Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Boletín médico del Hospital Infantil de México
versión impresa ISSN 1665-1146
Bol. Med. Hosp. Infant. Mex. vol.71 no.1 México ene./feb. 2014
Artículo de investigación
Aerobic actinomycetes that masquerade as pulmonary tuberculosis
Carlos Franco-Paredes
Hospital Infantil de México Federico Gómez, Mexico City, Mexico; Infectious Diseases Department, Phoebe Putney Memorial Hospital, Albany, GA, USA.
E-mail address:
carlos.franco.paredes@gmail.com
Received 2 November 2013
Accepted 12 December 2013
Abstract
Background: There is an increasing recognition of organisms in the order Actinomycetales including Nocardia sp. causing lung infections that mimic pulmonary tuberculosis or fungal pneumonias.
Methods: We retrospectively evaluated a cohort of patients in the southeastern United States in whom a presumptive diagnosis of pulmonary tuberculosis was initially entertained but who eventually were found to have infection caused by Rhodococcus sp. or Tsukamurella sp.
Results: Among a cohort of 52 individuals diagnosed as case suspects for pulmonary tuberculosis, we identified six patients who were infected with either Rhodococcus sp. or Tsukamurella sp. Of these six patients, two had co-infection with Mycobacterium tuberculosis.
Conclusions: Infection with aerobic actinomycetes may mimic pulmonary tuberculosis or may cause concomitant disease in patients with pulmonary tuberculosis.
Keywords: Rhodococcus equi; Tsukamurella sp.; Tuberculosis; Mycobacterium tuberculosis; Tuberculosis-like pneumonias.
Introduction
Pulmonary cavities are caused by tissue necrosis that leads to the exclusion of a portion of the pulmonary parenchyma via the bronchial tree.1,2 In general, the differential diagnosis of pneumonitis with cavitations includes infectious and noninfectious causes (Table 1). The infectious causes include bacteria such as community-associated methicillin-resistant Staphylococcus aureus,3 Actinomyces or Nocardia asteroids,1,2 Rhodococcus equi,4 Pseudomonas aeruginosa,1 melioidosis,2 polymicrobial necrotizing pneumonias or lung abscesses;2 mycobacteria including Mycobacterium tuberculosis5 or nontuberculous mycobacteria such as Mycobacterium kansasiii,6 Mycobacterium avium-intracellulare and others;7 fungi Aspergillus fumigatus,8 Histoplasma capsulatum,9 Cryptococcus neoformans or Cryptococcus gatti,10,11 Blastomyces dermatitidis,12 Coccidiodes immitis,13 Penicillium2 and others; and parasites Paragonimus westermani or cystic echinococcosis.2,14 Other infectious causes include septic emboli to the lung (right-sided endocarditis, Lemierre's syndrome).2 Among the non-infectious causes, the most salient etiologies include autoimmune diseases such as granulomatosis with polyangitis,15 rheumatoid arthritis,2 sarcoidosis,2 ankylosing spondylitis,16 pulmonary contusion and pulmonary infarction.2 In addition, bronchogenic carcinomas or metastatic neoplasms to the lung may develop cavities or a bronchogenic carcinoma may lead to postobstructive pneumonia with secondary cavitation.2 Thick wall cavities tend to be associated with malignancies.1,2
Pulmonary tuberculosis (TB), which is often associated with cavitary disease, continues to be a significant cause of morbidity and mortality in the U.S.17 Whereas the overall rates have declined to 3.4/100,000 persons in 2011, some specific geographic areas continue to be "hotspots" of TB transmission. In the U.S. state of Georgia, the number of tuberculosis cases (pulmonary and extrapulmonary) continues imposing substantial burden of disease in some districts.17 Interestingly, during the clinical evaluation of these cases, it has become evident that some cases suspected to be pulmonary TB from a clinical and radiographic evaluation were found as having infection due to aerobic actinomycetes such as Nocardia, Rhodococcus, or Tsukamurella. Recent reports confirm our findings.18-21
The Actinomycetales group of bacterial pathogens includes phylogenetically diverse but morphologically similar aerobic and anaerobic actinomycetes such as Actinomyces, Rothia, Nocardia, Williamsia, Gordonia, Tsukamurella, and Rhodococcus.18,19 This group of pathogens can cause human and veterinary disease leading to clinical syndromes involving the lung, bone and joints, soft tissue, and central nervous system similar to some fungal and mycobacterial infections.18,21 Indeed, lung infections with these organisms can produce cavitary disease manifesting with productive cough for many weeks, hemoptysis, fever, night sweats, weight loss and malaise, a clinical picture of pulmonary tuberculosis. These organisms may not only mimic pulmonary TB according to their clinical and radiographic features but they may also appear as acid-fast bacilli in sputum specimens.19
In this study we were interested in conducting a retrospective assessment of clinical cases of suspected pulmonary TB and in whom aerobic actinomycetes pathogens were isolated during the clinical investigation of cases of pulmonary TB. The association of Nocardia sp. and Actinomyces sp. with tuberculosis-like pneumonias has been previously reported.2,18,21 However, there is an increasing awareness of other members of the group of Actinomycetales causing lung infections with clinical and imaging similarities to those with patients suffering from pulmonary TB.
Subjects and methods
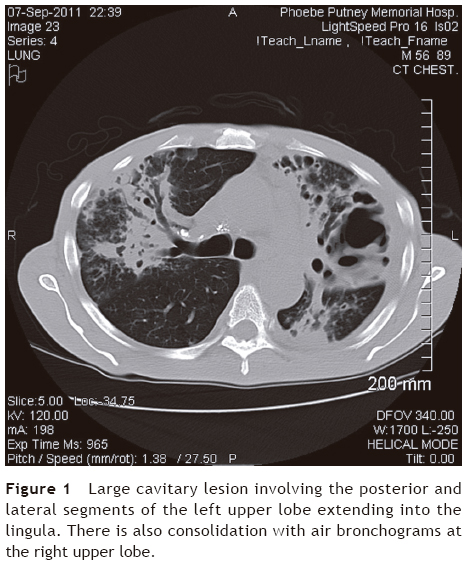
We conducted a descriptive, observational and retrospective review of clinical cases of suspected pulmonary TB (TB Class V as per the International Union Against Tuberculosis and Lung Disease) during a 2-year period (September 2011 to September 2013) in two large districts in southwest Georgia (U.S.) to identify the number of those infected with bacterial pathogens in the group of Actinomycetales. We were particularly interested in identifying those cases where Tsukamurella spp. or Rhodococcus spp. were isolated during the clinical workup of cases of presumed pulmonary TB. We defined cases according to their clinical presentation (fever, night sweats, productive cough for many weeks, malaise, weight loss, and hemoptysis) and chest radiograph or CT scan of the chest with findings compatible with pulmonary TB (cavitary lesions, endobronchial spread of pneumonia, or upper lobe pneumonias) (Fig. 1).
During the study period we included patients residing in the two largest districts in southwest Georgia (U.S.) evaluated by clinicians and referred to public health nurses. Microbiological studies were performed at local hospitals and also at the reference State Public Health Laboratory (Atlanta, GA). Standard microbiological methods for mycobacteria were used including acid-fast bacilli fluorochrome staining and high-performance liquid chromatography. Data were collected using an Excel spreadsheet and simple proportions were calculated. Patients were treated according to official tuberculosis treatment guidelines and those patients who were identified to have co-infection or infection with aerobic actinomycetes were referred for appropriate treatment by their clinician. The study was approved the Institutional Review Committee.
Results
During the study period we identified 52 individuals classified as case suspect for pulmonary TB case suspect (TB Class V) by clinical and radiographic criteria. A total of 43/52 (83%) cases were diagnosed as pulmonary TB. Among cases of pulmonary TB, 36/43 (84%) were confirmed microbiologically and 7/43 (16%) cases were classified as clinical cases (negative AFB staining and negative culture but having clinical response to the institution of anti-TB therapy for at least 6 weeks) (Table 2).
Other etiologies identified included nontuberculous mycobacteria: two cases of infection due to Mycobacterium kansasii and one case caused by Mycobacterium abscessus. We also identified 6/52 (12%) patients who were considered having disease produced by aerobic actinomycetes. Radiographic imaging demonstrated cavitary pneumonias in all six patients. Of these, two cases were diagnosed as having Rhodococcus equi infection and four patients with infection due to Tsukamurella sp. Importantly, two cases had co-infection with Mycobacterium tuberculosis (one with Tsukamurella sp. and one with Rhodococcus equi).19 No cases of Gordonia sp. or Nocardia sp. were identified in this cohort of patients.
Among those diagnosed with aerobic actinomycetes infection, the median age was 41 years and all were male. None of the six cases had a history of travel outside the state of Georgia. We identified smoking history in 5/6 (83%) patients and one patient (17%) had diabetes. All were HIV-seronegative (HIV testing is routinely performed during the initial workup of individuals classified as having TB Class V). A history of cancer was also not elicited among these six patients. All cases resided in rural areas where agriculture is the main occupation. All patients received adequate medical therapy22 and eventually recovered clinically and microbiologically. The only deaths reported during the 6-month treatment period were the patients co-infected with Mycobacterium tuberculosis and Rhodococcus.19
Discussion
In the presence of effective treatment, treatment failure of pulmonary TB is considered with continued or recurrently positive cultures during the course of anti-TB therapy after 4 months of treatment.23-25 Probable reasons for delayed conversion or treatment failure in patients receiving appropriate regimens include nonadherence to medications, underlying drug resistance, development of de novo drug resistance, decreased absorption of drugs, laboratory error, and the fact that some individuals may respond slowly due to biological variation.19,23-25 Additionally, there is a growing recognition that other pathogens may share, to some degree, the acid-fastness property and that may mimic pulmonary TB from a clinical standpoint. In fact, some respiratory microorganisms other than Mycobacterium tuberculosis and the nontuberculous mycobacteria share this property including Nocardia spp., Rhodococcus spp., Tsukamurella sp. and Legionella micdadei. Thus, clinicians should be aware of the potential for misdiagnosis of these organisms with the tubercle bacilli due also to the similarities found among these pathogens in their clinical presentation and radiographic imaging.19
Aerobic actinomycetes may either co-infect or be the actual pathogen among those in whom pulmonary TB was initially suspected as demonstrated in our series of patients.18 We identified cases of Rhodococcus sp. and Tsukamurella sp. but no cases of Nocardia sp. or Gordonia sp. were identified. All patients presented with a tuberculosis-like clinical syndrome and chest radiograph with cavitary disease and features consistent with pulmonary TB as the initial clinical impression. As a group, these organisms exhibit staining patterns such as filamentous branching structures, which may fragment into rods or coccoid forms. This is one of the morphological reasons why acid-fast bacilli organisms such as Mycobacterium, Tsukamurella, and Rhodococcus may occasionally be confused even by experienced microbiologists by staining patterns.19,21,26 Therefore, filamentous branching may not always be present in cases of Rhodococcus, Gordonia, or Tsukamurella. In addition, difficulties in culturing and identifying aerobic actinomycetes are responsible for delayed or inappropriate diagnosis.26 Different from other reports,18,21 we noted that most cases were patients who had a long-standing history of smoking and who live in rural agricultural-based settings. Only one patient had diabetes mellitus but no other comorbidities were identified.
Rhodococcus equi is an aerobic generally nonmotile Gram-positive rod that looks coccoid on solid media and in tissue.18,27 R. equi is preferentially a zoonotic pathogen but occasionally can cause severe and often fatal disease in humans.27,28 The acid-fastness of this organism with a modified acid technique stems from the presence of mycolic acids in the cell wall. During the last two decades, increases in the prevalence of R. equi disease coincide with the pandemic of HIV/AIDS and the use of highly immunosuppressive drugs used for transplantation, autoimmune diseases, and cancer chemotherapy.29 Aerosols and dust particles seem to be the predominate mode of acquisition. Frequently there is a history of contact with soil from horse farms or farming environments.4 The characteristic pathological findings associated with infection due to R. equi include the presence of malakoplakia, which is characterized by a dense infiltration of foamy histiocytes with intracellular coccobacilli and scattered concentric basophilic inclusions termed Michaelis-Gutmann bodies.27-29 R. equi infection manifests primarily as pulmonary disease but some may develop extrapulmonary infection (bacteremia, skin and soft tissue infections, mesenteric adenitis, and others).4 Rhodococcus spp. infection affects immunocompromised individuals and pulmonary disease is present in ~80% of cases with the most common radiographic findings being cavitary pneumonia located in the upper lobes. This is the reason for the potential misdiagnosis between R. equi and M. tuberculosis.19,29 Tsukamurella sp. such as T. paurometabola, but more frequently T. pulmonis and T. tyrosinosolvens, have been associated with lung infections.18 These veterinary and human pathogens are catalase-positive, gram-positive nonmycelium forming rods that can be identified as staining mildly to substantially with acid-fast staining.18,19
Previous studies demonstrated that bacterial pathogens in the Actinomycetales group, particularly Nocardia spp. and Actinomyces spp., may cause tuberculosis-like pneumonias. In our experience, we conclude that aerobic actinomycetes such as Gordonia, Rhodococcus, and Tsukamurella are also increasingly recognized clinical pathogens that may masquerade as pulmonary TB in patients with or in many cases without any identifiable immune deficiency.
Funding
None.
Conflict of interest
The authors declare no conflict of interest of any nature.
References
1. Gallant JE, Ko AH. Cavitary pulmonary lesions in patients infected with human immunodeficiency virus. Clin Infect Dis. 1996;22:671-82. [ Links ]
2. Gadkowski LB, Stout JE. Cavitary pulmonary disease. Clin Microbiol Rev. 2008;21:305-33. [ Links ]
3. Hidron AI, Low CE, Honig EG, Blumberg HM. Emergence of community-acquired methicillin-resistant Staphylococcus aureus strain USA300 as a cause of necrotizing community-onset pneumonia. Lancet Infect Dis. 2009;9:384-92. [ Links ]
4. Yamschikov AV, Schuetz A, Lyon MG. Rhodococcus equi infection. Lancet Infect Dis. 2010;10:350-9. [ Links ]
5. Davis JM, Ramakrishman L. The role of the granuloma in expansion and dissemination of early tuberculosis infection. Cell. 2009;136:37-49. [ Links ]
6. Wallace RJ Jr, Glassroth J, Griffith DE, Olivier KN, Cook JL, Gordin F, et al. Diagnosis and treatment of disease caused by nontuberculous mycobacteria. Am J Respir Crit Care Med. 1997;156(2 pt 2):S1-S25. [ Links ]
7. Griffith DE, Girard WM, Wallace RJ Jr. Clinical features of pulmonary disease caused by rapidly growing mycobacteria. An analysis of 154 patients. Am Rev Respir Dis. 1993;147:1271-8. [ Links ]
8. Walsh TJ, Anaissie EJ, Denning DW, Herbrecht R, Kontoyiannis DP, Marr KA, et al. Treatment of aspergillosis: clinical practice guidelines of the Infectious Diseases Society of America. Clin Infect Dis. 2008;46:327-60. [ Links ]
9. Wheat JL, Freifeld AG, Kleinman MB, Baddley JW, McKinsey DS, Loyd JE, et al. Clinical practice guidelines for the management of patients with histoplasmosis: 2007 update by the Infectious Disease Society of America. Clin Infect Dis. 2007;45:807-25. [ Links ]
10. Perfect JR, Dismukes WE, Dromer F, Goldman DL, Graybill JR, Hamill RJ, et al. Clinical practice guidelines for the management of cryptococcal disease: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis. 2010;50:291-322. [ Links ]
11. Harris JR, Lockhart SR, Debess E, Marsden-Haug N, Goldoft M, Wohrle R, et al. Cryptococcus gatti in the United States: clinical aspects of infection with an emerging pathogen. Clin Infect Dis. 2011;53:1188-95. [ Links ]
12. Chapman SW, Dismukes WE, Proia LA, Bradsher RW, Pappas PG, Threlkeld MG, et al. Clinical practice guidelines for the management of blastomycosis: 2008 update by the Infectious Diseases Society of America. Clin Infect Dis. 2008;46:1801-12. [ Links ]
13. Galgiani JN, Ampel NM, Blair JE, Catanzaro A, Johnson RH, Stevens DA, et al. Coccidioidomycosis. Clin Infect Dis. 2005;41:1217-23. [ Links ]
14. Martínez S, Restrepo CS, Carrillo JA, Betancourt SL, Franguet T, Varón C, et al. Thoracic manifestations of tropical parasitic infections: a pictorial review. Radiographics. 2005;25:135-55. [ Links ]
15. Bacon PA. The spectrum of Wegener's granulomatosis and disease relapse. N Engl J Med. 2005;352:330-2. [ Links ]
16. Franco C, Jurado R. Photo quiz. Diagnosis: apical fibrocavitary disease associated with ankylosing spondylitis. Clin Infect Dis. 2001;32:1062-103. [ Links ]
17. Centers for Disease Control and Prevention. Reported Tuberculosis in the United States 2011. Atlanta, GA: U.S. Department of Health and Human Services, CDC; 2012. [ Links ]
18. Savini V, Fazii P, Favaro M, Astolfi D, Polilli E, Pompilio A, et al. Tuberculosis-like pneumonias by the aerobic actinomycetes Rhodococcus, Tsukamurella, and Gordonia. Microbes Infect. 2012;14:401-10. [ Links ]
19. Franco-Paredes C, Ray S. Causes of persistent acid-fast positive smears in pulmonary tuberculosis. Am J Med. 2012;125:e3-e4. [ Links ]
20. Gupta M, Prasad D, Khara HS, Alcid D. A rubber-degrading organism growing from a human body. Int J Infect Dis. 2010;14:e75-e76. [ Links ]
21. Sullivan DC, Chapman SW. Bacteria that masquerade as fungi: actinomycosis/nocardia. Proc Am Thorac Soc. 2010;7:216-21. [ Links ]
22. Blumberg HM, Leonard MK Jr, Jasmer RM. Update on the treatment of tuberculosis and latent tuberculosis infection. JAMA. 2005;293:2776-84. [ Links ]
23. Al-Moamary MS, Black W, Bessuille E, Elwood RK, Vedal S. The significance of the persistent presence of acid-fast bacilli in sputum smears in pulmonary tuberculosis. Chest. 1999;116:726-31. [ Links ]
24. Lee JS, Kim EC, Joo SI, Lee SM, Yoo CG, Kim YW, et al. The incidence and clinical implication of sputum with positive acid-fast bacilli smear but negative mycobacterial culture in a tertiary referral hospital in South Korea. J Korean Med Sci. 2008;23:767-71. [ Links ]
25. Vidal R, Martin-Casabona N, Juan A, Falgueras T, Miravitlles M. Incidence and significance of acid-fast bacilli in sputum smears at the end of antituberculosis treatment. Chest. 1996;109:1562-5. [ Links ]
26. Franco-Paredes C. The reply. Am J Med. 2013;126:e17. [ Links ]
27. Prescott JF. Rhodococcus equi: an animal and human pathogen. Clin Microbiol Rev. 1991;4:20-34. [ Links ]
28. Weinstock DM, Brown AE. Rhodococcus equi: an emerging pathogen. Clin Infect Dis. 2002;34:1379-85. [ Links ]
29. Mistry NF, Dholakia Y, D'Souza DT, Taylor M, Hoffner S, Birdi TJ. Rhodococcus and Mycobacterium tuberculosis: masquerade or mixed infection. Int J Tuberc Lung Dis. 2006;10:351-3. [ Links ]