Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Archivos de cardiología de México
versión On-line ISSN 1665-1731versión impresa ISSN 1405-9940
Arch. Cardiol. Méx. vol.79 no.4 Ciudad de México oct./dic. 2009
Comunicaciones breves
Anomalous origin of the Left Coronary Artery from the Pulmonary Artery. Echocardiographic diagnosis
Origen anómalo de la arteria coronaria izquierda de la arteria pulmonar. Diagnóstico ecocardiográfico
Carlos Alva,1 Felipe David Gómez,2 Santiago Jiménez–Arteaga,2 Arturo Martínez–Sánchez, 2 José Ortegón–Cardeña,2 Lucelli Yánez,2 y Carlos Riera–Kinkel3
1 Director of Congenital Heart Disease Department, Centro Médico Nacional Siglo XXI.
2 Staff Member of Congenital Heart Disease Department.
3 Surgeon of Congenital Heart Disease Department.
Corresponding author:
Carlos Alva MD,
Director of Congenital Heart Disease Department,
Cardiology Hospital, Centro Médico Nacional Siglo XXI,
Cuauhtemoc Ave. 330, Col. Doctores,
Mexico City CP 06720, Mexico,
Phone: ++525555852443.
E–mail: echoca@yahoo.com
Received: October 22, 2008.
Accepted: April 07, 2009.
Abstract
The experience with echocardiographic diagnosis of five cases of Anomalous Origin of the Left Coronary Artery from the Pulmonary Artery (ALCAPA) is reviewed.
Material and Methods. all cases with a diagnosis of ALCAPA during a 10 year period were included.
Results. two age groups were clearly identified: infants and older patients. In the former, the echocardiographic findings included a dilated left ventricle with low ejection fraction (19% and 26%), mitral regurgitation, dilated right coronary artery and difficulties in identifying the origin of the left coronary artery. An 11–year–old asymptomatic boy, a 17–year–old young woman with dyspnea on effort and a 55–year–old woman with angina formed the older group. In these three cases, an abnormal upward flow was detected within the ventricular septum related to the collaterals and the inverse flow of the anterior descending artery. A reverse flow within the origin of the left coronary artery, probably related to an origin in the pulmonary artery, was observed. In all five cases the diagnosis was corroborated using selective right coronary artery angiography.
Conclusions: In the infants, the dilated left ventricle with impaired systolic function, mitral regurgitation and dilated right coronary artery make it necessary to discard the ALCAPA diagnosis. In the older group, even in asymptomatic patients, an abnormal ascending flow within the ventricular septum, combined with a dilated coronary artery, was the most reliable echocardiographic evidence for a diagnosis of ALCAPA.
Key words: ALCAPA; Pulmonary artery; Congenital anomalies; Coronary arteries; Echocardiography; Mexico.
Resumen
Se revisa la experiencia con el diagnóstico ecocardiográfico del origen anómalo de la arteria coronaria izquierda que nace en la arteria pulmonar (ALCAPA).
Material y Métodos. Se incluyeron todas las personas a quienes se les diagnosticó ALCAPA durante un periodo de 10 años.
Resultados. Se definieron dos grupos, el grupo de lactantes y el de enfermos mayores. En el primer grupo, los datos ecocardiográficos fueron dilatación del ventrículo izquierdo con fracción de expulsión disminuida (19% y 26%), insuficiencia mitral, dilatación de la arteria coronaria derecha y dificultad para identificar el origen de la coronaria izquierda. En el grupo de enfermos mayores se estudió a un niño asintomático de 11 años, una mujer de 17 años con disnea de esfuerzo y otra mujer de 55 años con angina. En estos tres casos, se observó flujo ascendente anormal en el tabique interventricular, relacionado con el flujo inverso de la descendente anterior a partir de colaterales, combinado con dilatación de la coronaria derecha. En todos los casos se corroboró el diagnóstico de ALCAPA mediante coronariografia selectiva.
Conclusiones. En los lactantes, la observación ecocardiográfica de dilatación del ventrículo izquierdo con función sistólica disminuida e insuficiencia mitral y coronaria derecha dilatada obliga a verificar si el diagnóstico es ALCAPA. En los enfermos de mayor edad, aun en pacientes asintomáticos, la observación de un flujo ascendente anormal en el tabique interventricular combinado con dilatación de la coronaria derecha fueron los datos característicos para diagnosticar ALCAPA.
Palabras clave: Origen anómalo de la arteria coronaria izquierda; ALCAPA; Anomalías congénitas coronarias; México.
Introduction
The anomalous origin of the left coronary artery from the pulmonary artery (ALCAPA), in contrast to the normal origin from the aorta, is the primary cause of myocardial ischemia in children. This fascinating congenital anomaly of the coronary arteries is characterized by cardiac failure and cardiogenic shock in small children. The mortality rate in untreated infants is 90%. However, in older patients and even in adults, it could be asymptomatic or only a minimal murmur can be detected. In adults, sudden death and severe arrhythmias have been previously described.1
ALCAPA was first described in 1886 by Brooks,2 then by Abbot in 1908,3 although a major interest emerged in this malformation arose in 1933, with the description of a girl with a myocardial infarction, corroborated in the autopsy findings, by Bland, Garland and White.4 Since then, many works have been published on improving diagnosis and treatment of this lesion.5–7 In this work, a 10 year experience of five cases with ALCAPA diagnosed by echocardiography is reported.
Materials and methods
From July 1998 to June 2008, five cases with ALCAPA were diagnosed using echocardiography and corroborated with coronary artery angiography. There were two patients under two years of age (three and 21 months old) and three patients 11,17 and 55 years of age, respectively. In all cases, the clinical file, the echocardiogram and cardiac catheterization were retrospectively analyzed looking for the most notable echocardiographic findings.
Results
The ALCAPA defect constitutes approximately 0.35% of all congenital heart diseases in our unit. The five patients can be clearly classified into two groups: the infant group with evident cardiac failure and the older patients, two of them asymptomatic, and one with dyspnea and angina on effort.
Infant group
It was interesting to see that the two infants were referred to our hospital with the diagnosis of dilated cardiomiopathy and symptoms of heart failure. In both patients a systolic mitral murmur was heard and both had a marked cardiomegaly. The electrocardiogram revealed anterior necrosis in one patient and anterolateral in the other. A dilated left ventricle, low ejection fraction and mitral regurgitation was observed by echocardiography in both (Figure 1a). In both patients it brought to our attention the dilated right coronary artery and the difficulty in identifying the origin of the left coronary artery (Figure 1b). The selective coronary artery angiogram revealed ALCAPA in both infants.

The first case, a 21 month old infant, with 19% ejection fraction, a surgical transfer of the coronary artery to the aorta was attempted. This patient died in surgery. In the second case the direct reimplantation procedure was successfully performed. Today, two years later, the patient is asymptomatic and the ejection fraction improved to 58% by echocardiography.
Older group
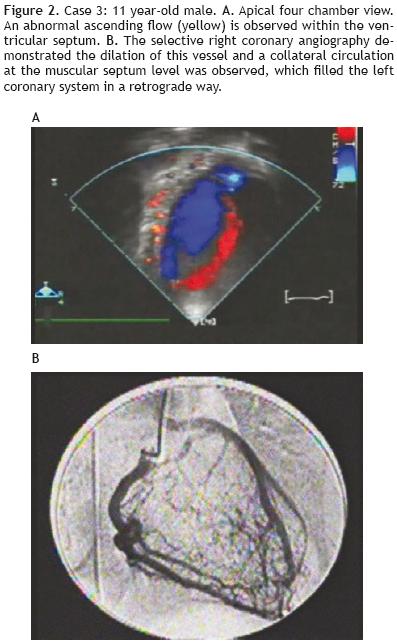
An asymptomatic 11 year–old male patient was referred to our unit for a grade I systolic cardiac murmur along the sternal border, when having a check–up, with no other findings during the physical exam. The electrocardiogram, chest x–ray and electrical stress test were normal. The echocardiogram revealed an abnormal ascending flow at the ventricular septum (Figure 2a). The right coronary artery was dilated and the origin of the left coronary appeared to be possibly at the pulmonary artery, with a backflow into the pulmonary artery. The selective angiography clearly demonstrated ALCAPA with evidence of filling of the left side from a wide number of septal collaterals and a discrete shunt of the pulmonary artery (Figure 2b). The left ventricular angiography showed this cavity with normal morphology, dimensions and mobility. The nuclear medicine stress test revealed discrete anterolateral ischemia. In this patient it was decided to continue only with medical treatment; in the event of hemodynamic compromise or major ischemia a reimplantation procedure will be scheduled. Close follow–up has taken place, with check–ups every two months, for the last four years with all clinical, echocardiographic and nuclear medicine examinations. The patient has shown normal growth and development, remains asymptomatic, with no changes in the left ventricular dimension and with the same nuclear medicine stress test results.
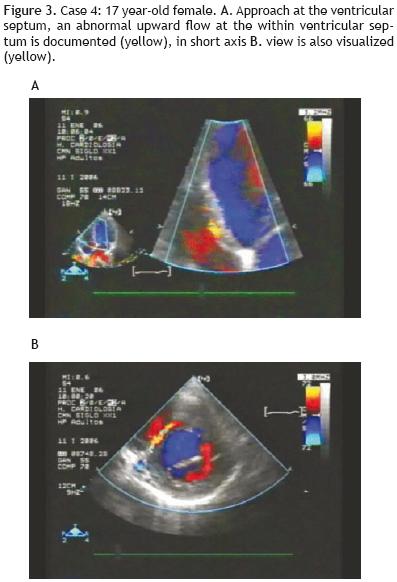
A 17 year–old female was referred to our center for dyspnea. She had normal ventricular functioning, but with an abnormal upward flow at the ventricular septum by echocardiography (Figures 3a and b). This finding, combined with the observation of a dilated origin of the right coronary artery, generated the suspicion of ALCAPA. Figure 4a, shows the suprasternal viewed at 45° from which it was evident that the left coronary originated from the pulmonary artery with reverse flow.
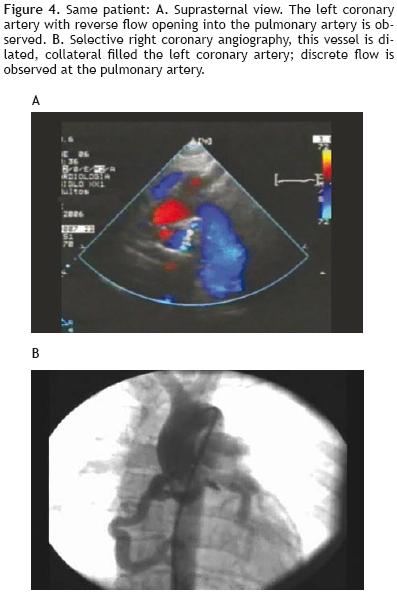
The selective right coronary angiography demonstrated dilation of this vessel and a collateral circulation at the muscular septum level was observed, which filled the left system in a retrograde fashion with a discrete flow of the contrasting agent at the pulmonary artery from the origin of the left coronary (Figure 4b). Reimplantation surgery was refused by the patient's parents.
Finally, case three is a 55–year–old female, referred for stress angina, who had an anterior ischemia diagnosed by electrocardiogram with a positive nuclear medicine test, without necrosis and 60% ejection fraction. The echocardiogram revealed an abnormal upward flow at the ventricular septum level with the left ventricle and right coronary artery slightly dilated. This patient was scheduled for coronary reimplantation, but did not show up for the surgery, claiming she felt well living in Veracruz, at sea level.
In the past 10 years, an additional adult was incorrectly diagnosed with ALCAPA by echocardiography when observing a dilated right coronary artery and an abnormal flow on the pulmonary artery. No collateral circulation was observed at the septum. The selective coronariography demonstrated that both coronary arteries originated normally, with a dilated fistula of the right coronary which ended into the pulmonary artery.
Discussion
ALCAPA is considered to be the most significant congenital coronary artery anomaly due to its prognostic and therapeutic implications. The incidence of ALCAPA ranges from 0.25% to 0.50% of all congenital cardiac anomalies.8 The clinical aspects clearly define two groups: the infant group, with severe cardiac failure, and the older group, frequently asymptomatic.9 The severity of ALCAPA forms a continuous spectrum and it may be detected at any age. The early or late clinical expression reflects the number of collaterals that the patient developed to prevent ischemia and necrosis in the left ventricle. It is interesting to note that a small number of asymptomatic infants under two months of age have been reported. This is due to the fact that during the first few weeks of life, the pulmonary diastolic pressure is still high, allowing for the perfusión of the left ventricle with blood flowing through the left coronary from the pulmonary artery. The heart tolerates hypoxemia better than low perfusión. In this scenario it is relevant to note the reported case of the catastrophic consequences of ligation of a large arterial duct, which supports the pulmonary pressure in a patient with ALCAPA. This patient died after the surgical ligation of the arterial conduct.10
The critical period of the left ventricle is reached once the pulmonary pressure drops. If the patient develops enough collaterals to join the right and left coronaries, then the perfusión from the right coronary will reach the left myocardium overcoming ischemia and preventing myocardial damage; these collaterals serve as a bridge between both coronaries. In the majority of cases with good collateral flow, a fraction of blood flow, up to the left coronary, is lost through from left–to–right shunt in the pulmonary artery. The well–established collateral circulation, which allows for normal development of the patient, even up to adulthood, combined with the upward flow of the anterior descending coronary artery, is diagnosed by echocardiography as an abnormal upward flow at the ventricular septum. Since its description by Houston and colleagues in 1990, this finding has become the most solid evidence for the diagnosis of ALCAPA by echocardiography in older patients.11 In this work, this finding was observed in all three cases belonging to the older group in which the dilation of the right coronary artery at its origin was also identified. The direct observation with transthoracic echocardiography of the origin of the left coronary from the pulmonary artery is difficult. We were able to clearly observe this malformation in one patient from the suprasternal view, which has not been previously reported (Figure 4a). In any case, the direct visualization by transthoracic echocardiography has led to false positives and negatives.12 Transesophageal echocardiography seems to be a more reliable method in the direct identification of anomalous formations of the left coronary from the pulmonary artery.13
In young children, collateral circulation is either absent of poorly developed, evidence consistent with the echocardiographic findings, as occurred in the two infants reported in this work. However, the dilation and hypokinesia of the left ventricle combined with the different degrees of mitral insufficiency and the difficulty in recognizing the origin of the left coronary, must lead to the probable diagnosis of ALCAPA, in particular, when it is associated to ischemia, necrosis or both, as occurred with our patients.
In children and adults, an abnormal flow at the main pulmonary artery, which does not run parallel to the lateral wall (as in the flow of a permeable arterial conduct), could be the first echocardiographic sign of ALCAPA. Next, the ventricular septum must be studied in search of a collateral flow or an upward flow from the left coronary. This should be performed, along with an analysis of a possible dilation of the origin of the right coronary, with the use of echocardiographic zoom.
Some authors consider the diagnosis by echocardiography sufficient clinical evidence to schedule the patients for surgery.14 Others, 15–16 share our point of view that ALCAPA must be verified by selective coronary artery angiography, since there could be other lesions, such as a coronary fistula opened to the pulmonary artery. Magnetic resonance imaging has been helpful in the diagnosis of ALCAPA.17 201–thallium dipirimadol SPECT studies have also been successfully used to non–invasively study ventricular functioning and perfusión.18
Conclusions
ALCAPA is a rare congenital disorder, but it is the most significant congenital coronary artery anomaly.
A thorough understanding of its clinical and electrical manifestations in infants and older groups is required.
In all infants with cardiac failure, left ventricular dilation with low systolic functioning and mitral insufficiency, as detected by echocardiography, is imperative to search for a right coronary dilation and the absence of a left coronary originating from the aorta.
In older patients, even those who are asymptomatic and with normal ventricular function, the presence of an upward flow at the ventricular septum, combined with a dilated right coronary artery and abnormal flow at the base of the pulmonary artery, as observed by echocardiography, strongly suggest the presence of ALCAPA.
Even though some groups consider diagnosis by echocardiography as sufficient evidence for its diagnosis, we believe that ALCAPA should be corroborated in all cases by selective coronary artery angiography.
During the revision of this article, another case, an 11 year–old female diagnosed by echocardiography with ALCAPA, all the findings described in older group, were documented and confirmed by the cardiac surgeon who performed the reimplantation of the left coronary artery.
Acknowledgments: The authors express their gratitude to Hector Alva, PhD, for his help with the English language.
Bibliography
1. Virmani R, Burke AP, Farb A: The pathology of sudden death in athletes. In Williams RA, ed. The Athlete and Heart Disease. Philadelphia: Lippincott Williams a Wilkins; 2000; 249–172. [ Links ]
2. Brooks HS: Two cases of an abnormal coronary artery of the heart arising from the pulmonary artery with some remarks upon effect of this anomaly in producing cirsoid dilatation of the vessels. J Anat Physiol 1886;20:26–29. [ Links ]
3. Abbot M. Congenital Cardiac Disease. In: Osier W, ed. Modern Medicine: its Theories and Practice. Philadelphia: Lea and Febiger, 1908; 4:323–425. [ Links ]
4. Bland EF, White PD, Garland J: Congenital anomalies of the coronary arteries. Am Heart J 1933;8:787–793. [ Links ]
5. Reul RM, Cooley DA, Hallman GL, Reul GJ: Surgical treatment of coronary artery anomalies: report of 371/2 year experience at the Texas Heart Institute. Texas Heart Inst J 2002;29:299–307. [ Links ]
6. Angelini PA, Velasco JA, Flamm S: Coronary anomalies. Incidence, pathophysiology, and clinical relevance. Circulation 2002;105:2449–2454. [ Links ]
7. Michielon G, Di Carlo D, Brancaccio G, Guccione P, Mazzera E, Toscano A, et al: Anomalous coronary artery origin from the pulmonary artery: Correlation between surgical timing and left ventricular function recovery. Ann Thorac Surg 2003; 76:581 –588. [ Links ]
8. Dodge–Khatamani A, Mavroudis C, Backer: Anomalous origin of the left coronary artery from the pulmonary artery: collective review of surgical therapy. Ann Thorac Surg 2002;74:946–955. [ Links ]
9. Smith A, Arnold R, Anderson RH, Wilkinson JL, Qureshi SA, Gerlis LM, et al: Anomalous origin of the left coronary artery from the pulmonary trunk. Anatomic findings in relation to pathophysiology and surgical repair. J Thorac Surg 1989;98:16–24. [ Links ]
10. Roh LS: Sudden death due to Anomalous origin of the left coronary artery from the pulmonary trunk. J Forensic Sci 1980;25:40–43. [ Links ]
11. Houston AB, Pollock JC, Doig WB, Gnanapragasam J, Jamieson MP, Lilley S, et al: Anomalous origin of the left coronary artery from the pulmonary trunk: Elucidation with colour Doppler flow mapping. Br Heart J 1990; 63:50–54 [ Links ]
12. Snider AR, Serwer GA, Ritter SB. Abnormal vascular connections and structures. In Echocardiography in Pediatric Heart Disease. St. Louis Mo. Mosby–Year Book, 1997 p. 486–488. [ Links ]
13. Lin PC, Chen YP, Chen TH: Anomalous origin of the left main coronary artery from the main pulmonary artery in a young adult. Circulation 2001; 104:1575–1576. [ Links ]
14. Hildreth B, Junkel P, Aliada V, Sintek C, Sapin S: An uncommon echocardiographic marker for anomalous origin of the left coronary artery from the pulmonary artery: visualization of intercoronary collaterals within the ventricular Septum. Pediatr Cardiol 2001;22:406–468 [ Links ]
15. Ya–Li Y, Navin NC, Xin–Fang W, Ming–Xing X, Quing L, Lin H, et al: Echocardiographic diagnosis of anomalous origin of the left coronary artery from the pulmonary artery. Echocardiography 2007;214:405–411. [ Links ]
16. Hauser M: Congenital anomalies of the coronary arteries. Heart 2005;91:1240–1245. [ Links ]
17. Schwerzmann M, Salehian O, Elliot T, Merchant N, Siu SC, Webb GD: Anomalous origin of the left coronary artery from the main pulmonary artery in adults. Circulation 2004; 110:e511–e513. [ Links ]
18. Chen ML, Lo HS, Chao IM, Su HY: Dipiridamole T1–201 myocardial single photon emission computed tomography of anomalous left coronary artery from the pulmonary artery. Clin Nucl Med 2007;32:940–943. [ Links ]














