Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Boletín médico del Hospital Infantil de México
versión impresa ISSN 1665-1146
Bol. Med. Hosp. Infant. Mex. vol.68 no.2 México mar./abr. 2011
Artículo
Bridging the innovation gap for neglected tropical diseases in México: capacity building for the development of a new generation of antipoverty vaccines
Maria Elena Bottazzi,1 Eric Dumonteil,2 Jesus G. Valenzuela,3 Miguel Betancourt–Cravioto,4 Roberto Tapia–Conyer,5 and Peter J. Hotez1,6
1 Department of Microbiology, Immunology and Tropical Medicine, George Washington University, Washington, D.C., USA
2 Laboratorio de Parasitology C.I.R., Universidad Autónoma de Yucatán, Mérida, Yucatán, México
3 Vector Molecular Biology Section, Laboratory of Malaria and Vector Research, NIAID, NIH, Rockville, Maryland, USA
4 Director de Soluciones Globales, Instituto Carlos Slim de la Salud, México, D.F., México
5 Facultad de Medicina, Universidad Nacional Autónoma de México, Dirección General, Instituto Carlos Slim de la Salud, México D.F., México
6 Sabin Vaccine Institute, Washington, D.C., USA
Correspondence:
Maria Elena Bottazzi,
PhD Department of Microbiology, Immunology and Tropical Medicine
E–mail: mtmmeb@gwumc.edu
Received for publication: 1–24–11
Accepted for publication: 1–27–11
Abstract
The neglected tropical diseases (NTDs) represent a group of chronic parasitic and related infections that promote poverty because of their impact on child development, pregnancy, and worker productivity. The estimated 20 million Mexicans who live below the poverty line suffer disproportionately from a high prevalence of neglected tropical diseases such as amebiasis, Chagas disease, dengue, leishmaniasis, soil–transmitted helminth infections, trachoma, and vivax malaria. However, because the NTDs occur predominantly among the poor, new industrial and financial models are required to establish innovative technologies to address these conditions in Mexico and elsewhere in Latin America. In response, the Slim Initiative for Antipoverty Vaccine Development was established to foster a public/private partnership between key academic, government, and industrial institutions in the U.S. and Mexico. Initial emphasis will be placed on developing new vaccines for Chagas disease and leishmaniasis, two of the highest burden NTDs in Mexico and Mesoamerica.
Key words: public/private partnerships, vaccines, product development, technology transfer, vaccine manufacture.
INTRODUCTION
Today in the Latin American and Caribbean región an estimated 40% of the population lives in poverty with more than 100 million people living on less than US$2 per day.1 Generally speaking, Latin America's "bottom one hundred million" live as subsistence farmers in rural areas and urban slums. Almost all of this region's most impoverished people suffer from one or more neglected tropical diseases (NTDs), generally defined as chronic parasitic or related infectious diseases that promote poverty because of their impact on child growth and development, pregnancy, and worker productivity.2 Some estímate that in Latin America the burden of disease resulting from the NTDs may exceed that of HIV/AIDS.1 Simultaneously, the NTDs represent important, yet often unrecognized, reasons why the poorest people in the Americas cannot escape poverty.
Mexico and the NTDs
The estimated 112 million people living in Mexico also suffer from their share of poverty and NTDs. The nation is ranked 56 in terms of its human development index (HDI), an HDI that is roughly equivalent to that of Panama, Malaysia, Trinidad, and Tobago.3 However, this level of development is highly maldistributed, with 47% of Mexico's population living below the poverty line as of 20044 and 3.3% (approximately 3.7 million Mexicans) living on wages lower than US$2 per day.5 This bottom 3.7 million people also disproportionately suffer from NTDs, especially in the southern most impoverished Mexican states. In Table 1 we show the total cumulative number of cases of the major infectious diseases identified in the past 10 years (2000–2010) as reported by the Ministry of Health in Mexico. However, with few exceptions, the actual number of persons infected with an NTD is greatly underestimated. Universally, NTDs are considered forgotten diseases among forgotten people.6
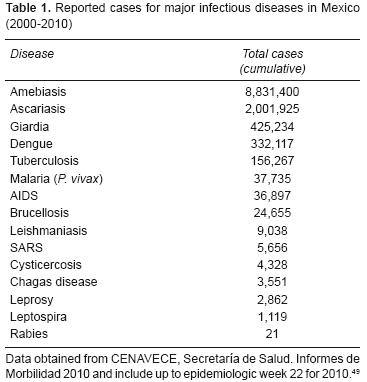
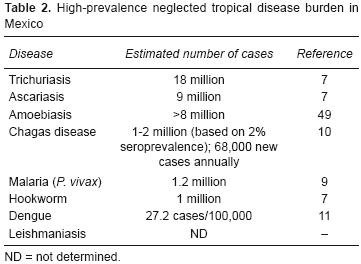
Alternative estimates for the prevalence of several NTDs in Mexico (Table 2) indicate that almost 20 million people are infected with one or more soil–transmitted helminth infections, i.e., trichuriasis (Trichuris trichiura whipworm infection), ascariasis (Ascaris lumbricoides roundworm infection), or hookworm infection caused predominantly by Necator americanus,1,1 an important figure given that some studies have shown that chronic hookworm infection in childhood reduces future wage–earning by 40% or more.2,8 In addition, 1.2 million Mexicans are infected with Plasmodium vivax malaria9 and, based on 2% seroprevalence estimates among prospective blood donors, at least two million suffer from Chagas disease, a disease that can cause serious and debilitating heart disease. However, some estimates suggest that as many as 6 million or more persons may be infected nationwide, whereas in some southern Mexican states more than 10% of the population may be infected.10 In addition, dengue fever and its dreaded complication, dengue hemorrhagic fever, are emerging in Mexico11 especially among people who live in poor–quality dwellings with poor sanitary conditions,12 a risk factor common to both dengue and Chagas disease. In addition, leishmaniasis remains endemic in Mexico, particularly in the south–southeast areas of the country and is unfortunately underreported. Other NTDs such as trachoma, cysticercosis, and onchocerciasis are also concentrated in areas near the states of Chiapas and Oaxaca.1
The Mexican population who live below the poverty line4,5 have a fundamental right to access not only essential medicines but innovation as well. However, because there is inadequate financial remuneration for new drugs, diagnostics, and vaccines for NTDs, most multinational companies have shied away from investing in research and development for these products.13 Nonetheless, new vaccines for the high prevalence NTDs, including hookworm infection, dengue, vivax malaria, Chagas disease, and leishmaniasis, could become new and important life–saving technologies. Because of the propensity of NTDs to promote poverty, vaccines to combat such NTDs also represent important economic development interventions and are even sometimes referred to as "antipoverty vaccines."14 However, in the absence of obvious financial incentives, almost all of the antipoverty vaccines are being developed either in academic laboratories or through new non–profit product development partnerships.13,15 Here we outline steps for the development of antipoverty vaccines for Mexico's leading NTDs, with initial emphasis on Chagas disease and leishmaniasis.
Chagas Disease and Leishmaniasis
Chagas disease is a parasitic infection caused by the protozoa Trypanosoma cruzi, which is transmitted to humans by infected triatomine kissing bugs. In the acute stages, symptoms include fever, swollen glands, liver and spleen and, in acute cases, meningitis and heart enlargement, which can lead to death. Between 30 and 40 percent of cases develop into a chronic stage of the disease, which leads to cardiac complications and dilation of the esophagus and the intestine.16 Cardiac manifestations from chronic chagasic cardiomyopathy are especially debilitating and can lead to sudden death. Chagas disease is one of the most common parasitic diseases in Latin America occurring in poor rural areas and, increasingly, in many new urban and periurban areas especially where the quality of dwellings is poor and kissing bugs may flourish. There are approximately eight to nine million existing cases of the disease and approximately 50,000 new cases annually, an incidence that is between five and ten times greater than malaria. Finally, Chagas disease causes an estimated 20,000 deaths in the region and its economic impact represents a significant percentage of the region's external debt.1
Leishmaniasis is a vector–borne parasitic infection caused by the protozoa of the genus Leishmania and transmitted to humans by the bite of infected sandflies. There are two forms of the disease: cutaneous leishmaniasis (ulcers at the site of the bite) and visceral leishmaniasis (where organs such as liver and spleen are affected and symptoms include fever, weight loss, grayish skin, swollen glands, anemia and loss of white blood cells). The cutaneous facial lesions are especially stigmatizing for women,17 whereas the visceral form can lead to death. Leishmaniasis is a common disease with 60,000 new cases reported annually in Latin America, although the actual incidence is probably far higher. The most important determinants for the emergence of both new world zoonotic cutaneous leishmaniasis (ZCL) and zoonotic visceral leishmaniasis (ZVL) include poverty, urbanization, and human migration.17 In these impoverished urban and periurban settings, the cracked walls and damp earth floors, together with an absence of sanitation and inadequate garbage collection, create optimal sandfly breeding sites. The disease has become an important problem in densely settled slums and favelas where there is also close contact with infected canine reservoirs.18 With the exception of Brazil, surveillance systems in Latin America have been limited in their capacity to as–sess the true burden of ZVL.1
Burden and Distribution of Leishmaniasis and Chagas Disease in Mexico
Leishmaniasis and Chagas disease are two of the most frequently neglected diseases of poverty (Table 1). In the case of Chagas disease, the first cases were identified in Mexico in the 1930s19 but, since then, information on Chagas disease burden has been scarce and the importance of the disease has been largely ignored. Very few cases have been reported to the Ministry of Health of Mexico during the past 15 years, although there has been a large increase of cases reported in recent years (Figure 1A). This is mostly due to improvements in epidemiological surveillance and the decentralization of diagnostic activities to regional laboratories. Nonetheless, Chagas disease remains grossly underreported, as evidenced by an increasing number of serologic surveys.10,16 Conservative estimates derived from national surveys indicate that about 1–2% of blood donors in the Mexican population are seropositive for T. cruzi,20,21 which would correspond to at least 1–2 million persons exposed to the parasite in the country, with about 70,000 new cases annually.22 However, several studies have identified much higher seroprevalence levels in specific regions, giving a national average of up to 5.9% of seropositivity,10 which would correspond to up to nearly 6.5 million infected patients. The southern states of Chiapas, Oaxaca, Puebla, Veracruz and Yucatan are among the most affected areas (where the prevalence may exceed 10%), although cases have been reported in most of the country.10,16 In fact, Mexico hosts one of the most diverse triatomine populations, with 39 species documented in the country, at least 21 of which have been found to be infected by T. cruzi, rendering them potential vectors of Chagas disease.10,24 The most important Chagas vectors belong to the phyllosoma and dimidiata species complexes within the Triatome phylum.23–25 Nonetheless, no national vector control program has been implemented to date for the prevention of Chagas disease in Mexico. However, blood donor screening is mandatory and cover–age has been steadily increasing.21,26
Leishmaniasis was first reported in Mexico in the early 20th century with Seidelin's observations of what is referred to as the Chiclero's ulcer on gum workers. For the past 15 years, leishmaniasis case reporting by the Ministry of Health of Mexico has been steady, with an average notification of slightly over 900 cases/year (Figure 1B), due to somewhat better epidemiological surveillance. However, underreporting of cases is also very likely and there may be up to several thousand cases/year. Similar to Chagas disease, leishmaniasis is mostly present in the southern states of Mexico, although it appears to be much more clustered around limited foci of infection. The state of Tabasco hosts the most active transmission cluster, and other foci have been described in Veracruz, Oaxaca, Chiapas, Campeche, Quintana Roo, and Nayarit. The entire spectrum of disease has been observed in Mexico, ranging from localized cutaneous leishmaniasis27 to mucosal and diffuse leishmaniasis28 as well as visceral leishmaniasis.29 Leishmania (Leishmania) mexicana is the most frequent causing cutaneous and diffuse cutaneous disease, followed by L. (Viannia) braziliensis, and cases of visceral leishmaniasis presumably by L. infantum chagasi (L. donovani complex) were also reported.29–32 Studies of sand flies species in Mexico, particularly species responsible for the transmission of leishmaniasis, showed that Lutzomyia olmeca olmeca is the principal vector of Leishmania mexicana in the southern states of Mexico.33 More recently, other sand fly species in addition to Lu. olmeca have been incriminated as vector for L. mexicana in the Yucatan state including Lu. ylephiletor, Lu. shannoni, Lu. cruciata, and Lu. panamensis.34 The presence of two potential vectors, Lu. longipalpis and Lu. evansi, for the transmission of visceral leishmaniasis in the state of Chiapas were reported.35
Approaches for the Development of New Control Tools against Neglected Tropical Diseases
In 2001, the World Health Organization Commission on Macroeconomics and Health reported that it is essential to invest in health to be able to achieve poverty reduction and promote economic development in middle– to low–income countries.36 Partnerships with high–income countries were identified as essential to advance health innovations such as the development of new products (vaccines, drugs, diagnostics and devices), new processes (novel ways to produce or administer health products or services) and new health policies and strategies. Since then, such efforts have been primarily spearheaded by so–called product development partnerships (PDPs). PDPs are nonprofit organizations that use industry practices for product development, control and quality assurance. The model used by these PDPs consists of applying strategies to bridge the development and innovation gaps. Through the establishment of links between the public and private sectors and encouraging the endemic countries to play a role, PDPs have achieved the advancement of knowledge and learning while improving best practices and reducing the costs of advancing a specific technology.37,38
One PDP in particular called "Sabin Vaccine Development" (a major program of the Sabin Vaccine Institute39), was established specifically to address the development of vaccines against NTDs such as hookworm, schistosomiasis and others.13,15 In the last decade it has built a sustainable infrastructure and capacity for research, development, and scale–up of vaccine candidates, operating primarily with laboratories based in an academic institution.13,15,40 Sabin Vaccine Development learned how to cross the so–called "valley of death" between initial discovery and pilot manufacture and clinical testing, and created processes and procedures needed to advance a vaccine target from the laboratory scale to mid–scale manufacturing and transfer the information to large–scale manufacturing facilities. The success of Sabin Vaccine Development has been achieved through its strong collaborations and partnerships established between academic institutions in the U.S. and public entities in innovative development countries such as Brazil (i.e., public sector vaccine manufacturers).13,15,40 Sabin Vaccine Development is now well poised to use a similar approach and establish links with public and academic entities in Mexico for the development of new control tools against Chagas disease and leishmaniasis.
Current Biotechnology and Research and Development Capabilities in Mexico
Mexico is one of the top 25 countries in the world ranked to have innovative efficiency as measured by the number of U.S. patents per GDP/capita (gross domestic product/ capita).38 In addition, a study conducted by the Global Forum for Health Research in 2007 noted that in 2005 Mexico's 12 National Institutes for Health spent approximately US $585,981,972 for health research for national needs.41 For these reasons, Mexico, together with nations such as Brazil, India, and China, belongs to a group of designated "innovative developing countries" (IDCs) referring to nations that are modest economic performers but have overachieved in terms of their research and development capabilities as evidenced by their number of papers in international scientific journals and patents.38 As noted by the Commission for the Future of Vaccines in Latin America (COFVAL),42 we believe that Mexico has all of the necessary tools ready for the advancement of public–private collaborations in the areas of research, development and manufacturing.
Furthermore, Mexico has approximately 98 institutions dedicated to various aspects of biotechnological research and development (R&D)43,44 and according to the Mexican National System of Investigators (SNI), between 1995 and 2005, the number of researchers registered in the areas of 1) biology and chemistry, 2) medicine and health sciences and 3) biotechnology and agricultural sciences nearly doubled.43 Presently there are more that 1,250 investigators dedicated to biotechnology R&D alone. Finally, the level and quality of publications also have increased notably in the last decade in Mexico. Publications in the area of microbiology are cited 39% higher than the world average, and plant and animal science publications are cited 42% higher than average.43,44 These indicators certainly provide evidence of the growing scientific and R&D capability in Mexico.
Strengthening Vaccine Development Capacity for NTDs in Mexico
Governmental scientific centers and universities continue to concentrate their efforts on biotechnological research and the training and development of a new cadre of young investigators.44 Most of the financial support for these institutions comes from entities such as the National Council for Science and Technology (CONACyT). There is, in addition, a small number of private initiatives such as the Carlos Slim Health Institute whose main mission is to enhance and augment biotechnological production in Mexico.
The Carlos Slim Health Institute was created in 2007 for the development and funding of programs aimed at contributing to the solutions for the main public health problems in Latin America and the Caribbean, guided by a spirit of innovation and with a clear concern for the most underserved populations.
Through its programs, the Institute works to meet the demand for services by helping National and State Health Departments in the creation of primary health care units. The Institute also facilitates access to health resources via communication technology. Through leading–edge educational communication activities it encourages individuals and populations to participate in maintaining their own health. Finally, the Institute provides support to other nonprofit organizations that promote the well–being of the population by encouraging them to favor their own physical health and to maintain healthy households as well as natural community environments.45
One of the methods implemented by the Carlos Slim Health Institute in order to achieve its goals is to establish alliances with strategic partners such as academic institutions and industry, with the aim of sharing economic and scientific risks for the development of social patents and public goods for the benefit of the wider population.
Future Perspective
Guided by its mission together with recommendations made by COFVAL, the Carlos Slim Health Institute launched "The Slim Initiative for Antipoverty Vaccine Development." This new initiative will promote the establishment of a biotechnology consortium, which will include several institutions in Mexico such as the Carlos Slim Health Institute, the Autonomous University of Yucatan and its Regional Research Center "Dr. Hideyo Noguchi," the Center for Research and Advanced Studies (CINVESTAV) and Biologics and Reagents Laboratories of Mexico (BIRMEX) and in the U.S. including the U.S. National Institute of Allergy and Infectious Diseases of the National Institutes of Health, the George Washington University, and Sabin Vaccine Development. Through the establishment of a product development public–private partnership, an initial focus will be on accelerating the development of new vaccines against Chagas disease and leishmaniasis. Ultimately, the goal will be to strengthen the capacity to translate scientific research into the development, manufacture and future distribution of biologics against NTDs through technology transfer to the Mexican institutions mentioned above. This will allow for the future production of vaccines on a large scale, which will benefit impoverished communities of Mexico and Latin America.
This new partnership proposes to enhance and strengthen capacity for translating bench discovery into development by employing an innovative system for antigen selection and pre–clinical evaluation. Sabin Vaccine Development has developed a scoring system that incorporates essential criteria for the rigorous selection of vaccine candidate antigens that show promise of being highly efficacious.46,47 For instance, the selection of vaccine candidates suitable for the development of a human hookworm vaccine included evaluation of candidates using animal models to determine specific endpoints such as blood loss, worm burdens and fecal egg counts. In addition, to aid in the analysis of the results obtained from the challenge animal models, molecules were evaluated based on their immunoepidemiological profile and whether targets were of known function/structure of protein.46
After targets are ranked and selected, early–stage process development will initiate to determine the type of vaccine expression systems that would be feasible and that ultimately would indicate the type of manufacturing process needed for mid– to large–scale production. These could include development of DNA as well as recombinant protein–based vaccine. Vaccine candidates will be assessed at the 10 liter fermentation scale, purified using low–cost resins and materials and then technology transferred to biotechnology partners in Mexico with the future goal to manufacture a final pilot lot. Ultimately, all of these processes would lead into entering the regulatory path for GMP manufacture, toxicology testing and clinical testing. A schematic of the proposed stages of a product development pipeline for the Slim Initiative for Antipoverty Vaccine Development is presented in Figure 2.
Influencing Vaccine Development Innovation in Mexico
In order for this new initiative to be successful and to establish a sustainable model for the development of vaccines for other diseases of poverty in Mexico and Central America, a different approach will be necessary (Table 3).
The model to be utilized by the Slim Initiative for Antipoverty Vaccine Development will be based on combining the expertise of industry–experienced researchers with knowledge of biotechnology and pharmaceutical procedures and practices and with traditional scientists from the academic and nonprofit sector. Continuous communications, transparency and a series of criteria for organizational innovation need to be applied among these alliances, and the collaborating scientists must embrace an attitude of persistence and creativity.
Furthermore, these collaborations will also focus on advancing vaccines specifically needed by the region. This will require that the technology and processes developed are affordable and easily transferable. This will also create incentives for local manufacturers through the partnership such as the feasibility of local governmental subsidies, tax credits or fast track regulatory processes. For example, we envision that if the diseases addressed by the initiative meet a significant unmet regional medical need, WHO would potentially allow submission of a regulatory file to its prequalification system as was done for the vaccine against pneumoccocus.48
Finally, many studies have validated the concept of using vaccines for both protection and therapy against these protozoan parasites in a variety of animal models. The challenge now will be to translate what has been achieved in these models into human vaccines of comparable efficacy. Also, genomemining and new antigen discovery strategies may provide new tools for a more rational search of novel vaccine candidates.
Acknowledgments
We wish to thank Ms. Violetta Yevstigneyeva for assistance in compiling literature for this manuscript. No funding was received for the compilation of any information submitted in this review and there are no economic or employment relationships of the authors of this paper from companies or institutions that may be favored with the information in this article.
REFERENCES
1. Hotez PJ, Bottazzi ME, Franco–Paredes C, Ault SK, Periago MR. The neglected tropical diseases of Latin America and the Caribbean: a review of disease burden and distribution and a roadmap for control and elimination. PLoS Negl Trop Dis 2008;2:e300. [ Links ]
2. Hotez PJ, Fenwick A, Savioli L, Molyneux DH. Rescuing the bottom billion through neglected tropical disease control. Lancet 2009;373:1570–1575. [ Links ]
3. International Human Development Indicators. Mexico. Available at: http://hdrstats.undp.org/en/countries/profiles/MEX.html (accessed December 31, 2010). [ Links ]
4. The World Bank. Mexico. Available at: http://data.worldbank.org/country/mexico (accessed January 5, 2011). [ Links ]
5. The World Bank. Poverty gap. Available at: http://data.worldbank.org/indicator/SI.POV.GAP2 (accessed January 5, 2011). [ Links ]
6. Hotez PJ. Forgotten People, Forgotten Diseases: The Neglected Tropical Diseases and their Impact on Global Health and Development. Washington DC: ASM Press; 2008. p. 218. [ Links ]
7. De Silva N, Brooker S, Hotez P, Montresor A, Engels D, Savioll L. Soil transmitted helminth infections: updating the global picture. Trends Parasitol 2003;19:547–551. [ Links ]
8. Bleakley H. Disease and development: evidence from hookworm eradication in the American South. Q J Econ 2007;122:73–117. [ Links ]
9. World Health Organization. World Malaria Report 2010. Available at: http://whqlibdoc.who.int/publications/2010/9789241564106_eng.pdf. [ Links ]
10. Cruz–Reyes A, Pickering–López JM. Chagas disease in Mexico: an analysis of geographical distribution during the past 76 years—a review. Mem Inst Oswaldo Cruz 2006;101:345–354. [ Links ]
11. Gómez–Dantés H, Willoquet JR. Dengue in the Americas: challenges for prevention and control. Cad Saude Publica 2009;25(suppl 1):S19–S31. [ Links ]
12. Brunkard JM, Robles López JL, Ramirez J, Cifuentes E, Rothenberg SJ, Hunsperger EA, et al. Dengue fever seroprevalence and risk factors, Texas–Mexico border, 2004. Emerg Infect Dis 2007;13:1477–1483. [ Links ]
13. Bottazzi ME, Brown AS. Model for product development of vaccines against neglected tropical diseases: a vaccine against human hookworm. Expert Rev Vaccines 2008;7:1481–1492. [ Links ]
14. Hotez PJ, Ferris M. The antipoverty vaccines. Vaccine 2006;24:5787–5799. [ Links ]
15. Bottazzi ME, Miles AP, Diemert D, Hotez PJ. An ounce of prevention on a budget: a nonprofit approach to developing vaccines against neglected diseases. Expert Rev Vaccines 2006;5:189–198. [ Links ]
16. Dumonteil E. Update on Chagas' disease in Mexico. Salud Publica Mex 1999;41:322–327. [ Links ]
17. Alvar J, Yactayo S, Bern C. Leishmaniasis and poverty. Trends Parasitol 2006;22:552–557. [ Links ]
18. Bern C, Maguire JH, Alvar J. Complexities of assessing the disease burden attributable to leishmaniasis. PLoS Negl Trop Dis 2008;2:e313. [ Links ]
19. Flisser A, Velasco–Villa A, Martínez–Campos C, González–Domínguez F, Briseno–García B, García–Suárez R, et al. Infectious diseases in Mexico. A survey from 1995–2000. Arch Med Res 2002;33:343–350. [ Links ]
20. Velasco–Castrejón O, Valdespino JL, Tapia Conyer R, Salvatierra B, Guzmán Bracho C, Magos C, et al. Seroepidemiologia de la enfermedad de Chagas en Mexico. Salud Publica Mex 1992;34:186–196. [ Links ]
21. Guzmán–Bracho C, García–García L, Floriani–Verdugo J, Guerrero–Martínez S, Torres–Cosme M, Ramírez–Melgar C, et al. Riesgo de transmisión de Trypanosoma cruzi por transfusión de sangre en México. Rev Panam Salud Publica 1998;4:94–99. [ Links ]
22. Ramsey J, Ordonez R, Tello–López A, Phols JL, Sanchez V, Peterson AT. Actualidades sobre la epidemiología de la enfermedad de Chagas en México. In: Ramsey J, Tello–López A, Pohls JL, eds. Iniciativa para la Vigilancia y el Control de la Enfermedad de Chagas en la República Mexicana. Cuernavaca, Morelos: Instituto Nacional de Salud Pública; 2003. pp. 85–103. [ Links ]
23. Ibarra–Cerdeña CN, Sánchez–Cordero V, Townsend Peterson A, Ramsey JM. Ecology of North American Triatominae. Acta Trop 2009;110:178–186. [ Links ]
24. Bargues M D, Klisiowicz DR, González–Candelas F, Ramsey J, Monroy C, Ponce C, et al. Phylogeography and genetic variations of Triatoma dimidiata, the main Chagas disease vector in Central America, and its position within the genus Triatoma. PLoS Negl Trop Dis 2008;2:e233. [ Links ]
25. Dorn PL, Calderón C, Melgar S, Moguel B, Solórzano E, Dumonteil E, et al. Two distinct Triatoma dimidiata (Latreille, 1811) taxa are found in sympatry in Guatemala and Mexico. PLoS Negl Trop Dis 2009;3:e393. [ Links ]
26. Schmunis GA, Zicker F, Cruz JR, Cuchi P. Safety of blood supply for infectious diseases in Latin American countries, 1994–1997. Am J Trop Med Hyg 2001;65:924–930. [ Links ]
27. Andrade–Narváez FJ, Vargas–González A, Canto–Lara SB, Damián–Centeno AG. Clinical picture of cutaneous leishmaniases due to Leishmania (Leishmania) mexicana in the Yucatan peninsula, Mexico. Mem Inst Oswaldo Cruz 2001;96:163–167. [ Links ]
28. Velasco O, Savarino SJ, Walton BC, Gam AA, Neva FA. Diffuse cutaneous leishmaniasis in Mexico. Am J Trop Med Hyg 1989;41:280–288. [ Links ]
29. Monroy–Ostria A, Hernandez–Montes O, Barker DC. Aetiology of visceral leishmaniasis in Mexico. Acta Trop 2000;75:155–161. [ Links ]
30. Canto–Lara SB, Cardenas–Maruffo MF, Vargas–González A, Andrade–Narváez F. Isozyme characterization of Leishmania isolated from human cases with localized cutaneous leishmaniasis from the state of Campeche, Yucatan Peninsula, Mexico. Am J Trop Med Hyg 1998;58:444–447. [ Links ]
31. Canto–Lara SB, Van Wynsberghe NR, Vargas–González A, Ojeda–Farfán FF, Andrade–Narváez FJ. Use of monoclonal antibodies for the identification of Leishmania spp. isolated from humans and wild rodents in the State of Campeche, Mexico. Mem Inst Oswaldo Cruz 1999;94:305–309. [ Links ]
32. Hernandez–Montes O, Monroy–Ostria A, McCann S, Barker DC. Identification of Mexican Leishmania species by analysis of PCR amplified DNA. Acta Trop 1998;71:139–153. [ Links ]
33. Rebollar–Téllez EA, Ramírez–Fraire A, Andrade–Narváez FJ. A two years study on vectors of cutaneous leishmaniasis. Evidence for sylvatic transmission cycle in the state of Campeche, Mexico. Mem Inst Oswaldo Cruz 1996;91:555–560. [ Links ]
34. Pech–May A, Escobedo–Ortegón FJ, Berzunza–Cruz M, Rebollar–Téllez EA. Incrimination of four sandfly species previously unrecognized as vectors of Leishmania parasites in Mexico. Med Vet Entomol 2010;24:150–161. [ Links ]
35. Ibáñez–Bernal S, Rodríguez–Domínguez G, Gómez–Hernández CH, Ricardez–Esquinca JR. First record of Lutzomyia evansi (Nuñez–Tovar 1924) in Mexico (Diptera: Psychodidae, Phlebotominae). Mem Inst Oswaldo Cruz 2004;99:127–129. [ Links ]
36. Sachs JD. Macroeconomics and health: investing in health for economic development. Report of the Commission on Macroeconomics and Health to the World Health Organization. Newton, MA, USA: Digital Design Group; 2001. Available at: http://www.cid.harvard.edu/archive/cmh/cmhreport.pdf. [ Links ]
37. Mahoney RT, Morel CM. A Global Health Innovation System (GHIS). Innovation Strategy Today 2006;2:1–12. [ Links ]
38. Morel CM, Acharya T, Broun D, Dangi A, Elias C, Ganguly NK, et al. Health innovation networks to help developing countries address neglected diseases. Science 2005;309:401–404. [ Links ]
39. Sabin Vaccine Institute. Available at: http://www.sabin.org/ (accessed January 3, 2011). [ Links ]
40. Hotez PJ, Brown AS. Neglected tropical disease vaccines. Biologicals 2009;37:160–164. [ Links ]
41. Burke MA, de Francisco A, Matlin SA. Monitoring the Financial Flows of Health Research 2007: Behind the Global Numbers. Geneva: Global Forum for Health Research; 2008. [ Links ]
42. The Commission on the Future of Vaccines in Latin America. Strengthening vaccination policies in Latin America. México D.F., México: Instituto Carso de la Salud, A.C., Fundación Carlos Slim; 2008. [ Links ]
43. San Diego Dialogue, a division of University of California San Diego Extension. Borderless Biotech & Mexico's Emerging Life Sciences Industry. 2007. Available at: http://www.sandiegodialogue.org/pdfs/Borderless_Biotech.pdf. [ Links ]
44. Possani LD. The past, present, and future of biotechnology in Mexico. Nat Biotech 2003;21:582–583. [ Links ]
45. Instituto Carlos Slim de la Salud. Informe 2009. Mexico: 2010. Available at: http://www.carlosslim.com/pdf/icss_informe2009.pdf. [ Links ]
46. Loukas A, Bethony J, Brooker S, Hotez P. Hookworm vaccines: past, present, and future. Lancet Infect Dis 2006;6:733–741. [ Links ]
47. Hotez PJ, Bethony JM, Diemert DJ, Pearson M, Loukas A. Developing vaccines to combat hookworm infection and intestinal schistosomiasis. Nat Rev Microbiol 2010;8:814–826. [ Links ]
48. Taylor K, Nguyen A, Stephenne J. The need for new vaccines. Vaccine 2009;27(suppl 6):G3–G8. [ Links ]
49. Secretaría de Salud. Informes de Morbilidad 2010. Sistema Nacional de Vigilancia Epidemiológica. México D.F., 2010. Available at: http://www.dgepi.salud.gob.mx/anuario/html/anuarios.html. [ Links ]














