Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Agrociencia
versión On-line ISSN 2521-9766versión impresa ISSN 1405-3195
Agrociencia vol.52 no.5 Texcoco jul./ago. 2018
Water-soils-climate
Alometric equations for estimating biomass and carbon in oil palm (Elaeis guineensis Jacq.) in the humid tropic of Chiapas, Mexico
1 Facultad de Ciencias Agrícolas Campus IV, Universidad Autónoma de Chiapas (UNACH).
2 Facultad de Ciencias Agronómicas Campus V, UNACH, Villaflores, Chiapas, México. (aleycoss@gmail.com).
3 Ganadería. Campus Montecillo. Colegio de Postgraduados. 56230. Montecillo, Estado de Mexico.
4 Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias, Campo Experimental Rosario Izapa.
The state of Chiapas, Mexico, has the climatic and soil potential for cultivation of the oil palm (Elaeis guineensis Jacq.). Currently, around 48 000 ha are planted with this species, representing a major carbon (C) sink. The objective of this study was to measure biomass and accumulated C in 12-year-old palms to generate the base line and methodology to permit predicting C fixation for this crop. Randomized destructive sampling was conducted on four plants per site. In three subsamples of stem and leaves, biomass, diameter at breast height (DBH), total height and number of leaves per plant were determined. In 20 subsamples of soil, root biomass was determined. Carbon content was estimated with the wet digestion technique of Walkley and Black. With the data, two alometric equations were generated to calculate biomass and a mathematical model in its linear expression for C, in which Y is the content of biomass or C, and X is the plant height. The concentration of C in stem, branches and root was 48.3, 48.1 and 48.2 %. The average C concentration per plant was 48.24 %. With the biomass and plant height, the model Y = 98.349X + 737.41 (R2 = 0.577) was obtained. To establish the variables of the predictive model of accumulated C per palm (CAP) in function of height, a procedure identical to the previous procedure was used, and CAP = 55.15X + 326.96 (R2 = 0.519). A larger number of field variables is necessary to obtain acceptable alometric equations (higher R2) for prediction of biomass and palm C stock. The methodology permitted determining biomass and C accumulated in the oil palm, thus establishing the bases for its study.
Key words: Elaeis guineensis Jacq.; alometric equations; greenhouse effect gasses; carbon sink
El estado de Chiapas, México, posee potencial agroclimático y edáfico para cultivar palma de aceite (Elaeis guineensis Jacq.). En la actualidad hay alrededor de 48 mil ha plantadas con esta especie, lo que representa un sumidero importante de carbono (C). El objetivo de esta investigación fue medir la biomasa y el C acumulado en plantas de palma de 12 años para generar la línea base y la metodología que permita predecir la fijación de C para este cultivo. Un muestreo destructivo, aleatorizado, se realizó en cuatro plantas por sitio. En tres submuestras de tallo y hojas se evaluó la biomasa, el diámetro a la altura de pecho (DAP), la altura total y el número de hojas por planta también se midieron. En 20 submuestras de suelo se determinó la biomasa de la raíz. El contenido de C se estimó con la técnica de digestión húmeda de Walkley y Black. Con los datos se generaron dos ecuaciones alométricas para calcular la biomasa y un modelo matemático en su expresión lineal para el C, en el cual Y es el contenido de biomasa o C y X es la altura de la planta. La concentración de C en fuste, ramas y raíz fue 48.3, 48.1 y 48.2 %. La concentración promedio de C por planta fue 48.24 %. Con la biomasa y la altura de las plantas se obtuvo el modelo Y = 98.349X + 737.41 (R2 = 0.577). Para establecer las variables del modelo predictivo de C acumulado por palma (CAP), en función de su altura, se usó un procedimiento idéntico al anterior, y CAP = 55.15X + 326.96 (R2 = 0.519). Un número mayor de variables de campo es necesario para obtener ecuaciones alométricas aceptables (R2 mayores), para predecir biomasa y C almacenado en la palma. La metodología permitió determinar la biomasa y C acumulado en palma de aceite, estableciendo así la base para su estudio.
Palabras clave: Elaeis guineensis Jacq.; ecuaciones alométricas; gases de efecto invernadero; sumidero de carbono
Introduction
The diagnostic reports of the Intergovernmental Panel on Climate Change (IPCC) indicate that Latin America and the Caribbean, like other regions of the world, are vulnerable to adverse effects of climate change (CC) and contribute 12 % of the world emissions of greenhouse effect gases (GEI)5. The concentration of CO2 in the atmosphere has increased because of the use of fossil fuels, passing from 280 ppm in the preindustrial era to 402 ppm in 2016 (Prentice et al., 2001; NOAA, 2016). CO2 is the main cause of the greenhouse effect, which has directly modified global temperature. In 2004, the increase was 0.54 ºC; that year was considered the hottest in history (Smith et al., 2005).
Together with the increase in the demand for energy by industrial processes and urban development, extended areas of forest and jungles are being converted to agriculture. For this reason, alternatives are being sought for agricultural and livestock production with low C emissions or for fixation of the C of CO2 in crop biomass. This demands public policies and sustained agricultural production systems with high potential for fixing C, mitigating and adapting to CC and generating sustainable renewable energy (Pacheco et al., 2007).
Annual world deforestation causes the release of 1.8 x 1012 Mg of C, equivalent to 20 % of the total of anthropogenic emissions. Vegetation assimilates atmospheric CO2 via photosynthesis by which it accumulates carbohydrates and increases biomass volume. Forests capture and preserve more C than annual crops, and grasslands participate with 90 % of the annual C flow between the atmosphere and the earth’s surface (Derner and Schuman, 2007; Follett and Reed, 2010).
The cultivation of oil palm (E. guineensis Jacq.) under the agroecological and edaphic conditions of Central America and Asia has the potential to accumulate 96.02 and 103.35 Mg C ha-1 C in shoot biomass (Leblanc et al., 2006). To estimate C sequester by the palm in the study area, an inventory of C was taken; that is, we obtained a “photograph” equivalent of the current state or quantity of C in shoot biomass and in soil (Andrade et al., 2008). The potential of loss or sequester of C is considered to be the average of this element stored at any moment in the palm system, during its life cycle (Hairiah et al., 2011).
Carbon stocking is defined as the process of accumulating C in tissues of the growing plant that remains in situ or is transformed into products that do not release it rapidly into the atmosphere. Thus, C sequester is the result of dynamic processes of decomposition and flows generated by photosynthesis over a long time (Castilla, 2004). However, C stocks in agricultural and agroforestry systems are not considered among the mechanisms of CC mitigation. Here lies the need to generate information relative to the capacity of C fixation in cultivated species, such as the oil palm, using allometric equations to predict their potential for C sequester. The objective of this study was to design a base methodology to estimate C captured in the shoot and root fractions of the oil palm in the Soconusco region, Chiapas, Mexico.
Materials and Methods
Study area and plant material
The study was conducted with samples of oil palm plants collected in 1) Tapachula de Córdoba (14°45´29.31” N, 92°32´16.35” W; altitude 17 m), 2) Huehuetán (14°58´52.20” N, 92°28´30.35” W; altitude 18 m), and 3) Villa Comaltitlán (15°06´54.70” N, 92°37´42.94” W; altitude 10 m) Soconusco region, Chiapas.
Shoot sampling
The aboveground biomass was collected from 12 plants of more than 12 years old. Plants were cut with a chainsaw (Truper, Mod. Mot-4518; China) where the stipe begins, and the stem separates from the leaves. The stems were measured for total height (Ht) and diameter and breast height (DBH: 1.30 m) with a metric measuring tape (Truper, model Tp20ME de 20 m; China). Each stem was divided into three or more sections to determine fresh weight in situ with an electronic platform scale (OHAUS; Mod. ES50L AM, 50 kg capacity; USA). Volume of biomass was calculated with the truncated cone geometric formula. The lower and upper diameters of each section were measured. Sections 10 cm thick were cut with the chainsaw and dry weight was determined in three subsamples. All the sections were obtained from the height where the lower stem diameter was determined. Leaf biomass was determined in four leaves, one per cardinal point, with an electronic platform scale. The stem and leaf subsamples were weighed immediately after cutting to determine fresh weight, placed in plastic bags and taken to the biotechnology laboratory of the Facultad de Ciencias Agrícolas, Campus IV, Huehuetán, Chiapas. The samples were dried to constant weight in a circulating air oven (VWR international, Mod. 1390FM, Sheldon Manufacturing, Inc. USA) for 96 h at 60 ºC. The ratio dry weight:fresh weight was used as the estimator of the amount of biomass of the stem and branches in samples and subsamples using the technique described by Díaz-Franco et al. (2007) and Acosta et al. (2002).
Root sampling
Carbon in the palm roots and C stored in the soil was quantified by humification of the biomass. The latter was obtained in 20 samples representative of the approximately 4.5 m diameter drip area at the stem base with a stainless steel drill (no brand name, 10 cm diameter, 50 cm long). Carbon stored in the soil (CSS; Mg ha-1) was quantified in 20 soil subsamples per plant using soil content (% CS), bulk density (BD) and depth (D) of sampling (1 to 50 cm), with the following relation: CSS = % CS*BD*D (Ávila et al., 2001). For this estimation, the technique used was that reported by Leblanc et al. (2006), modified because soil BD was determined at 3,927 cm3 in a sample (extracted with a stainless steel drill). Of each sample, roots were separated; CSS and C in roots were determined with the modified wet combustion method of Walkley and Black (Kaurichev et al., 1984; Schlichting et al., 1995).
Total biomass (Y) per plant was the sum of biomass from stem, leaves and root. In stem, it was determined with the formula Y = Volume (V) x Density (D) because the stem shape is conical and irregular, and the diameter of the base is larger than the upper diameter. The volume of each section and total volume was calculated with the above equation. Brown (2001) indicated that the biomass of the leaves accounts for 65 % of the stem weight. This relationship was used to estimate sample biomass and the total leaf biomass. To corroborate this value, V and D of the five leaves of each plant were determined after they were completely shredded and weighed. With this information, values of the leaves per plant were calculated.
Determination of specific density
Specific density (g cm-3) of stems, roots, soil, subsamples of stem sections and leaves was determined with the ratio dry weight:volume of fresh tissue. Dry weight was obtained with the methodology described above, and the volume of fresh tissue was determined by the method of water displacement. Specific density of sections, leaves and roots were compared with the Tukey test (SAS Institute Inc., 2009). The total sum of stem (Yf), canopy (Yc), and root (Yr) biomass was the total biomass (Y), Y = Yf + Yc +Yr of each plant (Acosta et al., 2002).
Determination of the allometric model
Average biomass of plants of the sites were analyzed together with an equation that would permit interpretation of plant development. To this end, a simple regression test was conducted following Aquino-Ramírez et al. (2015). A linear regression analysis was included with indicator variables, in this, case biomass, amount of C and DBH. With the data of total biomass (Y) and total height (X), the linear equation was fit from the potential expression. The model was fit by minimum squares, and the order to origin (b) and slope (k) were obtained according to Avendaño et al. (2009) and Acosta et al. (2002). The resulting model was Y = bX k (potential equation), where Y is total biomass, X is the diameter at breast height, and b and k are the function parameters. The equation was fit to a linear expression by transformation to natural logarithm of Y and X; thus, the expression of the equation in its linear form was ln (Y) = ln (b) + k ln (X) (Eq. 2) where b and k are the parameters to be estimated.
Determination of carbon content
The amount of C in stem, leaf and root biomass was determined by the modified Walkley and Black wet combustion method, based on incomplete oxidation of C, with an oxidizing mixture of K2Cr2O7, H2SO4 and heat by aqueous acid dilution. Boiling between 110 and 130 ºC depends on initial temperature, organic matter content and size of the reaction recipient. The amount of oxidizing agent consumed in the reaction was determined by spectrophotometry (spectrophotometer METASH, Mod. UV-6000, China), by measuring initial intensity of green Cr+3 and yellow K2Cr2O7 ions, and a sucrose calibration curve. In the reaction, the Cr+3 ions are proportional to the amount of oxidized C, according to the reaction: C6H12O6 + 4 K2Cr2O7 + 16 H2SO4 → 4 K2SO4 + 4 Cr2 (SO4)3 + 6 CO2 + 22 H2O.
Five mL of K2Cr2O7 and 10 mL sulfuric acid were added to 0.02 g dry sample; it was left to cool for 30 min and 35 mL distilled water was added. The mixture was shaken after each addition and left to stand overnight. Absorbance was measured at 590 nm. The total content of each sample was multiplied by the percentage of C obtained by spectrophotometry and by the sample biomass; the total C content per plant would be the sum of the total C content in stem, branches and roots. To obtain the allometric equation and estimate C, the allometric model that relates the content of C to plant height was used (Swift, 2000; Avendaño et al., 2009). Descriptive statistical analysis and correlation tests (α = 0.05) were applied to the data.
Results and Discussion
Biomass estimation
The general average of stem, leaf and root biomass was 1104.34 ± 87.85 kg. Plants from Huehuetán had more biomass than plants from Tapachula and Villa Comaltitlán. The plants of the two latter sites had higher density (Table 1). These values were not reflected in total biomass, since average DBH and height were larger in Huehuetán than in other sites. According to the information provided by growers, variability of the field data is due to differences in: 1) agronomic management of the crop, such as the application of essential macro and micro minerals; 2) water supply: drought in high areas and flooding in low areas and in areas close to the sea; and 3) environmental conditions, such as temperature and precipitation. This coincides with Corley and Gray (1982). All the factors mentioned are determinant for the crop since maximum development is achieved after 6 to 12 years, although the plant can continue growing at a rate of 35 to 75 cm per year, depending on environmental conditions (Corley and Gray, 1982; Cayón, 1999). In our study, the plants evaluated were about 12-years-old.
Table 1 Dasometric characteristics of oil palms (Elaeis guineensis Jacq) in Soconusco, Chiapas, México.
| Planta número | DAP (cm) |
Altura de planta
(m) |
Densidad de
madera (g cm-3) |
Biomasa total
(kg) |
Carbono total
(%) |
Cantidad de carbono
(kg planta-1) |
| Sitio 1. Tapachula | ||||||
| 1 | 83.25 | 3.25 | 0.16 | 1052.01 | 47.34 | 498.02 |
| 2 | 55.54 | 2.66 | 0.14 | 1027.77 | 46.78 | 480.79 |
| 3 | 100.03 | 4.56 | 0.15 | 1208.35 | 45.36 | 548.11 |
| 4 | 72.25 | 3.45 | 0.13 | 1068.5 | 48.25 | 484.67 |
| Promedio | 77.77 | 3.48 | 0.15 | 1089.16 | 46.93 | 502.90 |
| σ | 18.71 | 0.79 | 0.01 | 81.20 | 1.21 | 31.03 |
| Sitio 2. Huehuetán | ||||||
| 1 | 62.35 | 3.01 | 0.10 | 1087.07 | 47.56 | 517.01 |
| 2 | 86.36 | 3.96 | 0.10 | 1076.18 | 51.25 | 551.54 |
| 3 | 96.25 | 4.25 | 0.10 | 1095.07 | 46.35 | 507.56 |
| 4 | 115.45 | 4.55 | 0.12 | 1325.45 | 51.23 | 614.35 |
| Promedio | 90.10 | 3.94 | 0.11 | 1145.94 | 49.10 | 547.62 |
| σ | 22.09 | 0.67 | 0.01 | 119.92 | 2.52 | 48.34 |
| Sitio 3. Villa Comaltitlán | ||||||
| 1 | 66.58 | 3.25 | 0.24 | 1064.06 | 42.34 | 450.52 |
| 2 | 96.25 | 4.69 | 0.25 | 1155.49 | 52.36 | 605.01 |
| 3 | 45.36 | 3.25 | 0.23 | 1006.74 | 54.25 | 546.16 |
| 4 | 86.25 | 3.89 | 0.25 | 1085.36 | 58.25 | 588.81 |
| Promedio | 73.61 | 3.77 | 0.24 | 1077.91 | 51.80 | 547.63 |
| σ | 22.51 | 0.68 | 0.01 | 61.46 | 6.77 | 69.33 |
| Promedio general | 80.49 | 3.73 | 0.16 | 1104.34 | 49.28 | 532.71 |
| σ | 20.50 | 0.68 | 0.06 | 87.85 | 4.35 | 51.92 |
DAP: diameter at breast height, CD: diametric category. The biomass of each palm was obtained by destructive sampling. The amount of C is an average of stem, leaves and roots of each plant.
The linear allometric model was Y = 3.571X +816.86 (R2 = 0.695) and Y = 98.349X + 737.41 (R2 = 0.577) to determine biomass based on DBH and plant height, respectively (Figures 1A and 1B). The coefficients of determination for both variables reduce reliability of the equations to determine biomass in 12-year-old palm plants, but they are not ruled out. We suggest increasing the number of replications and older plants to estimate applicable allometric equations (Thenkabail et al., 2004: Leblanc et al., 2006). However, for growers, it is difficult to provide plants for sampling. For this reason, the number of plants for destructive sampling was reduced in the Soconusco region. The proportion of root biomass and shoot biomass in this study was 0.06, which is notably low relative to other studies.
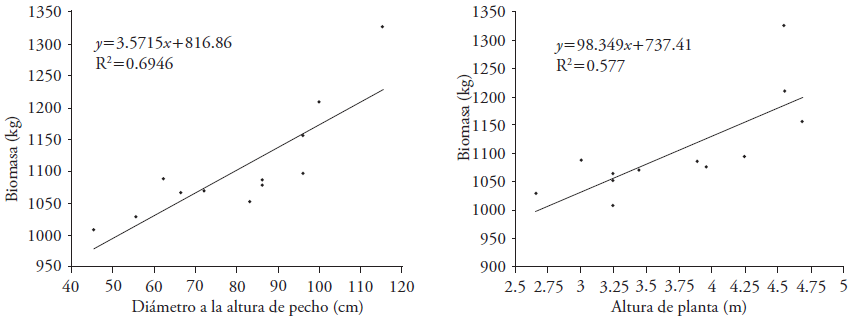
Figure 1 Dispersion of the measured values and regression line generated with the linear model to estimate biomass based on diameter at breast height (DAP) and total height of E. guineensis Jacq.
The allometric equations and similar transformations were reported in Abies religiosa (Kunth) Schltdl. & Cham. (Avendaño et al., 2009) and other tree species, such as Cupania dentata DC., Alchornea latifolia Sw. and Inga punctata Willd (Aquino-Ramírez et al., 2015). Therefore, the results of our study still do not corroborate the fit of the allometric model to determine biomass and sequestered C if DBH is known. However, by using plant height the model could be more precise. For this reason, it is necessary to conduct more studies with more samples to approach the acceptable models used in the literature with other species.
The relationship between DBH and plant height was positive (Figure 2). The linear equation was height (m) = 0.029(DBH) + 1.439 and R2 = 0.762. This equation can thus be used to estimate plant height (m) with data on DBH (cm) of oil palm in the wet tropical region of Chiapas.
Allometric equation to determine carbon stored in oil palm plants
The amount of C can depend on multiple factors. Leblanc et al. (2006) pointed out that among the main factors are nutrient management, plant age, and climatic factors such as precipitation, and type of soil. In our study, regardless of the variability of the determined values, the quantities of C were not different among sites nor among plants (p > 0.05). Oil palms about 12 years old at a density of 143 plants ha-1 in the Soconusco region has the potential to store an average of 76.18 ± 0.047 Mg C ha-1 and the annual rate of fixation is 6.53 Mg C ha-1. The values per hectare and the fixation rate were higher than the 42.87 Mg C ha-1 in plants reported by Khalid et al. (2000) and 31.21 Mg C ha-1 in 12-year-old plants reported by Rodríguez et al. (2000)6. In contrast, Thenkabail et al. (2004) found 14.75 and 14.94 Mg C ha-1 using satellite images (IKONOS Satellite Images), but this technique differs from that applied in our study. Pulhin et al. (2014) used a system of destructive sampling like that of our study and reported 55.0 Mg C ha-1, a C fixing potential of 6.1 Mg C ha-1 year-1, similar to that found in our study.
Table 2 Carbon stored (kg) in oil palm plants (E. guineensis Jacq.) in Soconusco, Chiapas, México.
| Sitio | Planta (número) | Tallo | Hojas | Raíz | Total |
| Tapachula | 1 | 281.12 | 185.40 | 31.50 | 498.02 |
| 2 | 244.95 | 209.06 | 26.77 | 480.79 | |
| 3 | 298.86 | 223.74 | 25.51 | 548.11 | |
| 4 | 259.22 | 204.68 | 20.77 | 484.67 | |
| Promedio (σ) | 271.04±23.8 | 205.72±15.8 | 26.13±4.4 | 502.90±31.0 | |
| Huehuetán | 1 | 297.71 | 193.51 | 25.79 | 517.01 |
| 2 | 317.85 | 208.46 | 25.23 | 551.54 | |
| 3 | 271.59 | 215.01 | 20.96 | 507.56 | |
| 4 | 330.06 | 255.97 | 28.32 | 614.35 | |
| Promedio (σ) | 304.30±25.6 | 218.24±26.7 | 25.08±3.1 | 547.62±48.3 | |
| Villa Comaltitlán | 1 | 261.82 | 165.74 | 22.97 | 450.52 |
| 2 | 349.96 | 228.81 | 26.24 | 605.01 | |
| 3 | 324.62 | 193.86 | 27.68 | 546.16 | |
| 4 | 336.97 | 222.48 | 29.36 | 588.81 | |
| Promedio (σ) | 318.34±39.1 | 202.72±28.9 | 26.56±2.7 | 547.63±69.3 |
Average (σ): average value by sampling site ± standard deviation.
With the total C content (stem, leaves and root) of the 12 sampled plants, allometric equations were generated to estimate the amount of stored C relative to plant height since, when DBH was used, the coefficient of determination (R2) was not acceptable. The equation with the best fit was the linear model (y = β0 + β1x) (Figure 3). The small number of field samples could have determined the low coefficient of determination for the linear equation between the amount of C and plant height; this improved when the evaluation was between the amount of C and DBH.
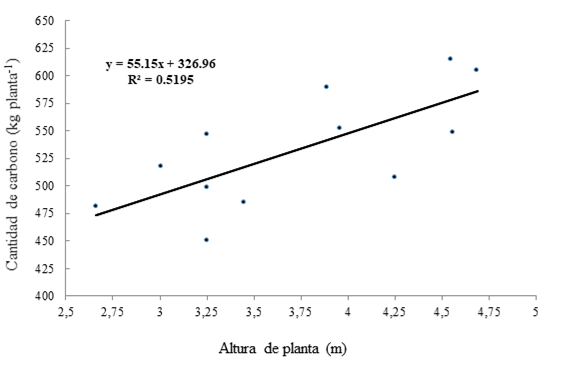
Figure 3 Dispersion of observed values for estimation of C in oil palm plants (E. guineensis Jacq.) and the generalized regression line with the linearized model.
In Central America and Asia, cultivation of oil palm has the potential to sequester C, with values of 96.02 and 103.35 Mg C ha-1, in the 0 to 30 cm and 0 to 50 cm soil profiles (Leblanc et al., 2006; Ibrahim et al., 2007). These quantities surpass those we registered in the root fraction and soil in our study. Leblanc et al. (2006) and Ibrahim et al. (2007) reported sequester of 22.68 Mg C ha-1 in shoot biomass, lower than the average of C determined in the thee municipalities in the Soconusco region. Thus, the C stock in oil palm could increase with the interactions of factors, such as favorable geographic location for this crop, annual precipitation of 1800 to 2200 mm, temperatures of 25 to 28 ºC, suitable soils (site 1, Cambisols and Acrisols; site 2, Cambisols and Solonchak; site 3, Feozen, Cambison and Solonchak), which are deeper and salty because of proximity to the Pacific coast (INEGI, 2010). Specifically, agronomic management of the plantation with applications of mixtures of micro and macro nutrients necessary for adequate crop development could be determinant in carbon sequester.
In the literature, quantities from 18.2 to 57.3 Mg C ha-1 sequestered by sugarcane agricultural production systems were reported in Saccharum oficinarum L. Variability is determined by agroclimatic factors and soil quality (Aryal et al., 2014). In other agricultural systems in Chiapas C sequester (Mg C ha-1) was estimated at 116.8 in secondary shrub, 46.7 in plantations of live fences, 116.8 in livestock grazing pastures, 130.2 in coffee plantations and fences and 180.8 in milpas with paths. The highest sequester was recorded in the trees established in each of these systems and recycling was through wood formation (Alegre et al., 2004; Lapeyre et al., 2004; Díaz-Franco et al., 2007). In annual crops, the sequester rate is 0.1 to 0.8 Mg C ha-1 for maize-bean systems. These systems generate higher emissions of greenhouse effect gases (GEG) when agrochemicals are used and when harvest residues are burned. In grasslands used for livestock grazing C sequester is 0.1 to 0.2 Mg de C ha-1; it is 4 to 8 Mg C ha-1 in reforested areas, and 0.2 to 3.1 Mg C ha-1 in agrosilvopastoral systems (crops plus poaceae).
Although methods, protocols and management systems do exist, it is necessary to standardize the processes to determine and calculate emissions and C sequester to have real, reproducible and comparable results and to follow the C footprint in the crop. The method in this study can be reliable for those estimations in E. guineensis in Chiapas, as it has been applied in other forest species (Castilla, 2004; Avendaño et al., 2009).
Conclusions
The potential of the oil palm to sequester carbon is linked to its potential to form biomass, and it is an important crop in mitigating climate change. The greatest accumulation of this element is in the stem, followed by the branches and roots. The technique used allowed estimation of biomass and carbon sequestered by this crop. The allometric equations generated were not reliable for prediction; thus, the number of replications or samples should be increased to reduce the error in sampling based on DBH and even on plant height. The evaluation technique used in this study generated the basis for evaluating accumulation of carbon in the aboveground and underground fraction of the plant in the wet tropics of southeastern Mexico.
REFERENCES
Acosta, M. M., J. J. Vargas, A. M. Velázquez, y J. D. Etchevers. 2002. Estimación de la biomasa aérea mediante el uso de relaciones alométricas en seis especies arbóreas en Oaxaca, México. Agrociencia 36: 752-736. [ Links ]
Alegre, J., L. Arévalo, A. Ricse, D. Callo-Concha, y C. Palm. 2004. Secuestramiento de carbono con sistemas alternativos en el Perú. In: Muller, M., A. Gama-Rodrigues, I. Silva Fontes, e M. Carvalho (eds). Sistemas Agroflorestais, Tendencia da Agricultura Ecologica nos Tropicos, Sustento da Vida e Sustento de Vida. Sociedadee Brasileira de sistemas Agroflorestais, Comissao Executiva do plano da lavoura cacauria e Universidade Estadual do Norte Fluminense, Ilehues- BA. pp: 27-32. [ Links ]
Andrade, H. J., R. Brook, and M. Ibrahim. 2008. Growth, production and carbon sequestration of silvopastoral systems with native timber species in the dry lowlands of Costa Rica. Plant Soil 308: 11-22. [ Links ]
Aquino-Ramírez, M., A. Velázquez-Martínez, J. F. Castellanos-Bolaños, H. De los Santos-Posadas, y J. D. Etchevers-Barra. 2015. Partición de la biomasa aérea en tres especies arbóreas tropicales. Agrociencia 49: 299-314. [ Links ]
Aryal, D. R., B. H. De Jong, S. Ochoa-Gaona, L. Esparza-Olguin, and J. Mendoza-Vega. 2014. Carbon stocks and changes in tropical secondary forests of Southern Mexico. Agric. Ecosys. Environ. 195: 220-230. [ Links ]
Avendaño, D. M., M. M. Acosta, F. A. Carrillo, y J. D. Etchevers. 2009. Estimación de biomasa y carbono en un bosque de Abies. Fitotec. Mex. 32: 233-238. [ Links ]
Ávila, G., F. Jiménez, J. Beer, M. Gómez, y M. Ibrahim. 2001. Almacenamiento, fijación de carbono y valoración de servicios ambientales en sistemas agroforestales en Costa Rica. Agrofor. Am. 8: 32-35. [ Links ]
Brown, S. 2001. Measuring carbon in forests: current status and future challenges. Environ. Poll. 116: 363-372. [ Links ]
Castilla, C. 2004. Potencial de captura de carbono por la palma de aceite en Colombia. Palmas 25: 366-371. [ Links ]
Cayón, S. D. G. 1999. Apuntes sobre fisiología del crecimiento y desarrollo de la palma de aceite (Elaeis guineensis Jacq.). Palmas 20: 43-54. [ Links ]
Corley, R.V.H., and B. S. Gray. 1982. Growth and morphology. In: Corley, R.V.H., J. J. Hardon, and B. J. Wood (eds). Oil Palm Research. Elseiver, Amsterdam. pp: 7-21. [ Links ]
Derner, J. D., and G. E. Schuman. 2007. Carbon sequestration and rangelands - a synthesis of land management and precipitation effects. J. Soil Water Conserv. 62: 77-85. [ Links ]
Díaz-Franco, R., M. Acosta-Mireles, F. Carrillo-Anzures, E. Buendía-Rodríguez, E. Flores-Ayala, y J. D. Etchevers-Barra. 2007. Determinación de ecuaciones alométricas para estimar biomasa y carbono en Pinus patula Schl. et Cham. Madera y Bosques 13: 25-34. [ Links ]
Follett, R. F., and D. Reed. 2010. Soil carbon sequestration in grazing lands, societal benefits and policy implications. Rangel. Ecol. Manage. 63: 4-15. [ Links ]
Hairiah, K., S. Dewi, F. Agus, S. Velarde, A. Ekadinata, S. Rahayu, and M. van Noordwijk. 2011. Measuring Carbon Stocks Across Land Use Systems: A Manual. Bogor, Indonesia. World Agroforestry Centre (ICRAF), SEA Regional Office, 154 pages. http://www.worldagroforestry.org/sea/Publications/files/manual/MN0050-11/MN0050-11-1.pdf (Consulta: agosto 2016). [ Links ]
Ibrahim, M., M. Chacón, C. Cuartas, J. Naranjo, G. Ponce, P. Vega, F. Casasola, y J. Rojas. 2007. Almacenamiento de carbono en el suelo y la biomasa arbórea en sistemas de usos de la tierra en paisajes ganaderos de Colombia, Costa Rica y Nicaragua. Agrofor. Am. 47: 27-36. [ Links ]
Kaurichev, I. S., N. P. Panov, M. V. Stratonovich, I. P. Grechin, V. I. Savich, N. F. Ganzhara, y A. P. Mershin. 1984. Prácticas de Edafología. Ed. Mir, Moscú, URRS. 287 p. [ Links ]
Khalid, H., Z. Z. Zakaria, y J. M. Anderson. 2000. Cuantificación de la biomasa de la palma de aceite y su valor nutritivo en una plantación desarrollada. I. La biomasa encima del suelo. Palmas 21: 67-77. [ Links ]
Lapeyre, T., J. Alegre, y L. Arévalo. 2004. Determinación de las reservas de carbono de la biomasa aérea, en diferentes sistemas de uso de la tierra en San Martín, Perú. Ecol. Apl. 3: 35-44. [ Links ]
Leblanc H., R. Russo, J. J. Cueva, y E. Subía. 2006. Fijación de carbono en palma aceitera en la región tropical húmeda de Costa Rica. Tierra Trop. 2: 143-148. [ Links ]
NOAA (National Oceanic and Atmospheric Administration). 2016. Trends in atmospheric carbon dioxide concentration. NOAA. http://www.esrl.noaa.gov/gmd/ccgg /trends/ . (Consulta: septiembre 2016). [ Links ]
Pacheco, E. F. C., A. Aldrete, A. G. Gómez, A. M. Fierros, A. B. Cetina, y H. H. Vaquera. 2007. Almacenamiento de carbón en la biomasa aérea de una plantación joven de Pinus greggii Engelm. Rev. Fitotec. Mex. 30: 251-254. [ Links ]
Prentice, I. C., G. D. Farquhar, M. J. R. Fasham, M. Goulden, M. Heimann, V. J. Jaramillo, H. S. Kheshgi, C. Le Quéré, R. J. Scholes, and D. W. R. Wallace. 2001. The carbon cycle and atmospheric carbon dioxide. In: Houghton, J. T. (ed.). Climate Change: The Scientific Basis. Contribution of Working Group I to the Third Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press. Cambridge and New York, USA. pp: 183-237. [ Links ]
Pulhin, F. B., R. D. Lascob, and J. P. Urquiolab. 2014. Carbon sequestration potential of oil Palm in Bohol, Philippines. Ecosys. Develop. J. 4: 14-19. [ Links ]
SAS Institute, Inc. 2009. SAS/STAT User’s Guide: Statistics. 8th ed. SAS Institute, Inc. Cary, US. 846 p. [ Links ]
Schlichting, E., H. P. Blume, and K. Stahr. 1995. Bodenkundliches Praktikun, Berlin/Wien. Blockwell. Wissenschafts-Verlag. 295 p. [ Links ]
Smith, T. M., T. C. Peterson, J. N. Lawrimore, and R. W. Reynolds. 2005. New surface temperature analyses for climate monitoring. Geophysical Research Letters, Vol. 32, L14712. http://www.ncdc.noaa.gov/oa/climate/research/Smith-comparison.pdf (Consulta: febrero 2016). [ Links ]
Swift, R. S. 2001. Sequestration of carbon by soil. Soil Sci. 166: 858-871. [ Links ]
Thenkabail, P. S., N. Stucky, B. W. Griscom, M. S. Aston, J. Diels, B. Van Deer Meer, and E. Enclona. 2004. Biomass estimation and carbon stock calculations in the oil palm plantations of African derived savannas using IKONOS data. Int. J. Remote Sens. 25: 5447-5472. [ Links ]
5Towards new scenarios for analysis of emissions, climate change, impacts and response strategies. Informe de la reunión de expertos del IPCC celebrada del 19 al 21 de septiembre de 2007 en Noordwijkerhout (Países Bajos), Grupo Intergubernamental de Expertos sobre el Cambio Climático, Ginebra, Suiza. 132 p.
Received: March 2017; Accepted: July 2017











 texto en
texto en 



