Services on Demand
Journal
Article
Indicators
Related links
Share
Revista de la Sociedad Química de México
Print version ISSN 0583-7693
Rev. Soc. Quím. Méx vol.47 n.4 Ciudad de México Oct./Dec. 2003
Investigación
Layered Silicates and Mesostructured Aluminosilicates Promoters of Benzyltoluenes
Bernardo Francisco,1* Gabriel A. Arroyo,1 Olivia Noguez,1 Juan M. Aceves,1 José M. Domínguez,2 Francisco Delgado,3 René Miranda1
1 Facultad de Estudios Superiores Cuautitlán, Universidad Nacional Autónoma de México, Av. 1 de Mayo s/n Cuautitlán Izcalli, Estado de México, C. P. 54740 tel/fax 00 52 5 6232056. e-mail garroyo@servidor.unam.mx.
2 Instituto Mexicano del Petróleo, Eje Lázaro Cárdenas 152 Gerencia de Competencia. 07730 México D.F.
3 Escuela Nacional de Ciencias Biológicas, Instituto Politécnico Nacional, Prolongación Carpio y Plan de Ayala, Col. Casco de Santo Tomás, México, D. F., CP 11340, Tel 7-29-63-00 ext. 62413. e-mail fdelgado@woodward.encb.ipn.mx.
Recibido el 15 de agosto del 2003.
Aceptado el 14 de octubre del 2003.
Abstract
Several hydrous layered silicates, modified kanemites, aluminosilicates and various mesoporous materials, were hydrothermally synthesized, and then evaluated as new catalysts for obtaining benzyltoluenes. In addition, the dependence of the catalyst concentration, the influence of the reaction time and the effect of the temperature were studied.
Key words: Layered silicates, kanemites, catalysis, benzyltoluenes.
Resumen
Varios materiales mesoporosos como los silicatos laminares hidratados y kanemitas modificadas fueron obtenidos hidrotérmicamente y posteriormente evaluados como nuevos catalizadores en la obtención de benciltoluenos. Asimismo, se estudia la dependencia de la concentración del catalizador, la influencia del tiempo de reacción y el efecto de la temperatura.
Palabras clave: Silicatos laminares, catálisis, kanemitas, benciltoluenos.
Introduction
The remarkable ability of aluminesilicates to perform interesting transformations has found widespread applications and uses in research chemistry and industrial areas [1]. Mainly these materials have acquired important interest in the catalytical field due to their usefulness and high activity. Therefore, this kind of material are of current interest and many groups among us [2], are now working in this area in order to seek for novel catalysts as well as their corresponding applicative alternatives.
The family of hydrous layered silicates includes natural minerals such as kanemite [3], NaHSi2O4(OH)2 2H2O, a sodium hydrated silicate with laminated structure that shows large possibilities in catalysis due to its structural and superficial characteristics as well as to its facile synthesis [4-6].
In relation to this subject and due to the importance that represents for the industry, this paper deals with the potential of kanemite and mesoporous materials to induce the one pot promotion of benzyltoluenes from simple reagents: toluene and benzylchloride. It is worth mentioning, that the target molecules are largely employed as insulating oils in high-voltage electrical devices [7], and as high octane fuels for aircraft engines [8], among others uses [9].
These new catalytic materials offer considerable advantages over conventional homogeneous acid catalysts; in particular, the products are obtained after simple filtration of the catalyst. Conversely, the common Friedel-Crafts alkylation using typical Lewis acids, generally affords complex reaction mixtures requiring laborious work-up to isolate the products [10]; furthermore, they are highly toxic, pollutants of the environment and of high-cost [11]. Thus, layered silicates and aluminosilicates represent an important alternative as new heterogeneous catalysts for the production of the target molecules.
Results and Discussion
The results of several experiments performed with various layered silicates and mesostructured aluminosilicates in order to obtain the benzyltoluenes are summarized in the table 1; it is note worthy that the reaction crude mixture does not showed by-products, as detected by GC-MS. In general, good yields and pure compounds were obtained at low reaction times, being the work-up procedures very simple. Therefore, these new options are highly competitive with those methods previously informed in the literature. These results, in addition to the easy preparation of the kanemites and the mesoporous material led us to conclude that these catalysts are interesting alternatives for the production of ortho and para-benzyltoluenes.
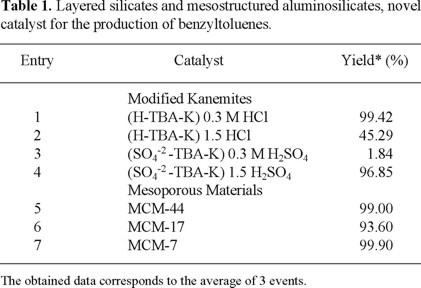
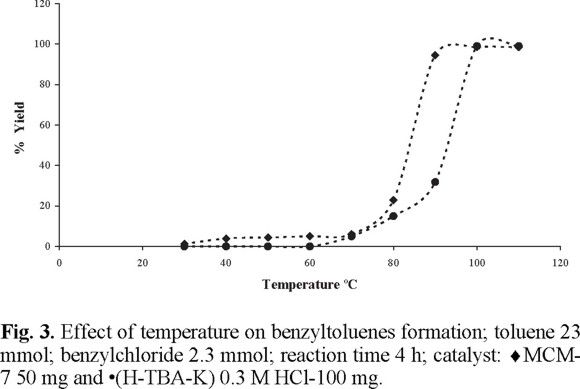
Finally, the effect of the catalyst concentration, the time and the temperature of reaction were investigated in the cases of MCM-7 and (H-TBA-K) 0.3 M HCl, in order to establish the corresponding optimum reaction conditions for these two novel catalysts. Thus the results of the dependence of catalyst concentration, the role of the reaction time and the effect of the temperature on the promotion of the benzyltoluene are reported in figures 1-3 (2) respectively. As can be seen, the mesoporous material (MCM-7) is a higher effective Lewis - catalyst to promote these new options of Friedel-Crafts toluene alkylation, since it required less concentration, 50 mg, as well as lower temperatures and reaction times to reach better yields, all this in comparison to the kanemitic moiety, (H-TBA-K) 0.3 M HCl.

Experimental
General. Toluene (Aldrich) was dried prior to use (Na/benzophenone). The benzylchloride was also purchased from Aldrich and used as received. The modified kanemites and mesoporous materials were synthesized using previously reported procedures [4,12]. Purified products were characterized by spectroscopic means: 1H NMR spectra were recorded in a Varian FT-200 spectrometer using CDCl3 as solvent and TMS as internal reference; EIMS (70 ev) spectra and GC-MS analysis were obtained using a JEOL JMS AX505HA mass spectrometer. For the optimization of the experiments a Polyscience Corporation Temperature Controller Model 73 was employed, the corresponding formation in percent data were acquired by a Varian Gas Chromatographer 3400 equipped with a flame ionization detector and a 30 m x 0.53 mm column packed with polyethylenglicol, the relative proportion of the products were calculated assuming that the detector gave equal response for each experiment.
Benzyltoluenes. To 2.5 ml of anhydrous toluene were added 0.25 ml of benzylchloride and 100 mg of catalyst, followed by magnetic stirring under reflux during 4 hrs. The reactions were monitored by GC-MS until the total disappearance of the reagent. The reaction mixture was filtered on a celite bed, washed with NaHCO3 (5 %, 3 × 10 ml), the organic phase was dried (Na2SO4 anh.), and the remaining toluene was eliminated under reduced pressure. Finally the product was purified by column chromatography. The mixture of ortho and para -benzyltoluenes was characterized by spectroscopic means and compared with reported data(1): 1H NMR (CDCl3, TMS, 200 MHz) δ 2.20 (s, 3H), 2.32 (s, 3H), 3.92 (s, 2H), 4.00 (s, 2H), 7.2 (m, 9H); EIMS (70 ev) m/z (%) 182 (48) M+·, 91 (100) [C7H7]+.
Acknowledgement
Financial support from DGAPA-UNAM (PAPIIT-IN215598) is acknowledged.
References
1. Meurig-Thomas, J. Scientific American 1992, 266, 112-118. [ Links ]
2. Miranda, R.; Arroyo, G. A.; Penieres, G.; Salmón, M.; Cabrera, A.; Álvarez, C.; Delgado, F. Research Trends 2003, in press. [ Links ]
3. Johan, Z.; Maglione, G. F. Bull. Soc. Fr. Mineral. Cristallogr. 1972, 95, 371-382. [ Links ]
4. Terres, E.; Domínguez, J. M. "Synthesis and Modifications Of Layered Silicates: Kanemite and Megadite" Symp. Synthesis of Zeolites, Layered Compounds and Other Microporous Materials, Div. Petr. Chem. Inc. 209th Natl. Meet. ACS. Anaheim CA., U.S.A. april 2-7, 1995, 261. [ Links ]
5. Yanigisawa, T.; Shimizu, T.; Kuroda, K.; Kato, C. Bull. Chem. Soc. Jpn. 1990, 988. [ Links ]
6. Wong, S. T.; Cheng, S. Chem. Mater. 1993, 5, 770. [ Links ]
7. Berger, N.; Jay, P. IEEE Trans. Electr. Insul. 1986, EJ-21, 59. [ Links ]
8. Lamneck, J. H.; Wise, P. H. Naatl, Adris, Comm. Aeronaut, Tech. Notes, 1950,17, 2230. [ Links ]
9. Fu, X.; He, M.; Lei Q.; Luo, B. Synth. Commun. 1991, 21, 1273. [ Links ]
10. Berger, N.; Jay, P. IEEE Trans. Electr. Insul. 1986, EJ-21, 59. [ Links ] Lamneck, J. H. Wise, P. H. Naatl, Adris, Comm. Aeronaut Tech. Notes. 1950, 17, 2230. [ Links ]
11. Salmón, M.; Miranda, R.; Angeles, E. J. Chem. Soc., Chem Commun. 1990, 1188; [ Links ] High Lights Chem. and Ind. 1991, 26. The Janssen Chimica Catalog / Handbook of Fine Chemicals for Research and Industry 1993-1994, 23, 317-37. [ Links ] Miranda, R.; Aceves, J. M.; Corona-Cortés, H.; Domínguez, J. M.; Cabrera A.; Salmón, M. Synth. Commun. 1994, 24, 727. [ Links ]
12. Terrés, E.; Ramírez, S. S. Domínguez, J. M. Montoya, A.; Navarrete, J.; Gómez-Cisneros, M. Microporous and Macroporous Materials; Materials Research Society; Symposium Proceedings 1996, 431, 111; Symposium April 8-11, , San Francisco CA, USA. [ Links ]





![Síntesis y evaluación del efecto ansiolítico de un nuevo derivado pirimido[1,6-a]pirimidínico](/img/en/next.gif)








