Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Hidrobiológica
versão impressa ISSN 0188-8897
Hidrobiológica vol.15 no.2 Ciudad de México 2005
Artículo
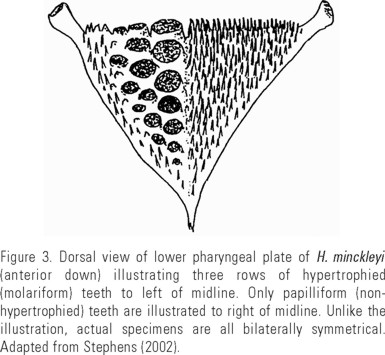
Habitat Segregation among Trophic Morphs of the Cuatro Ciénegas Cichlid (Herichthys minckleyi)
Segregación de hábitat entre formas tróficas de la mojarra de Cuatro Ciénegas (Herichthys minckleyi)
Adam E. Cohen1, Dean A. Hendrickson1,2*, Camille Parmesan1 and Jane C. Marks3
1 Section of Integrative Biology, 1 University Station #C0930, The University of Texas at Austin, Austin, Texas 78712 USA.
2 Texas Memorial Museum, Texas Natural History Collections, The University of Texas at Austin, PRC 176 / R4000, 10100 Burnet Road, Austin, TX 78758-4445 USA.
3 Department of Biological Sciences, Northern Arizona University, Box 5640, Flagstaff, AZ 86011-5640 USA.
Recibido: 4 de octubre 2004.
Aceptado: 31 de mayo de 2005.
Abstract
Herichthys minckleyi is an endangered, trophically polymorphic cichlid endemic to the Cuatro Ciénegas basin of Coahuila, Mexico. A molariform morph has stout pharyngeal teeth whereas a papilliform morph has numerous fine pharyngeal teeth. Individuals with intermediate pharyngeal dentition also exist, as does yet another morph, called piscivore. Previous studies indicated that morphs utilize different food sources, thus suggesting morph-specific spatial segregation, since food resource availability is spatially heterogeneous. We present data from an observational study of all morphs (but focusing on the three most common, normal-bodied ones - molariform, papilliform and intermediate) in a single spring pool, Poza Mojarral Oeste. We analyzed morph distribution in relation to habitat types, and document morph-specific differences in feeding behavior. Spatio-temporal habitat partitioning was also investigated. Habitat use by molariform, papilliform, and intermediate morphs was found to be non-random. Morphs differed in habitat use, albeit with considerable overlap. Strong segregation among morphs was not detected in any season or time of day, but habitat use patterns varied seasonally within each morph and were consistently different among morphs. All morphs behave basically as feeding generalists. This endangered species may prove more difficult to manage than other, non-polymorphic species. It is clearly important to manage not only for the maintenance of the species, but also for maintenance of its different morphs, which our study indicates may each require different mixes of habitat types. We thus hypothesize that any changes in habitat heterogeneity will lead to altered proportions of the different morphs of the species.
Key words: Habitat partitioning; resource partitioning; intraspecific interactions; trophic polymorphism; Cuatro Ciénegas.
Resumen
Herichthys minckleyi, una mojarra en peligro de extinción endémica de Cuatro Ciénegas, Coahuila, es tróficamente polimórfica. La forma llamada molariforme tiene dientes faríngeos robustos, mientras que la papiliforme los tiene numerosos y finos. También existen individuos con dentición intermedia, así como una tercera forma, llamada piscívora. Los estudios previos indicaban que cada forma utiliza distintos recursos alimentarios, lo que sugiere una segregación espacial específica para cada forma, ya que la disponibilidad espacial de los alimentos es heterogénea. Se presenta un estudio observacional de todas las formas (aunque con énfasis en las tres más comunes: molariforme, papiliforme e intermedia) en la Poza Mojarral Oeste. Analizamos la distribución de las formas en función del hábitat y documentamos diferencias específicas para cada forma en conducta alimentaria. Se investigó también el reparto espacio-temporal del hábitat. El uso de hábitat resultó no-aleatorio. Las formas difirieron en uso de hábitat, aunque con traslape considerable. No se detectó segregación entre las formas por época del año ni hora del día, pero el uso de hábitat varió estacionalmente para cada forma, y fue diferente entre ellas. Todas las formas son básicamente generalistas tróficos. El manejo de esta especie en peligro puede resultar más difícil que para especies no-polimórficas. Será importante buscar no sólo la permanencia de la especie, sino también la de sus diversas formas, para lo cual se requerirán diferentes mezclas de tipos de hábitat. Predecimos que cualquier cambio en la heterogeneidad del hábitat causará alteración en las proporciones de las diferentes formas de esta especie.
Palabras clave: Reparto de habitat; reparto de recursos; interacciones intraespecíficas; polimorfismo trófico; Cuatro Ciénegas.
Introduction
The Cuatro Ciénegas Valley is a unique desert ecosystem with unusually high levels of endemism. Minckley (1969) described many aspects of the valley, its geology and aquatic biota. In north central Coahuila, Mexico, this 700-km2 valley lies at about 740 m above sea level and harbors minimally several hundred springfed pools ranging in area from a few square meters to several hectares. Water temperatures are highly variable among pools and warm and cold springs often emerge within a few meters of each other, coalescing into streams, rivers, and marshes. High levels of endemism in both terrestrial and aquatic communities, as well as imminent threats in the form of dewatering and exotic species, make conservation an important issue in what is now a federally declared Natural Protected Area for Flora and Fauna (Secretaría de Desarrollo Social, 1994).
The endemic Cuatro Ciénegas cichlid, Herichthys minckleyi (Kornfield & Taylor, 1983) is one of the most common fishes in the basin. Because it is trophically polymorphic, it may perform at several levels in the food web and surely plays an important ecosystem role. The polymorphism is complex, involving multiple characters, yet morphs are difficult to distinguish without close examination of pharyngeal teeth or dissection (Kornfield & Taylor, 1983; LaBounty, 1974). The molariform or molluscivore morph has stout pharyngeal teeth and jaws with hypertrophied associated musculature. The papilliform or detritivore morph has numerous papilliform (fine) pharyngeal teeth embedded in comparatively less-stout jaws, with reduced associated musculature and a relatively longer intestine. Another morph of intermediate pharyngeal morphology, with characters of both molariform and papilliform morphs, was said by Kornfield and Taylor (1983) to exist at frequencies less than 5% of the total population. The pharyngeal tooth morphs are indistinguishable on the basis of overall body shape, but some rare individuals with elongated heads and fusiform bodies have been referred to as the piscivore morph, which may have either molariform or papilliform pharyngeal dentition. Molariform and papilliform morphs with normal body shape interbreed and belong to a single Mendelian population (Kornfield et al., 1982; Kornfield & Koehn, 1975; Sage & Selander, 1975; Trapani, 2003), but the rarer morphs (i.e. intermediate and piscivore) have not yet been studied genetically.
Inter-morph differences in food resource utilization are well supported. Stable isotope ratios, which integrate diet over time, indicate that molariform and papilliform morphs utilize different food sources (Hendrickson et al., in press). Sage and Selander (1975) analyzed 128 guts from 3 pools (lumped together) sampled in summer and winter, and found a strong correlation between pharyngeal morphology and gut content. Molariform individuals ate snails whereas papilliform specimens did not, eating plant matter instead. Fusiform individuals ate fishes, which were very rare in the guts of all normal-bodied specimens. Swanson et al. (2003) monitored individuals in situ and found significant differences among molariform and papilliform morphs in microhabitat use and feeding behaviors. One study, however, indicates broad dietary overlap among morphs, although snail consumption was restricted to molariform individuals. Smith (1982) performed gut analyses of individuals from three pools. His findings were consistent with studies of other cichlid species that often act as generalists despite their apparent morphological trophic specializations (Liem, 1984, 1990). It is likely that dietary segregation may occur only seasonally during periods of low food availability. Smith (1982), sampling during summer of 1979 and not during winter, may have missed the time of year when intraspecific competition could be most intense and morphs would be specializing.
Little is known about the distribution of morphs across habitats in the Cuatro Ciénegas Basin. Sage and Selander (1975) reported little spatial variation in morph distributions; however, our own recent data from the Poza Escobedo system suggest a correlation between snail density and morph proportions, and we find large differences in morph ratios across localities distributed across the entire valley (Hendrickson et al., in press).
Resource partitioning along temporal or spatial axes is considered to be a consistent factor influencing spatial distributions, evolution, and elements of natural histories. The importance of resource partitioning in allowing different species or guilds (sex, morph, size class) within a species to coexist in the same habitat has been demonstrated in many communities in diverse ecosystems for various plant (French & Chambers, 1996; Webb & Peart, 2000) and numerous animal taxa, including many vertebrates (Skúlason & Smith, 1995) as well as invertebrates such as spiders (Aiken & Coyle, 2000), insects (Wilhelm et al., 2001), and lice (Reed et al., 2000). It has been well documented among fishes, both among species (Johannes & Larkin, 1961; Hixon, 1980; Robinson et al., 1993; Robinson & Wilson, 1994; Skúlason & Smith, 1995) and among guilds of single species (Alanärä et al., 2001; McKaye, 1980; Sandheinrich & Hubert, 1984). Especially among cichlids, there is evidence that resource partitioning is an important component maintaining species coexistence. Lake Malawi's diverse cichlid species (called Mbuna) partition resources on a fine scale, with different species using different microhabitats and generally eating different foods (Bootsma et al., 1996: Genner et al., 1999; Reinthal, 1990; Ribbink et al., 1983). However, Reinthal (1990) found that fishes with different trophic structures often eat the same foods and also noted that Mbuna are opportunistic feeders, gorging themselves on fish eggs or plankton when the opportunity arises.
Resource partitioning by food choice, habitat use, or migratory routes has been shown in numerous polymorphic species, including mammals (Chatti et al., 1999), birds, reptiles, amphibians, and fishes (Skúlason & Smith, 1995). Polymorphic fishes demonstrated to express inter-morph resource partitioning include arctic charr (Salvelinus alpinus (Linnaeus 1758)) (Jonsson & Jonsson, 2001), brook charr (S. fontinalis (Mitchill 1814)), brown trout (Salmo trutta Linnaeus 1758), sockeye salmon (Oncorhynchus nerka (Walbaum 1792)), lake whitefish (Coregonus clupeaformis (Mitchill 1818)), Scandinavian whitefish (Coregonus spp.), rainbow smelt (Osmerus mordax (Mitchill 1814)), three-spined stickle-back (Gasterosteus aculeatus Linnaeus 1758), bluegill sunfish (Lepomis macrochirus Rafinesque 1818), pumpkinseed sunfish (Lepomis gibbosus (Linnaeus 1758)), the paradontid (Characiformes) genus Saccodon spp., Cuatro Cienégas cichlids (Herichthys minckleyi), scale-eating cichlids (Perissodus spp.) (Hori, 1993), Midas cichlids (Cichlasoma citrinellum (Günther 1864)) (McKaye, 1980) (see Skúlason & Smith, 1995 for a general description of these polymorphisms) and numerous other examples from 17 families (Robinson & Wilson, 1994).
H. minckleyi is an excellent model for studying habitat partitioning in situ. Individuals of this species are common in almost all pools in Cuatro Ciénegas, and are usually easy to catch, thus facilitating large sample sizes necessary for high statistical power. After short periods of adjustment, their behavior seems little affected by observers only a meter away. They tolerate handling well and survivorship of marked individuals is high. Near total absence of diurnal predators on adults minimizes that possible confounding factor.
Here we use marking and observation of individuals in one springfed pool to test the hypothesis that pharyngeal morphs of H. minckleyi segregate onto specific substrate types and, detecting such segregation, test for temporal variation in it. We also describe different feeding behaviors deployed differentially according to substrate type.
Materials and methods
Poza Mojarral Oeste (102.1229 W, 26.9219 N) was described by Minckley (1969), but has more recently become locally known as Poza Azul. Its clear, blue spring inflow deviates annually no more than one degree from 33°C, although edge habitats may fluctuate more. The pool has two deep pits (eastern and western) with spring inflow in the western pit moving at low velocities across a broad, shallow reach to exit the pool through either a siphon in the eastern pit or a shallow surface canal at its northeastern extremity. There are no surface inflows to the pool, though formerly one existed (Arnold, 1972).
The pool was mapped (Fig. 1) using a theodolite and elevation rod with a rod level. Major features were surveyed and a non-random array of transects with surveyed end points was set up; depths and habitat information were recorded at intervals along each transect. Undercuts were measured and a map was produced and manipulated in the program ArcViewTM (Environmental Systems Research Institute, Inc., Version 3.2). To calculate bottom substrate surface areas, the map was converted to a grid with square cells (50 cm on a side) and average slopes were calculated for each square of the grid. Surface areas were then calculated for each square, and summed to get surface areas for each of the five substrate types existing in the pool (Fig. 2).

In order to further characterize these substrate types, invertebrates were sampled in August 2000 using 0.5-mm mesh dip nets in each of the substrates until no new species were encountered. Samples were picked and sorted in trays in the field and all live individuals counted and identified in the lab. Obtaining comparable sampling effort among physically very different substrates was difficult and for this reason absolute, quantitative analyses were not possible and only relative comparisons (given as a proportion of the within habitat total) among substrate types are reported here.
Since fishes in general can be prone to seasonal food switching (Gerking, 1962; Naiman, 1979; Taylor & Gerking, 1980) and there can be considerable dietary variation among individuals (Bryan & Larkin, 1972), observations were repeated in five consecutive seasons: summer 1999 (Aug), fall 1999 (Oct), winter 2000 (Jan), spring 2000 (Mar), and summer 2000 (Jul). At each sampling, fish were caught with a combination of gears, including cast net, dip net, rod and reel, bare hands (at night), and gill nets. Individuals were anesthetized using clove oil (Prince & Powell, 2000), measured (SL mm), weighed and photographed, and pharyngeal morphology was determined using an otoscope. Individuals were classified to pharyngeal morph using the system of Stephens (2002), as illustrated in Fig. 3: molariform if the lower pharyngeal plate had greater than 50% areal coverage by obviously hypertrophied (molariform) teeth, and papilliform if it had less than 10% coverage by hypertrophied teeth. Intermediate morphs had between 10% and 50% molariform teeth. Individuals were then tagged with uniquely numbered plastic flags (made of surveyors' tape) sewn into the dorsal musculature anterior to the dorsal fin with chromic catgut suture thread designed to dissolve after several days. To prevent observer bias, numbered tags did not identify the individual's pharyngeal morphology. After marking, and recovery in buckets of fresh pool water, marked individuals were released centrally in the pool. Preliminary observations indicated that tagged individuals appeared to behave normally after a 24-hour recovery period, so fish were allowed at least a day, and no more than three days, to recover before observations were made. After recovery, a population estimate was made using the Petersen method by trained observers swimming along a fixed transect (Fig. 1) that passed over each of the five discrete substrate types.
Detailed observations of tagged individuals were noted by observers snorkeling along the fixed transect. Visibility was almost invariant (about 10 m) at the start of each observation period, but flocculent sediments are easily disturbed and visibility invariably dropped during each observation period; however, it was never less than about 5 m. Observers recorded data on any marked specimens seen on or off the transect and were allowed to leave the transect to follow a marked individual's movements. Five observers, composed of combinations of the first author and trained volunteers, made observations simultaneously throughout each sample effort. For each marked fish encountered, observers recorded time of day, total time the specimen spent over each substrate type, the number of picks (soft mouthing at substrate) and stabs (forceful thrust of mouth into substrate) in each substrate type, as well as frequencies of other less common feeding behaviors: "stabs at edible-sized fish" (an attack on a small fish presumably with intent to consume), "vegetation rough up" (oral manipulation of loose vegetation - usually expelled afterwards), "detritus wiggle" (wiggling of body to remove a layer of flocculent sediment followed by a search for exposed food items). Observers attempted to follow and record data on each tagged fish encountered for complete 10 minute periods before moving along the transect to find another tagged fish. In some instances fish were lost from view before 10 minutes elapsed. Observers attempted to minimize disturbances of fishes being observed by themselves or other observers, detouring from the transect if necessary. In each season data were collected over 2 days. On each of these days, observations were made in 3 observation periods (morning, mid-day, and evening) for a total of 6 discrete periods each season. Morning was defined as the two hours immediately after sunrise; evening the two hours immediately before sunset; and mid-day the two hours approximately equitemporal between end of the morning observation and beginning of the evening observation.
Times spent over different substrate types were summed for each individual. An electivity index was created for substrate using the formula:
(A / B) / (X / Y) = Et
where A = the time that an individual spent over a particular substrate, B = the total time the individual was observed (regardless of substrate), X = the total surface area of the substrate type in the pool, and Y = the total pool substrate surface area. Thus Et is the proportion of time spent over substrate divided by the proportional areal representation of that substrate in the pool. A second, similar electivity index was calculated for feeding behaviors using the formula:
(P / Q) / (X / Y) = Ef
where P = the number of feeding acts that the individual made in a particular substrate type, Q = the total number of feeding acts observed regardless of substrate, and X and Y are as defined above. Thus Ef is the proportion of feeding acts observed in a certain substrate divided by the proportional areal representation of that substrate's surface area in the pool. Electivity indices greater than one thus indicate a positive selection for a particular substrate type and lower than one indicate avoidance. Electivity index scores were used as dependent variables in a PROC GENMOD analysis in SAS statistical program (SAS Institute) using morph, substrate, season, time of day, and standard length as independent variables. In addition to analysis using electivity indexes, chisquare tests were performed, using frequencies of individuals displaying each of the observed feeding behaviors, to test the hypothesis that morphs differ in their use of certain feeding behaviors.
Results
Habitat characterization and availability. Poza Mojarral Oeste has 1377.5 m2 of total bottom substrate area, depths up to 4.88 m, and 5 well defined substrate types: (1) Travertine is a biogenic, stratified calcareous formation with a rich, living algal and bacterial fauna in the surface layers (Winsborough, 1990) that covers 12.54% of the total pool area. In some parts of the pool it is covered with a fine detrital dust, but is most often clean and exposed. In some places travertine is associated with steep drops to depths of 0.5 - 4.88 m. Current is substantial in some areas of travertine near spring vents and in the siphon. Travertine in this pool has a highly irregular surface, with numerous crevices and conduits of varying sizes used extensively as cover by H. minckleyi and other fishes. (2) Detritus is composed almost entirely of snail feces and the snail Mexipyrgus churinceanus Taylor 1966. This light-colored flocculent substance, that is easily disturbed to cloud the water, covers 74.75% of the total pool substrate. Snail densities are higher in this substrate than in any other; though they live within this flocculent material they are not usually visible on its surface. (3) Nymphaea (the water lily, Nymphaea ampla (Salisbury) de Candolle) has two growth forms; one with floating leaves and another with submerged, ruffled leaves close to the bottom. The submerged form predominates in Poza Mojarral Oeste, covering 6.6% of the total pool area. (4) Terrestrial vegetation is composed of roots, leaves, and branches of terrestrial vegetation that covers the banks and overhangs much of the pool's edge. Aquatic species such as bullrush (Scirpus sp.) are found here and bladderwort (Utricularia sp.) is often tangled in the submerged roots. This area of overhang offers good cover for fishes and makes up 3.01% of the total pool area. (5) Broken travertine and snail shell is found in the deepest areas, mainly in two patches corresponding to high current velocities that together comprise 3.09% of the total pool area. This substrate consists of bits of travertine that have broken off of the larger overhanging travertine formations, and a collection of dead snail shells (mostly Mexipyrgus, but some Mexithauma as well — for snail descriptions see Hershler 1984).
Substrate types differ in much more than simple physical characteristics (Table 1). All substrates except Nymphaea and travertine differ significantly in the mean depths at which they occur (ANOVA, p<0.0001), ranging from a few centimeters for terrestrial vegetation to as deep as 4.88 m for broken travertine and snail shell. The only invertebrate recorded in detritus is Mexipyrgus churinceanus. In broken travertine and snail shell, M. churinceanus makes up approximately 20% of the invertebrate fauna and Hyalella sp. approximately 70%. Within Nymphaea, M. churinceanus comprises about 84% of the invertebrate fauna and Mexithauma quadripaludium Taylor 1966 14%. Within terrestrial vegetation Hyalella sp. comprises 95% of total invertebrates. Within travertine, Hyalella sp. makes up about 52% of invertebrates, Mexithauma quadripaludium about 36%, and Nymphophilus minckleyi 10%. Other aquatic insects are also found in some substrates, including members of the families Chironomidae (Diptera), Elmidae (Coleoptera), Corduliidae and Coenagrionidae (Odonata), although in low densities. Several other fishes are also found in the pool. Cyprinodon bifasciatus Miller 1968 and Gambusia marshi Minckley & Craddock 1962 are both very common and Cyprinella xanthicara (Minckley & Lytle 1969), Astyanax mexicanus (de Filippi 1853), and Ictalurus lupus (Girard 1858) are present, but uncommon.
Feeding behaviors. Over the five-season course of this study, 230 individuals (60% of the 383 marked) were observed for a total of 208 hours and 11 minutes. Of the observed individuals, 37 were intermediate, 67 were molariform, and 126 were papilliform (see Table 2 for additional summary data). Only 17 of these had piscivore body morphology. Observation sessions (time spent watching an individual) averaged 7.783 minutes.
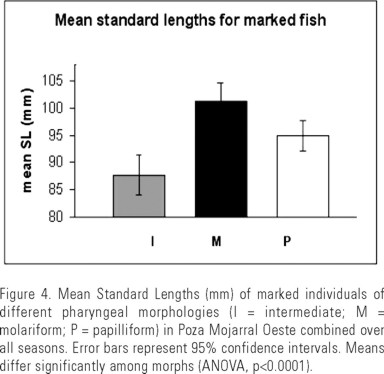
Mean SL of individuals was 96 mm, but their sizes differed significantly among morphs (ANOVA, p<0.0001) with marked molariform individuals (mean SL = 101 mm) on average larger than papilliforms (mean SL = 95 mm), and both were on average larger than intermediate individuals (mean SL = 88 mm, Fig. 4). Active feeding (observation of any type of feeding behavior) was observed for many individuals of each morph and frequencies of individuals displaying feeding behaviors did not differ significantly among morphs (p-values for Chi square tests were all >=0.17, Table 3). None of the feeding behaviors recorded were unique to any pharyngeal morph.
On average, marked individuals picked every 2.23 minutes and stabbed every 4.85 minutes, making these the most common feeding behaviors, while other behaviors were comparatively rare. Individuals tended to be sporadic in their feeding behaviors, at times stabbing and then suddenly shifting to picking. Approximately 48% of observed individuals displayed stab behavior at least once, and 72% displayed pick behavior. Only ten individuals (4% of observed individuals) were observed making stabs at edible-sized fish, but this behavior was observed in unmarked individuals as well and was always directed towards the pupfish, Cyprinodon bifasciatus. Of the marked individuals observed to exhibit this behavior, none had piscivore morphology, but we have frequently observed piscivores preying on pupfish in the course of other studies.
Detritus wiggle appears to be a behavior used mostly by small (<70 mm) individuals, apparently to expose and then feed on snails. This behavior was observed mostly among smaller, unmarked individuals outside of the focus of the study, and was observed in only 6 marked individuals (3% of observed individuals). The other method for catching snails, restricted to larger individuals, involves stabbing the head into the detritus and thus clearing off a layer of detritus to allow visual searching of the area for exposed snails. Vegetation rough up, displayed by 12% of observed individuals, was rare compared to stabs or picks, but was more than twice as frequently observed as was detritus wiggle or stabs at edible-sized fish.
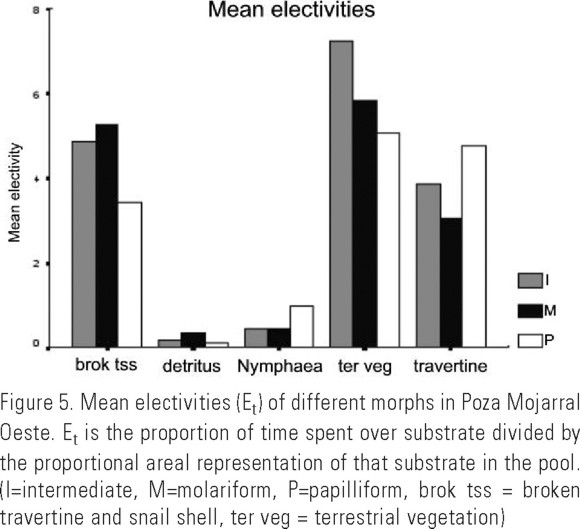
Substrate electivity. Though individuals of all three pharyngeal morphs were observed in each of the five substrates, some substrates were apparently positively selected (Et>1) by all morphs, and others were avoided (Et<1) (Fig. 4). Both Nymphaea and detritus were avoided by all morphs with average Et (for all individuals) of 0.73 and 0.18, respectively. The highest mean Et was recorded for terrestrial vegetation (5.63), followed by broken travertine and snail shell (4.18), and travertine (4.13).
An interaction was found between morph and substrate type using Et as a dependent variable (PROC GENMOD, p < 0.0001). For 4 of the 5 substrates, intermediate individuals had average Et values between those of molariform and papilliform. Et for intermediates for terrestrial vegetation exceeded Et values for other morphs for this substrate, but not significantly. Et values indicate that papilliforms spent more time in travertine than did molariforms (Mann-Whitney U test, Et as dependent variable, p < 0.0001), and molariforms spent more time in detritus than did papilliforms (Mann-Whitney U test, Et as dependent variable, p < 0.0001). Likewise, when Ef was used as a dependent variable an interaction was found between morph and substrate type (PROC GENMOD, p < 0.0001). Ef values indicate that papilliforms fed more in travertine than did molariforms (Mann-Whitney U test, Ef as dependent variable, p < 0.0001), and molariforms fed more in detritus than did papilliforms (Mann-Whitney U test, Ef as dependent variable, p < 0.0001). Ef values also indicate that papilliform morphs elected to feed in Nymphaea more so than did molariform morphs (Mann-Whitney U test, Ef as dependent variable, p = 0.001). In addition to interactions between morph and substrate, an interaction between fish size and substrate type was detected (PROC GENMOD, Et as dependent variable, p=0.0424). Small individuals tended to spend more time in habitats that offered cover (travertine, Nymphaea, and terrestrial vegetation), while medium to large fish tended to spend more time in open habitats (detritus and broken travertine and snail shell).
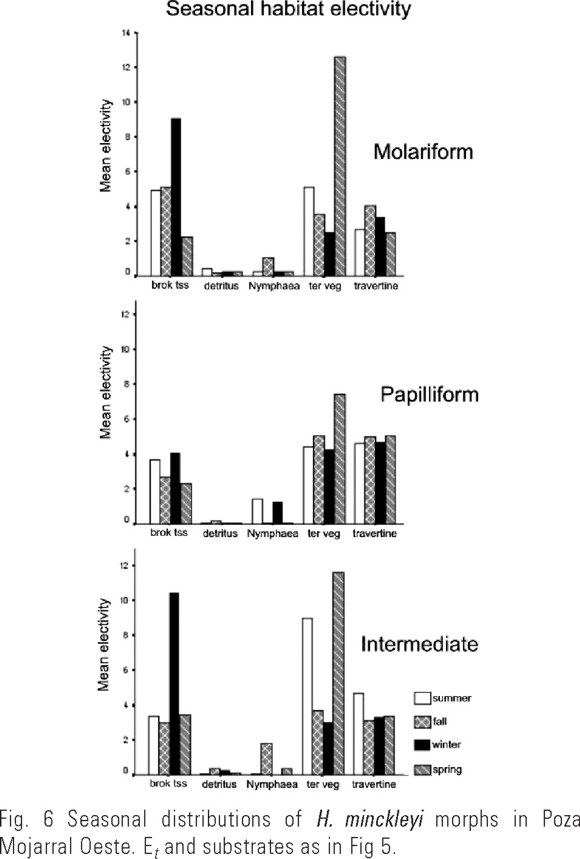
Significant differences among morphs in diel patterns of substrate use were not detected, but seasonal patterns of substrate use did vary significantly among morphs. A significant interaction was found between substrate type, morph (when intermediates were excluded), and season (PROC GENMOD, Et as dependent variable, p = 0.0024, Fig. 5). When intermediates were included in the analysis no interaction was found. Molariforms showed a dramatic shift from broken travertine and snail shell in winter to terrestrial vegetation in spring. Papilliforms tended to display more even use of substrate types throughout the year, but still showed a tendency to use terrestrial vegetation more in the spring than in other seasons. In general papilliforms appeared to move to shallow areas with travertine and terrestrial vegetation in the spring and fall, and to deeper areas of broken travertine and snail shell and Nymphaea in winter and summer. Intermediate individuals showed seasonal substrate type use patterns similar to molariforms, but an increase in use of terrestrial vegetation in the summer as well.
Discussion
Finding that mean standard lengths differed significantly among the three morphs is consistent with other (unpublished) data we have collected for populations across the valley. This pattern suggests that intermediate individuals may be simply juveniles that have yet to fully differentiate into either molariform or papilliform morphs, and is compelling since mate selection in other New World cichlids is known to be influenced by size (Barlow 1998, 2000). If this is also true for H. minckleyi, it seems unlikely that mating would be random among morphs that differ in size.
We found the population to consist of 14.1 % intermediates, while others found intermediates to comprise only about 5% of the population (Kornfield and Taylor (1983). This discrepancy may be due to natural variation over time and/or habitats; however, we cannot rule out differences that may exist in how morphology was classified in the field by different researchers.
Our behavioral data indicate that all H. minckleyi pharyngeal morphs appear to act as feeding generalists, a finding consistent with other studies concluding that other cichlids often act as generalists despite morphological specializations (Liem, 1984, 1990). That intermediate individuals had average Et values between those of molariform and papilliform individuals indicates that intermediates behave as habitat generalists more than do the other pharyngeal morphs.
Despite general similarity in feeding behaviors, diet partitioning among H. minckleyi morphs may occur indirectly via habitat segregation. The relationships found here between pharyngeal morph and substrate type show that morphs do not use substrate types randomly and that habitat electivity differs among morphs. Since the relationship between size and substrate is weak compared to that between morph and substrate, it is probable that morph is more important than size in determining substrate type use.
That molariforms spent more time over, and fed more in detritus than did papilliforms, and that papilliforms spent more time over, and fed more in travertine and also fed more in Nymphaea than did molariforms, is not surprising considering how individuals might utilize their specialized pharyngeal jaws and teeth. Papilliforms may use their brush-like pharyngeal teeth to separate filamentous algae and organic components from coarser and more brittle calcium carbonate in travertine. They may also use their pharyngeal teeth to feed on Nymphaea leaves by separating softer parts of the leaves from less digestible, harder parts.
Molariforms, with stronger musculature and thickened teeth, are specialized for crushing snails and thus might be expected to utilize substrate types with more snails. Though snails are abundant in detritus, our data indicate that molariforms did not prefer this substrate to others (electivities for detritus were less than 1, indicating avoidance). Instead, they avoid it less than do papilliforms, and may use this habitat as a refuge at times when more preferred foods are unavailable in other habitats.
Discovery of habitat partitioning by H. minckleyi morphs is congruent with speciation in progress via a "divergence with gene flow" model (Leim & Kaufman, 1984; Mayr, 1982, 1984). This sympatric speciation model, with resource partitioning by morphs as an initial step, has been touted as being one of the most plausible mechanisms for sympatric speciation in cichlids, and many other animals (Bush, 1994; Diekmann & Doebeli, 1999; Galls & Metz, 1998; Meyer, 1990; Rice & Hostert, 1993; Seehausen, 1996; Skúlason & Smith, 1995; Smith, 1966). According to this model, resource partitioning could lead to assortative mating and eventually evolution along separate trajectories.
Exploitative competition for limited resources is considered to be a strong cause of resource partitioning, however other causal factors are possible. Predator avoidance can result in partitioning of resources and confound competition as a causative factor in resource partitioning. Of these factors, exploitative competition for limited resources probably exerts strong influences on H. minckleyi since diurnal predators are rare and individuals of all morphs come into close contact regularly with only occasional aggression by reproductively active individuals. Swanson et al. (2003) concluded, through a series of in situ cage experiments in which growth of H. minckleyi was increased in mixed morph treatments relative to equal-density single-morph treatments, that the poly-morphism reduces competition on food resources.
Relationships among morph (excluding intermediates), habitat, and season indicate not only that individuals seasonally shift substrate type electivity, but also that molariform and papilliform morphs show different patterns of seasonal substrate use. Deep-water substrates such as broken travertine and snail shell and certain areas of Nymphaea may offer a refuge for papilliform individuals from winter and summer temperature extremes while shallow substrates such as terrestrial vegetation and certain areas of travertine may offer a valuable food source during spring and fall when allochthonous input may be greater to these near-edge habitats. Molariform seasonal habitat use patterns may be influenced by similar factors. Differences between molariform and papilliform seasonal substrate use patterns may serve to reduce inter-morph competition year-round. Although movements are constrained by the overall small size of Poza Mojarral Oeste, more extensive movements and morph-segregation may characterize other habitats in the valley where larger distances often separate substrate types.
Our data show no evidence of daily variation in patterns of substrate type utilization, but we did not include night-time observations. Our observations made outside of this study indicate that movement decreases dramatically at night and, especially on moonless nights, H. minckleyi individuals 'sleep' rather soundly, resting motionless on the bottom and being relatively unaware of potential predators. In this state, skilled quiet divers using indirect light can capture individuals by hand.
Laboratory experiments by Liem and Kaufman (1984) suggest that feeding specialization only occurs during times of low food availability. They fed several food types, including shrimp, flake food, fish, or tubifex worms to H. minckleyi of differing levels of satiation. When starved, molariform fish tended to feed on snails, while papilliform fish tended to feed on other food types. When satiated, both morphs tended to eat foods other than snails. They concluded that the polymorphism is supported by relatively short periods of strong selection during times of low food availability, and so during these times morphs would be expected to segregate into preferred habitats. Our data tend to support this hypothesis because there appears to be a general movement of both morphs to terrestrial vegetation in the spring, a time and substrate where food resources would be high, and increased segregation over other substrates during the rest of the year. More detailed studies and gut analyses may provide more rigorous tests of this hypothesis by providing more direct indicators of food choice, higher resolution of fine scale spatial partitioning, and more subtle differences in feeding behaviors.
There are three possible explanations for the apparent low recovery of marked individuals. First, some marked individuals may not have been observed due simply to chance. Second, some fish died after handling and release to the pool. This is particularly true for winter observations since large temperature differences between air (~5°C) and water (33°C), with frequent strong winds, caused considerable stress during handling of specimens despite our best efforts to minimize it. Finally, knot slippage resulted in some tags shedding as documented by recovery of several tags found loose on the bottom. Many other shed tags likely were quickly buried in sediments or drifted out of the pool. Despite these factors, however, proportions of intermediates, molariforms, and papilliforms observed were roughly comparable to natural morph proportions in the total population of the pool, thus indicating no differential mortality or likelihood of being observed with respect to morphology.
Of the 290 individuals observed, 170 were seen to feed, indicating full recovery to normalcy following marking and little or no modification of behavior related to marking or observation activities. Comments recorded for only 8 marked individuals indicated any abnormal or "scared" behavior. Proportions of time spent in substrates and proportions of feeding acts in those substrates are similar, indicating that fish were probably in those substrates on their own accord, rather than having been scared or otherwise forced into them.
By summing multiple observations of an individual within a season into one record, pseudoreplication (Hurlbert, 1984) within seasons was avoided. Some may argue however, that, at least to a limited extent, some individuals may have been used in more than one season, and thus appear in the data set multiple times (albeit with new identification numbers each time). Efforts were made to avoid this by not using individuals with obvious scarring indicative of marking during previous seasons. Nevertheless, we cannot exclude the possibility that some individuals may have been re-marked in different seasons. Based on our having marked an average of 76 fish per effort, and an estimated population size of 524 (calculated Petersen equation), the probability that we marked a previously marked fish was relatively low, but obviously increased as the study progressed (summer 1999 = 0.00, fall 1999 = 0.101, winter 2000 = 0.232, spring 2000 = 0.361, summer 2000 = 0.458).
Our study is relevant to conservation in this unique protected area by pointing out the importance of protecting substrate type heterogeneity over relatively small scales so all morphs have access to a diversity of substrates. This study and our other observations indicate that morph abundances may be influenced by the level of substrate heterogeneity available to each population. Another concern for managers is the threat created by the introduced jewel cichlid, Hemichromis guttatus, now found in Mojarral Este immediately downstream of Poza Mojarral Oeste (Hendrickson et al., in press). The diet of this species with papilliform pharyngeal morphology has been shown to overlap with the diet of papilliform H. minckleyi (Hendrickson et al., in press). Interactions with it might be hypothesized to shift morph ratios in H. minckleyi toward predominance of the molariform morph and/or alter feeding behaviors of all morphs.
Acknowledgements
Space limitations preclude listing names, but we thank all 47 volunteers who spent over 200 total man-hours in the water observing marked specimens. Doyle Mosier's survey expertise was invaluable for production of Fig. 1 and quantifying areal representation of substrate types. Valerie Virta assisted with the survey. Eric Dinger helped complete the macroinvertebrate survey of Poza Mojarral Oeste. The local Protected Area (SEMARNAT) office provided access to the study site and facilitated all aspects of our research. Lastly, we would like to thank the Nature Conservancy, Earthwatch Institute, and the Durfee Foundation for financial support. Dr. Francisco García de León devoted considerable effort to helping us obtain the permits required to carry out our research (Secretaría de Agricultura, Ganadería, Desarrollo Rural, Pesca y Alimentación, Comisión Nacional de Acuacultura y Pesca, Dirección General de Ordenamiento Pesquero y Acuícola: 180202-613-03-050 [and amendments], 041099-213-03 and 140102-613-03).
References
AIKEN, M. & F.A. COYLE, 2000. Habitat distribution, life history and behavior of Tetragnatha spider species in the Great Smoky Mountains National Park. Journal of Arachnology 28: 97-106. [ Links ]
ALANÄRÄ, A., M.D. BURNS & N.B. METCALFE, 2001. Intraspecific resource partitioning in brown trout, the temporal distribution of foraging is determined by social rank. Journal of Animal Ecology 70: 980-986. [ Links ]
ARNOLD, E.T., 1972. Behavioral Ecology of Two Pupfishes (Cyprinodontidae, Genus Cyprinodon) from Northern Mexico. PhD Thesis, Arizona State University. Tempe. [ Links ]
BARLOW, G.W., 1998, Sexual-selection models for exaggerated traits are useful but constraining. American Zoologist 38: 59-69. [ Links ]
BARLOW G.W., 2000. The Cichlid Fishes: Nature's Grand Experiment in Evolution. Perseus. Cambridge. 335 p. [ Links ]
BOOTSMA, H.A., R.E. HECKY, R.H. HESSLEIN & G.F. TURNER, 1996. Food partitioning among Lake Malawi nearshore fishes as revealed by stable isotope analyses. Ecology 77: 1286-1290. [ Links ]
BRYAN, J.E. & P.A. LARKIN, 1972. Food specialization by individual trout. Journal of the Fisheries Research Board of Canada 29: 1615-1624. [ Links ]
BUSH, G.L., 1994. Sympatric speciation in animals: new wine in old bottles. Trends in Ecology and Evolution 9: 285-288. [ Links ]
CHATTI, N., G. GANEM, K. BENZEKERI, J. CATALAN, J. BRITTON-DAVIDIAN & K. SAÏD, 1999. Microgeographical distribution of two chromosomal races of house mice in Tunisia, pattern and origin of habitat partitioning. Proceedings of the Royal Society of London, Series B 266: 1561-1569. [ Links ]
DIEKMANN, U. & M. DOEBELI, 1999. On the origin of species by sympatric speciation. Nature 400: 354-357. [ Links ]
ENVIRONMENTAL SYSTEMS RESEARCH INSTITUTE, INC., 1992-1998. Arcview GIS Version 3.1. Environmental Systems Research Institute, Inc. Redlands. [ Links ]
FRENCH, T.D. & P.A. CHAMBERS, 1996. Habitat partitioning in riverine macrophyte communities. Freshwater Biology 36: 509-520. [ Links ]
GALLS, F. & J.A.J. METZ, 1998. Why are there so many cichlid species? Trends in Ecology and Evolution 13: 1-2. [ Links ]
GENNER, M.J., G.F. TURNER & S.J. HAWKINS, 1999. Foraging of rocky habitat cichlid fishes in Lake Malawi: coexistence through niche partitioning? Oecologia 121: 283-292. [ Links ]
GERKING, S.D., 1962. Production and food utilization in a population of bluegill sunfish. Ecological Monographs 32: 31-78. [ Links ]
HENDRICKSON, D.A., J.C. MARKS, A.B. MOLINE, E. DINGER & A.E. COHEN, In press. Combining ecological research and conservation: a case study in Cuatro Ciénegas, Mexico. In: Stevens, L. and V.J. Meretsky (Eds.) Every Last Drop: Ecology and Conservation of North American Desert Springs. University of Arizona Press. Tucson. [ Links ]
HERSHLER, R., 1984. The hydrobiid snails (Gastropoda: Rissoacea) of the Cuatro Ciénegas Basin: systematic relationships and ecology of a unique fauna. Journal of the Arizona-Nevada Academy of Science 19: 61-76. [ Links ]
HIXON, M.A., 1980. Competitive interactions between California reef fishes of the genus Embiotoca. Ecology 61: 918-931. [ Links ]
HORI, M., 1993. Frequency-dependent natural selection in the handedness of scale-eating cichlid fish. Science 260: 216-219. [ Links ]
HURLBERT, S.H., 1984. Pseudoreplication and the design of ecological field experiments. Ecological Monographs 52: 187-211. [ Links ]
JOHANNES, R.E. & P.A. LARKIN, 1961. Competition for food between red-side shiners Richardsonius balteatus) and rainbow trout (Salmo gairdneri) in two British Columbia lakes. Journal of the Fisheries Research Board of Canada 18: 203-220. [ Links ]
JONSSON, B. & N. JONSSON, 2001. Polymorphism and speciation in Arctic charr. Journal of Fish Biology 58: 605-638. [ Links ]
KORNFIELD, I.L. & R.K. KOEHN, 1975. Genetic variation and speciation in New World cichlids. Evolution 29: 427-437. [ Links ]
KORNFIELD, I.L. & J.N. TAYLOR, 1983. A new species of polymorphic fish, Cichlasoma minckleyi, from Cuatro Ciénegas, Mexico (Teleostei: Cichlidae). Proceedings of the Biological Society of Washington 96: 253-269. [ Links ]
KORNFIELD, I.L., D.C. SMITH, P.S. GAGNON & J.N. TAYLOR, 1982. The Cichlid fish of Cuatro Ciénegas, Mexico: evidence of conspecificity among distinct trophic morphs. Evolution 36: 658-664. [ Links ]
LABOUNTY, J.F., 1974. Materials for the revision of cichlids from northern Mexico and southern Texas, USA (Perciformes: Cichlidae). PhD thesis, Arizona State University. Tempe. [ Links ]
LIEM, K.F., 1984. Functional versatility, speciation, and niche overlap: are fishes different? In: Meyers DG, Strickler JR (Eds.) Trophic interactions within aquatic ecosystems. Westview Press. Boulder. [ Links ]
LIEM, K.F., 1990. Aquatic versus terrestrial feeding modes: possible impacts on the trophic ecology of vertebrates. American Zoologist 30: 209-221. [ Links ]
LIEM, K.F. & L.S. KAUFMAN, 1984. Intraspecific macroevolution: functional biology of the polymorphic cichlid species Cichlasoma minckleyi. In Echelle A & I. Kornfield (Eds.) Evolution of fish species flocks. University of Maine at Orono Press. Orono. [ Links ]
Mayr, E., 1982. Speciation and Macroevolution. Evolution 36: 1119-1132. [ Links ]
MAYR, E., 1984. Evolution of fish species flocks: a commentary. In Echelle A. & I. Kornfield (Eds.) Evolution of fish species flocks. University of Maine at Orono Press. Orono. [ Links ]
MCKAYE, K.R., 1980. Seasonality in habitat selection by the gold color morph of Cichlasoma citrinellum and its relevance to sympatric speciation in the family Cichlidae. Environmental Biology of Fishes 5: 75-78. [ Links ]
MEYER, A., 1990. Trophic polymorphism in cichlid fish: Do they represent intermediate steps during sympatric speciation and explain their rapid adaptive radiation? In: Schröder, J.H. (Ed.) Trends in Ichthyology. Paul Perey. Berlin. [ Links ]
MINCKLEY, W.L., 1969. Environments of the bolsón of Cuatro Ciénegas, Coahuila, Mexico. Texas Western Press, University of Texas. El Paso. 63 p. [ Links ]
NAIMAN, R.J., 1979. Preliminary food studies of Cyprinodon macularius and Cyprinodon nevadensis (Cyprinodontidae). The Southwestern Naturalist 24: 527-541. [ Links ]
PRINCE, A. & C. POWELL, 2000. Clove oil as an anesthetic for invasive field procedures on adult rainbow trout. North American Journal of Fisheries Management 20: 1029-1032. [ Links ]
REED, D.L., M.S. HAFNER, S.K. ALLEN & M.B.SMITH, 2000. Spatial partitioning of host habitat by chewing lice of the genera Geomydoecus and Thomomydoecus (Phthiraptera: Trichodecridae). Journal of Parasitology 86: 951-955. [ Links ]
REINTHAL, P.R., 1990. The feeding habits of a group of herbivorous rock-dwelling cichlid fishes (Cichlidae: Perciformes) from Lake Malawi, Africa. Environmental Biology of Fishes 27: 215-233. [ Links ]
RICE, W.R. & E.E. HOSTERT, 1993. Laboratory experiments in speciation: what have we learned in 40 years? Evolution 47: 1637-1653. [ Links ]
RIBBINK, A.J., B.A. MARSH, A.C. MARSH, A.C. RIBBINK & B.J. SHARP, 1983. A preliminary survey of the cichlid fishes of rocky habitats in Lake Malawi. South African Journal of Zoology 18: 149-310. [ Links ]
ROBINSON, B.W . & D.S. WILSON, 1994. Character release and displacement in fishes: a neglected literature. The American Naturalist 144: 598-627. [ Links ]
ROBINSON, B.W., D.S. WILSON, A.S. MARGOSIAN & P.T. LOTITO, 1993. Ecological and morphological differentiation of pumpkinseed sunfish in lakes without bluegill sunfish. Evolutionary Ecology 7: 451-464. [ Links ]
SAGE, RD, & R.K. SELANDER, 1975. Trophic radiation through polymorphism in cichlid fishes. Proceedings of the National Academy of Science 72: 4669-4673. [ Links ]
SANDHEINRICH, M.B. & W.A. HUBERT, 1984. Intraspecific resource partitioning by yellow perch Perca flavescens in a stratified lake. Canadian Journal of Fisheries and Aquatic Sciences 41: 1745-1752. [ Links ]
SAS INSTITUTE, 2000-2001. SAS Software Version 8.1 TS1 MO for Windows. [ Links ]
SECRETARÍA DE DESARROLLO SOCIAL, 1994. Decreto por el que se declara como área protegida, con el carácter de protección de flora y fauna, la región conocida como Cuatrociénegas, municipio de Cuatro Ciénegas, Coahuila. Diario Oficial De La Federación, México CDXCIV.5: 5-11. [ Links ]
SEEHAUSEN, O., 1996. Lake Victoria Rock Cichlids. Verduyn Cichlids. Zevenhuisen. [ Links ]
SKÚLASON, S. & T.B. SMITH, 1995. Resource polymorphisms in vertebrates. Trends in Ecology and Evolution 10: 366-370. [ Links ]
SMITH, D.C., 1982. Trophic ecology of the cichlid morphs of Cuatro Ciénegas, Mexico. M.S. Thesis, University of Maine. Orono. [ Links ]
SMITH, M.S., 1966. Sympatric speciation. The American Naturalist 100: 637-650. [ Links ]
STEPHENS, M.J., 2002. Pharyngeal morphology and spatial and temporal variation in trophic composition in the polymorphic cichlid, Herichthys minckleyi (Teleostei: Cichlidae) from the Cuatro Cienegas Basin, Mexico. M.S. Thesis, Southwest Texas State University. San Marcos. 61 p. [ Links ]
SWANSON, B.O., A.C. GIBB, J.C. MARKS & D.A. HENDRICKSON, 2003. Trophic polymorphism and behavioral differences decrease intraspecific competition in a cichlid, Herichthys minckleyi. Ecology 84: 1441-1446. [ Links ]
TAYLOR, W.W. & S.D. GERKING, 1980. Population dynamics of Daphnia pulex and utilization by the rainbow trout (Salmo gairdneri). Hydrobiologia 72: 277-287. [ Links ]
TRAPANI, J., 2003. Geometric morphometric analysis of body-form variability in Cichlasoma minckleyi, the Cuatro Cienegas cichlid. Environmental Biology of Fishes 68: 357-369. [ Links ]
WEBB, C.O. & D.R. PEART, 2000. Habitat associations of trees and seedlings in a Bornean rain forest. Journal of Ecology 88: 464-478. [ Links ]
WILHELM, S.I., D.J. LARSON & A.E. STOREY, 2001. Habitat preference of two burying beetles (Coleoptera: Silphidae: Nicrophorus) living among seabirds. Northeastern Naturalist 8: 435-442. [ Links ]
WINSBOROUGH, B.M., 1990. Some Ecological Aspects of Modern Freshwater Stromatolites in Lakes and Streams of the Cuatro Ciénegas Basin, Coahuila, México. Ph.D. Thesis, University of Texas. Austin. [ Links ]














