Servicios Personalizados
Revista
Articulo
Indicadores
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Archivos de cardiología de México
versión On-line ISSN 1665-1731versión impresa ISSN 1405-9940
Arch. Cardiol. Méx. vol.78 no.4 Ciudad de México oct./dic. 2008
Investigación clínica
Blood pressure and associated cardiovascular risk factors in adolescents of Mexico City
Tensión arterial y su asociación con factores de riesgo cardiovascular en adolescentes de México, D.F.
Juan Gabriel Juárez–Rojas,* Guillermo C Cardoso–Saldaña,* Rosalinda Posadas–Sánchez,* Aída Xóchitl Medina–Urrutia,* Liria Yamamoto–Kimura,** Carlos Posadas–Romero*
* Department of Endocrinology, Instituto Nacional de Cardiología Ignacio Chávez, Mexico City, Mexico.
** Department of Public Health, School of Medicine, Universidad Nacional Autónoma de Mexico, Mexico City, Mexico.
Correspondence:
Dr. Posadas–Romero Carlos.
Instituto Nacional de Cardiología Ignacio Chávez
(INCICH, Juan Badiano Núm. 1, Col. Sección XVI,
Tlalpan 14080. México D.F.).
Tel: (+ 5255) 55732911 Ext. 1272.
Fax: (+5255) 55734687.
E–mail: cposadasr@yahoo.com
Recibido: 8 de octubre de 2007
Aceptado: 22 de mayo de 2008
Abstract
Objective: To determine the prevalence of high blood pressure and associated cardiovascular risk factors in Mexican adolescents.
Methods: A cross–sectional study was conducted in 770 male and 1076 female students (12 to 16 years old) from eight randomly selected high schools in Mexico City. Anthropometry, blood pressure and fasting lipids and lipoproteins were measured.
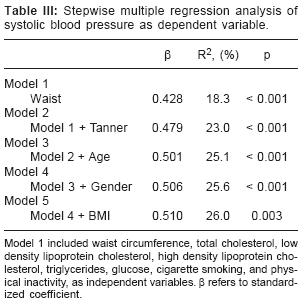
Results: Blood pressure levels were adjusted for age, gender, and height. The prevalence rates of hypertension (systolic blood pressure (SBP) and/or diastolic (DBP) > 95th percentile), and pre–hypertension (SBP or DBP > 90th but < 95th percentile) were 10.6 and 10%, respectively. Compared to normotensive subjects, those with high blood pressure showed a significantly higher prevalence of obesity, overweight, and dyslipidemia. A stepwise multiple regression analysis showed that waist (18.3%), Tanner stage (4.7%), age (2.1 %), gender (0.6%), and body mass index (BMI, 0.3%) accounted for 26% of the variance in SBP; whereas BMI (8.7%), age (4.8%), Tanner stage (1.7%), waist (0.4%), and gender (0.4%) accounted for 15.9% of the variance in DBP.
Conclusions: These results reveal a high prevalence of high blood pressure in adolescents living in Mexico City. Pre–hypertensive and hypertensive subjects showed a higher prevalence of cardiovascular risk factors, suggesting that, as adults, these adolescents will be at a higher risk of developing cardiovascular disease.
Key words: Adolescents. Hypertension. Obesity. Cardiovascular risk.
Resumen
Objetivo: Determinar la prevalencia de tensión arterial elevada y su asociación con factores de riesgo cardiovascular en adolescentes mexicanos.
Métodos: Se realizó un estudio transversal en 770 hombres y 1,076 mujeres (12 a 16 años de edad) de ocho escuelas secundarias seleccionadas aleatoriamente de la Ciudad de México. Se determinaron las medidas antropométricas, la tensión arterial, los lípidos y las lipoproteínas.
Resultados: Las mediciones de tensión arterial fueron ajustadas por edad, género y estatura. La tasa de prevalencia de hipertensión (tensión arterial sistólica y/o la tensión arterial diastólica > a la percentila 95) y la prevalencia de pre–hipertensión (tensión arterial sistólica y diastólica > percentila 90 y < a la percentila 95) fueron 10.6% y 10.0%, respectivamente. Comparados con los adolescentes normotensos, los hipertensos mostraron una prevalencia significativamente elevada de obesidad, sobrepeso y dislipidemia. El análisis de regresión múltiple mostró que la cintura (18.3%), el Tanner (4.7%), la edad (2.1%), el género y el índice de masa corporal (0.9%) contribuyeron con 26% de la variación en la tensión arterial sistólica, mientras que el índice de masa corporal (8.7%), la edad (4.8%), el Tanner (1.7%), la cintura (0.4%) y el género (0.4) contribuyeron con el 15.9% de la variación en la presión arterial diastólica.
Conclusiones: Estos resultados muestran una elevada prevalencia de hipertensión arterial en los adolescentes de la Ciudad de México. Los adolescentes pre–hipertensos e hipertensos mostraron una mayor prevalencia de factores de riesgo cardiovascular, lo que sugiere que, como adultos, estos adolescentes tendrán un mayor riesgo de desarrollar enfermedad cardiovascular.
Palabras clave: Adolescentes. Hipertensión. Obesidad. Riesgo cardiovascular.
Introduction
Coronary heart disease (CHD) is the leading cause of death in most of the developed1,2 and in developing countries, like Mexico.3 Available evidence indicates that atherosclerosis begins in childhood and is associated to different coronary risk factors.4,5 These findings and the observations that these factors tend to track from childhood into adult life1 emphasize the importance of identifying children with these conditions.
The prevalence of obesity has been increasing around the world6 and is accompanied by increases of other coronary risk factors, such as arterial hypertension.7,8 Numerous studies9–11 conducted among different ethnic and racial groups have shown that there are geographic variations in the childhood hypertension prevalence (1 to 25%).
As in many countries, the prevalence of excess body weight in Mexican children has increased during the past years.12 Several epidemiologic studies in Mexican adult populations have shown a high and increasing prevalence of hypertension.13,14 However, little is known about hypertension rates and its association with other cardiovascular risk factors in Mexican adolescents. Therefore, the purpose of the present study was to determine the prevalence of high blood pressure and associated cardiovascular risk factors in a population of adolescents residing in Mexico City.
Methods
Study design and participants
A cross–sectional survey was conducted as previously described.15 Briefly, eight urban public junior high schools (Mexico City) were randomly selected from a list of high schools, provided by the Ministry of Education. The Epi–info software version 6.04 (CDC, USA; WHO, Geneva, Switzerland) was used to calculate the sample size, considering 5% of high blood pressure levels (intervals 3.5% to 6.5%) for a population survey or descriptive studies,16 with a power of 80% and a 95% confidence interval. The calculated sample size was 810 adolescents.
Recruitment of participants
Because school authorities did not allow a random selection, all students 12 to 16 years of age attending the selected schools were invited to participate. A sample of 1,846 adolescents (770 males and 1,076 females) who accepted to participate and whose parents signed the informed consent were included. To assess potential bias introduced by differences between participants and non–participants, a random subsample of non–participants answered the questionnaire and had anthropometry and blood pressure measurements taken and no statistical differences were found. Adolescents with an organic pathology or under chronic medical treatment were excluded. The protocol was approved by the Research Coordination of the Department of Public Health at the Medical School of the National Autonomous University of Mexico and the authorities of the Ministry of Education.
Sources, methods, and techniques for data collection
Questionnaire application, anthropometry and blood pressure measurements were performed between 8:00 to 11:00 AM from Monday to Thursday. Adolescents answered individually questions about cigarette smoking and in order to encourage truthful answers, students were reassured that nobody (parents and/or teachers) would receive the information about smoking habits. A participant was defined as a smoker when he/she smoked at least one cigarette per week.17 Adolescents, who did not participate in any sport or physical activity in their leisure time were considered to have a sedentary life style.18
Blood pressure
After at least 10 minutes rest, systolic (Korotkoff phase I) and diastolic (Korotkoff phase V) blood pressure (SBP and DBP) values were recorded with a mercury sphygmomanometer (Welch Allyn Tycos, USA) using an appropriate cuff size for each student. Three readings were recorded for each individual. Average of the second and third measurements was defined as the subject's blood pressure. The blood pressure values were adjusted for age, height, and gender to classify subjects in three groups: 1) normotensives (NT) (SBP and DBP < 90th percentile); 2) pre–hypertensives (Pre–HT) (SBP or DBP > 90th but < 95th percentile); and 3) hypertensives (HT) (SBP and/or diastolic DBP > 95th percentile).19
Anthropometry measurements
Body weight was measured and recorded to the nearest 0.1 kg, height to 0.5 cm, and waist circumference to the nearest 0.5 cm at the midpoint between the bottom of the rib cage and above the top of the iliac crest. Body mass index (BMI) was calculated, and we used the cutoff points of overweight (25 kg/m2) and obesity (30 kg/m2) according to Cole.20
Lipid and lipoprotein measurements
Venous blood samples were obtained after 12–h overnight fast. Total cholesterol (TC) and triglycerides (TG) were measured by enzymatic methods (Roche–Syntex/Boehringer Mannheim, Germany). High density lipoprotein cholesterol (HDL–C) was quantified after precipitation of lipoproteins containing apo B with phosphotungstate/Mg2+. Low density lipoprotein–cholesterol (LDL–C) was estimated by Friedewald's formula.21 Plasma glucose was determined by the glucose–oxidase method (Roche–Syntex/Boehringer Mannheim, Germany). Accuracy and precision were under the surveillance of the Center for Disease Control Lipid Standardization Program (Atlanta, GA). The intra–assay coefficient of variation (CV) for TC, TG, HDL–C, and glucose were 0.43, 0.89, 1.72, and 1.0%, respectively. The interassay CVs were 1.76, 2.03, 3.24, and 1.7 %, respectively. Hypercholesterolemia was defined as TC > 200 mg/dL, high LDL–C as LDL–C > 130 mg/dL, hypertriglyceridemia as TG level > 150 mg/dL, and low HDL–C as HDL–C level < 35 mg/dL.1
Pubertal development
Sexual development was assessed using the method described by Tanner.22 Self–assessment is a valid method to assess sexual maturity when feasibility of physician examination is low in clinical evaluation, and as a research tool.23 In the case of girls, the date of menarche was also recorded.
Statistical analysis
Descriptive statistics, including mean and standard deviations (SD) for continuous variables and proportions for categorical variables, were used. All variables were tested for normality using the Kolmogorov–Smirnov test. Variables that were not normally distributed were log–transformed and then parametric statistics were used. Comparison of means by gender was evaluated by two–tailed t–test. Differences between more than two groups were evaluated using ANOVA test. Prevalence values between genders and between adolescents with normal and those with high blood pressure levels were compared using X2 test. Pearson correlation analysis was used to examine the association between systolic and diastolic blood pressure and other cardiovascular risk factors. The independence of associations was determined by stepwise multiple regression analysis. A p value < 0.05 indicated statistical significance. Statistical analysis was performed using SPSS version 10 software (SPSS, Chicago, IL).
Results
The clinical, anthropometric, and metabolic characteristics according to gender are shown in Table I. Compared with girls, boys had significantly lower mean values of BMI, DBP, TC, LDL–C, and TG; whereas, girls had significantly lower glucose and SBP values than boys. Theprevalence of Pre–HT(9.6%)and HT(9.9%) among boys was lower but not significantly different from that among girls (Pre–HT 10.3%; HT 11.1%). The cardiovascular risk factors studied were generally more common in HT and Pre–HT adolescents of both genders than in normal blood pressure subjects. As shown in Figure 1a, overweight and low HDL–C levels were the most frequent risk factors observed in HT boys, but obesity, high total and LDL cholesterol were also more prevalent than in NT subjects. In Pre–HT adolescents, overweight and obesity were also significantly higher than in normotensive subjects. Although several risk factors were more commonly seen in HT than in Pre–HT subjects, the differences did not reach statistical significance. Among girls (Fig. 1b), physical inactivity was the most common risk factor irrespective of blood pressure group. As in boys, HT girls showed higher prevalence of overweight, obesity, and elevated total and LDL cholesterol than normotensive ones. Smoking and low HDL–C was significantly higher in Pre–HT than in those girls with normal blood pressure. As in boys, no significant differences were observed between Pre–HT and HT girls. Compared with boys, girls had higher overweight prevalence in the NT group, were more physically inactive in all three groups, and had lower prevalence of low HDL–C in the NT and HT groups. In addition, there were fewer smokers among girls than among boys in the NT group.
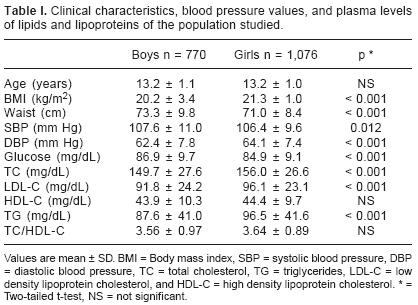
Because overweight and obesity were observed in almost one fourth of adolescents and because these two conditions are associated to higher prevalence of both hypertension and dyslipidemia, we examined the effect of body mass index and blood pressure levels on lipid concentrations. For this analysis, we considered two blood pressure groups: hypertensive (HT) and non–hypertensive (Pre–HT and NT combined), and according to their BMI they were categorized as obese, overweight, and normal weight adolescents. As expected, obese boys and girls were significantly more common in the HT group as compared with the non–HT group (Table II). For males and females, in both HT and non–hypertensive groups, TC, LDL–C, TC/HDL–C ratio, and TG mean values were higher in obese, intermediate in overweight, and lower in normal weight adolescents, except for TC and LDL–C in HT girls; whereas, HDL–C mean values were low, intermediate, and high for obese, overweight, and normal weight adolescents, respectively. When non–HT and HT subgroups were compared, the only difference observed was in triglycerides, which were higher in lean non–HT than in lean HT boys (p = 0.038).
A stepwise multiple regression analysis was used to determine the independent contribution of age, gender, Tanner stage, cigarette smoking, physical inactivity, and anthropometric and metabolic measurements to the variance in blood pressure levels. Waist circumference (18.3%), Tanner stage (4.7%), age (2.1%), gender (0.6%), and BMI (0.3%) accounted independently for 26% of the variance in SBP (Table III); whereas BMI (8.7%), age (4.8%), Tanner stage (1.7%), waist circumference (0.4%), and gender (0.4%) explained 15.9% of the variance in DBP (Table IV).

Discussion
Because adolescence is often considered a critical period for the development of some cardiovascular risk factors, several studies have suggested the screening of children to identify those at high risk for developing essential hypertension as adults.1,6 In the present study, a high prevalence of elevated blood pressure levels was found (10% for pre–hypertension, and 10.6% for hypertension). In addition, boys and girls with high blood pressure showed higher prevalence of other coronary risk factors as compared with normotensive subjects. The multiple regression analysis showed that the most important determinants of blood pressure were waist circumference for systolic and BMI for diastolic pressure. The prevalence of high blood pressure observed in our adolescents was generally higher than that found in other Latin American populations: 9.5% in Chileans,24 12% in Argentineans,25 and 3.0% in Colombians.26 It is also higher than the prevalence reported in European countries: 15% in North Ireland,9 12% in Germany,10 and 7.2% in Spain;11 or in Asian countries: < 10% in India27 and 4.8% in Turkey.28 Age, life style, dietary habits, environment, and differences in criteria to define high blood pressure levels may be the underlying factors for these variations in the prevalence of high blood pressure among adolescents from different countries.
Several studies have shown that age,19 gender,29 race,30 heritability,19,29 physical activity, and nutrition19,30 are associated to blood pressure in adolescents. Furthermore, it has been reported31 that obesity increases the occurrence of hypertension and the development of dyslipidemia. Our results clearly confirm these findings by showing that excess body weight (obesity and overweight) was significantly (p < 0.001) more prevalent among HT (males, 38.2%; females, 44.5%) than among non–HT (males, 21.3%; females, 28.1%) and by demonstrating that greater adiposity explained the higher values for TC, LDL–C, TG, and TC/HDL–C ratio, as well as lower concentrations of HDL–C, not only in HT but also in non–HT subjects (Table II). Further, as already mentioned, in the multiple regression analysis, waist circumference and BMI accounted for most of the variance in SBP and DBP, respectively.
In both, children and adults, obesity increases 3 to 8 times the prevalence of hypertension.6,32 Previous studies7,33 in over 56,300 children found that, irrespective of race, gender, or age, the risk of elevated blood pressure was significantly higher for children in the upper than in the lower BMI group, with odd ratios for hypertension ranging from 2.4 to 4.5. In another study,34 overweight was found to be the primary contributing factor to hypertension among different ethnic groups. In addition, Rosner33 reported a linear increase in the prevalence of diastolic hypertension in children of all races, gender and age combinations as BMI increased above the "normal" range.
Obesity–induced hypertension is likely due to an overlap or combinations of three main patho–physiological mechanisms. First, the link between obesity and hypertension may be mediated in part by sympathetic nervous system hyperactivity.35 Second, the insulin resistance associated with obesity may prevent insulin–induced glucose uptake but leave the renal sodium retention effects of insulin relatively preserved, thereby resulting in chronic volume overload and maintenance of blood pressure elevation.8 Third, lower arterial compliance, lower distensibility, and lower endothelium–dependent and independent function in severely obese than in control children have been reported.36
Recent research has shown that the pathogenesis of obesity may be, in part, inherited, but genetics cannot account for the rapid increases in overweight around the world.2 Lack of safe outdoor play areas and households with two working parents or a single parent limit children's ability to engage in active physical exercise or recreational sports, and reliance on sedentary entertainment, including television viewing, video games, and computers, has increased. In the present study, sedentary life style was significantly more prevalent in girls than in boys (40.9% vs. 14.5%, p < 0.001). The higher prevalence of overweight in girls (20.3% vs 18.3% in boys; p < 0.001) might be a reflection of the lower physical activity in girls. Health professional caring for children and adolescents are in a key position to help prevent and treat obesity by promoting behavioral and environmental changes.
This study has several limitations. First, Tanner scores were obtained by self–assessment. There are some studies, conducted mainly in obese children37,38 showing inconsistent percentages of agreement between physician's evaluation and self–assessment. However, other studies23,39,40 have found a good agreement between the scoring by this method and that obtained by direct observation. Therefore, it is unlikely that our results are invalidated by the use of self–assessed puberty. Second, we were unable to obtain information about diet, which is a factor with an important influence on blood pressure levels. Third, our study is based on cross–sectional data and it cannot address questions of causality inferences. Finally, because geographic, socio economic, cultural and ethnic characteristics are different around the country, the results obtained in this work, cannot be generalized to all Mexican adolescents. However, the data provide insights for the risk of cardiovascular disease in adolescents of Mexico City.
In conclusion, these results reveal a high prevalence of high blood pressure in adolescents living in Mexico City. Pre–hypertensive and hypertensive subjects also showed a high prevalence of other cardiovascular risk factors, mainly overweight and obesity. Our findings suggest that, as adults, these adolescents will be at a higher risk of developing cardiovascular disease. There is an urgent need to establish preventive measures to decrease high blood pressure in this population. Avoidance or correction of excess body weight should be the main target.
Acknowledgments
This work was partially supported by grant 0187PM9506 from the Consejo Nacional de Ciencia y Tecnología (CONACyT) and the National Institute of Cardiology "Ignacio Chavez", Mexico. We are grateful to parents, school authorities, and particularly to adolescents who made the conduction of the survey possible.
References
1. KAVEY RE, DANIELS SR, LAUER RM, ATKINS DL, HAYMAN LL, TAUBERT K: American Heart Association. American Heart Association guidelines for primary prevention of atherosclerotic cardiovascular disease beginning in childhood. Circulation 2003; 107: 1562–6. [ Links ]
2. WILLIAMS CL, HAYMAN LL, DANIELS SR, ROBINSON TN, STEINBERGER J, PARIDON S, BAZZARRE T: Cardiovascular health in childhood: A statement for health professionals from the Committee on Atherosclerosis, Hypertension, and Obesity in the Young (AHOY) of the Council on Cardiovascular Disease in the Young, American Heart Association. Circulation 2002; 106: 143–60. [ Links ]
3. LARA A, ROSAS M, PASTELIN G, AGUILAR C, ATTIE F, VELAZQUEZ MONROY O: Hipercolesterolemia e hipertensión arterial en México. Consolidación urbana actual con obesidad, diabetes y tabaquismo. Arch Cardiol Mex 2004; 74: 231–45. [ Links ]
4. BERENSON GS, SRINIVASAN SR, BAO W, NEWMAN WP 3RD, TRACY RE, WATTIGNEY WA: Association between multiple cardiovascular risk factors and atherosclerosis in children and young adults. The Bogalusa Heart Study. N Engl J Med 1998; 338: 1650–6. [ Links ]
5. NEWMAN WP 3RD, FREEDMAN DS, VOORS AW, GARD PD, SRINIVASAN SR, CRESANTA JL, ET AL: Relation of serum lipoprotein levels and systolic blood pressure to early atherosclerosis. The Bogalusa Heart Study. N Engl J Med 1986; 314: 138–44. [ Links ]
6. BAO W, THREEFOOT SA, SRINIVASAN SR, BERENSON GS: Essential hypertension predicted by tracking of elevated blood pressure from childhood to adulthood: the Bogalusa Heart Study. Am J Hypertens 1995; 8: 657–65. [ Links ]
7. HAFFNER SM, MIETTINEN H, GASKILL SP, STERN MP: Metabolic precursors of hypertension. The San Antonio Heart Study. Arch Intern Med 1996; 156: 1994–2001. [ Links ]
8. SOROF J, DANIELS S: Obesity hypertension in children: a problem of epidemic proportions. Hypertension 2002; 40: 441–7. [ Links ]
9. BOREHAM C, SAVAGE JM, PRIMROSE D, CRAN G, STRAIN J: Coronary risk factors in schoolchildren. Arch Dis Child 1993; 68: 182–6. [ Links ]
10. MICHEL U, RIECHERS B: Cardiovascular risk factors in schoolchildren. J Am Coll Nutr 1992; 11: 36S–40S. [ Links ]
11. ELCARTE LOPEZ R, VILLA ELIZAGA I, SADA GONI J, GASCO EGUILUZ M, OYARZABAL IRIGOYEN M, SOLA MATEOS A, ET AL: The Navarra study. Prevalence of arterial hypertension, hyperlipidemia and obesity in the infant–child population of Navarra. Association of risk factors. An Esp Pediatr 1993; 38: 428–36. [ Links ]
12. DEL RIO–NAVARRO BE, VELAZQUEZ–MONROY O, SANCHEZ–CASTILLO CP, LARA–ESQUEDA A, BERBER A, FANGHANEL G, ET AL: Encuesta Nacional de Salud 2000 Working Group, National Health Survey 2000. The high prevalence of overweight and obesity in Mexican children. Obes Res 2004; 12: 215–23. [ Links ]
13. ROSAS M, PASTELIN G, MARTINEZ–REDING J, HERRERA–ACOSTA J, ATTIE F: Comité Institucional para el Estudio y Prevención de la Hipertensión Arterial Sistémica. [Hypertension guidelines in Mexico]. Arch Cardiol Mex 2004; 74: 134–57. [ Links ]
14. LORENZO C, SERRANO–RIOS M, MARTINEZ–LARRAD MT, GABRIEL R, WILLIAMS K, GONZALEZ–VILLALPANDO C, ET AL: Prevalence of hypertension in Hispanic and non–Hispanic white populations. Hipertensión 2002; 39: 203–8. [ Links ]
15. YAMAMOTO–KIMURA L, POSADAS–ROMERO C, POSADAS–SANCHEZ R, ZAMORA–GONZALEZ J, CARDOS OSALDANA G, MENDEZ RAMIREZ I: Prevalence and interrelations of cardiovascular risk factors in urban and rural Mexican adolescents. J Adolesc Health 2006; 38: 591–8. [ Links ]
16. KISH L: Survey sampling, New York, John Wiley & Sons, 1965. [ Links ]
17. BEWLEY BR, DAY I, IDE L: Smoking by children in Great Britain. A review of the literature. Medical Research Council and Social Science Research Council, London, 1972. [ Links ]
18. SALONEN JT, SLATER JS, TUOMILEHTO J, RAUHRAMEE R: Leisure time and occupational physical activity. Risk of death from ischemic heart disease. Am J Epidemiol 1988; 127: 87–94. [ Links ]
19. National High Blood Pressure Education Program Working Group on High Blood Pressure in Children and Adolescents: The Fourth Report on the Diagnosis, Evaluation, and Treatment of High Blood Pressure in Children and Adolescents. Pediatrics 2004; 114 (Suppl 2): 555–76. [ Links ]
20. COLE TJ, BELLIZZI MC, FLEGAL KM, DIETZ WH: Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ 2000; 320: 1240–3. [ Links ]
21. DELONG DM, DELONG ER, WOOD PD, LIPPEL K, RIFKIND BM: A comparison of methods for the estimation of plasma low– and very low–density lipoprotein cholesterol. The Lipid Research Clinics Prevalence Study. JAMA 1986; 256: 2372–7. [ Links ]
22. TANNER JM: The Development of the reproductive system. In: Growth at Adolescence. (Tanner JM, ed.). Oxford. Blackwell Scientific. 1962; pp 28–39, [ Links ]
23. SCHMITZ KE, HOVELL MF, NICHOLS JF, IRVIN VL, KEATING K, SIMON GM, ET AL: A Validation Study of Early Adolescents Pubertal Self–Assessments. Journal Early Adolescence 2004; 24: 357–84. [ Links ]
24. NORERO C, VARGAS NA, MAYNE D, MONTI A, KUTZ M, SAITO R: Blood pressure levels in urban school–age population in Chile. Hypertension 1981; 3: 238–41. [ Links ]
25. ENNIS IL, GENDE OA, CINGOLANI HE: Prevalence of hypertension in 3154 young students. Medicina (B.Aires) 1998; 58: 483–91. [ Links ]
26. USCATEGUI PENUELA RM, ALVAREZ URIBE MC, LAGUADO SALINAS I, SOLER TERRANOVA W, MARTINEZ MALUENDAS L, ARIAS ARTEAGA R, ET AL: Cardiovascular risk factors in children and teenagers aged 6–18 years old from Medellin (Colombia). An Pediatr (Bare) 2003; 58: 411–7. [ Links ]
27. SINGH AK, MAHESHWARI A, SHARMA N, ANAND K: Lifestyle associated risk factors in adolescents. Indian J Pediatr 2006; 73: 901–6. [ Links ]
28. UCAR B, KILIC Z, COLAK O, ONER S, KALYONCU C: Coronary risk factors in Turkish schoolchildren: randomized cross–sectional study. Pediatr Int 2000; 42: 259–67. [ Links ]
29. SINAIKO AR: Hypertension in children. N Engl J Med 1996; 335: 1968–73. [ Links ]
30. Update on the 1987 Task Force Report on High Blood Pressure in Children and Adolescents: a working group report from the National High Blood Pressure Education Program. National High Blood Pressure Education Program Working Group on Hypertension Control in Children and Adolescents. Pediatrics 1996; 98(4 Pt 1): 649–58. [ Links ]
31. FREEDMAN DS, DIETZ WH, SRINIVASAN SR, BERENSON GS: The relation of overweight to cardiovascular risk factors among children and adolescents: the Bogalusa Heart Study. Pediatrics 1999; 103(6 Ptl): 1175–82. [ Links ]
32. SCHMIEDER RE, MESSERLI FH, RUDDEL H: Risks for arterial hypertension. Cardiol Clin 1986; 4: 57–66. [ Links ]
33. ROSNER B, PRINEAS R, DANIELS SR, LOGGIE J: Blood pressure differences between blacks and whites in relation to body size among US children and adolescents. Am J Epidemiol 2000; 151: 1007–19. [ Links ]
34. SOROF JM, LAI D, TURNER J, POFFENBARGER T, PORTMAN RJ: Overweight, ethnicity, and the prevalence of hypertension in school–aged children. Pediatrics 2004; 113(3 Pt 1): 475–82. [ Links ]
35. GUTIN B, OWENS S, SLAVENS G, RIGGS S, TREIBER F: Effect of physical training on heart–period variability in obese children. J Pediatr 1997; 130: 938–43. [ Links ]
36. TOUNIAN P, AGGOUN Y, DUBERN B, VARILLE V, GUY–GRAND B, SIDI D, ET AL: Presence of increased stiffness of the common carotid artery andendot–helial dysfunction in severely obese children: a prospective study. Lancet 2001; 358: 1400–4. [ Links ]
37. HERGENROEDER AC, HILL RB, WONG WW, SANGIHAGHPEYKAR H, TAYLOR W: Validity of self–assessment of pubertal maturation in African American and European American adolescents. J Adolesc Health 1999; 24: 201–5. [ Links ]
38. BONAT S, PATHOMVANICH A, KEIL MF, FIELD AE, YANOVSKI JA: Self–assessment of pubertal stage in overweight children. Pediatrics 2002; 110: 743–7. [ Links ]
39. DUKE PM, LITT IF, GROSS RT: Adolescents' self–assessment of sexual maturation. Pediatrics 1980; 66: 918–20. [ Links ]
40. BROOKS–GUNN J, WARREN MP, ROSSO J, GARGIULO J: Validity of self–report measures of girls' pubertal status. Child Dev 1987; 58: 829–41. [ Links ]














