INTRODUCTION
Heavy metals are broadly defined, high-density chemical elements that occur naturally in the environment in low concentration levels (Calamari and Naeve 1994). They are relevant for coastal management and public health because of their increased levels in the environment due to human activities, and represent highly harmful pollutants (Han et al. 2000).
Heavy metals are released from land-based anthropogenic and natural sources; discharged through river runoffs, drainage and atmospheric deposition; and finally collected into receiving systems, such as estuaries and mangroves (Amirah et al. 2013). Coastal mangroves provide regulating ecosystem services, like waste decomposition, detoxification and retention because their sediments actas a chelating matrix for metals (Amaro-Pinheiro et al. 2012).
Thus, mangroves reduce the mobilization of metals to plant tissues and dependent trophic webs. However, mangrove deforestation is reducing this capacity, potentially allowing previously trapped metals to disperse and enter adjacent ecosystems (Sandilyan and Kathiresan 2014). For this reason, the accumulation of heavy metals in mangroves has become a worldwide threat (NOAA 1990, Macfarlane et al. 2007, Lewis et al. 2011).
Some aquatic organisms living in mangrove ecosystems tend to accumulate heavy metals that may relate to several public health effects. Animals that live in close association with sediments, in which they bury and feed, seem to be more exposed to these contaminants than other organisms (Storelli 2008).
Crabs have been extensively used for the study of heavy metals bioaccumulation because they feed mainly on litter, promote organic matter incorporation to sediments, display slow growth rates and long life cycles, and are abundant and easy to collect (Amaro-Pinheiro et al. 2012). These popular seafood items represent a pathway for heavy metal transfer to human populations (Chien et al. 2002, Martí-Cid et al. 2007, Chen et al. 2008, Dórea 2008).
Documented toxicity mechanisms of heavy metals in humans are related to enzymes of cellular respiration and central nervous system control of respiratory movements, among others (Marques et al. 2010). Effects include reduced central nervous system functioning, lower energy levels, damage to blood composition, lungs, kidneys, liver, and other organs (Amirah et al. 2013).
Long-term exposure to heavy metals may result in Alzheimer’s and Parkinson’s diseases, muscular dystrophy, multiple sclerosis, and several types of cancer (Han et al. 2000, Amirah et al. 2013). Heavy metals are considered among the most concerning causes of major foodborne diseases (WHO 2015).
Fish consumption has increased worldwide in the last five decades, as it represents an excellent source of nutrients (FAO 2016). In Ecuador, annual fish consumption is 5-10 kg/person, the average for developing countries, and seafood is mainly provided by the artisanal sector (FAO 2003, 2016).
The Ecuadorian government has recognized the negative impact of contamination on fisheries, and among its national policies is the prevention, control and mitigation of contamination in maritime areas and coastal zones (STM 2015). However, Ecuador does not conduct continuous monitoring of water quality (CPPS 2012, MAE 2014, Nolivos et al. 2015). In this review, the available information of heavy metal contamination in the main estuary of the country is synthesized, focusing on artisanal fisheries products.
Study area
The Gulf of Guayaquil (GG) is the largest estuarine ecosystem on the Pacific coast of South America. Its surface area is estimated to cover 13 711 km2 (Fig. 1) and its depth varies between 20 and 180 m. It receives water from 24 out of the 79 drainage basins in the country, resulting in an annual average flow of 1654.5 m3/s. The catchment covers 53 299 km2 and includes 12 of the 24 provinces in the country (Montaño-Armijos and Sanfeliu-Montolío 2008).
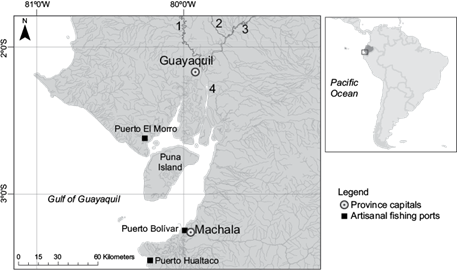
Fig. 1 Study area. Location of the Gulf of Guayaquil in South America, and main sites mentioned in the text. Rivers: (1) Daule, (2) Vinces, (3) Babahoyo, (4) Guayas
Economic activities across the GG’s watershed include agriculture, cattle raising, artisanal fishing, aquaculture, tourism, mining, manufacturing, navigation and urban development. Crops like rice, sugar cane, corn, banana, oil palm, cacao and coffee represent the main agricultural products of the region (Montaño-Armijos and Sanfeliu-Montolío 2008).
The GG’s influence area includes the city of Guayaquil, the largest in Ecuador, with more than 2.3 million inhabitants (INEC 2010). It is the main port city and a hub for economic, commercial and industrial activities.
Guayaquil is located on the banks of the Guayas river (Fig. 1), which is formed by the union of Daule, Vinces and Babahoyo rivers (Montaño-Armijos and Sanfeliu-Montolío 2008). The landscape around the Guayas river system is dominated by banana, rice, sugar cane, cacao, corn, passion fruit and soy fields, while shrimp ponds are abundant near the river mouth.
The GG accounts for 42 % of the Ecuadorian continental platform area and holds the largest fisheries production in the country (Montaño-Armijos and Sanfeliu-Montolío 2008, CPPS 2012). Official landings data, however, is only available for the red mangrove crab (Ucides occidentalis) and ark (Anadara spp., a bivalve), which are highly demanded seafood products. In 2011, more than 14 million crabs and 12 million black arks were landed in the GG (INP 2013).
Objectives
The GG is under strong pressure from human activities that have polluted its aquatic environment (CPPS 2012). Yet, this situation is not well represented in international scientific literature.
Therefore, this systematic literature review intends to provide an overview of the current situation and identify major information gaps. It focuses on local studies about heavy metals concentrations in water, sediments and major artisanal seafood products of the region.
The specific objectives of this contribution are to: (a) evaluate data quality to describe the robustness of available evidence, (b) assess the level of potential risk of heavy metal contamination, and (c) explore spatial patterns of heavy metal contamination across the region.
MATERIALS AND METHODS
Data collection
A search for empirical studies of heavy metal concentrations in mangrove ecosystems of the GG was done. Public online catalogues, such as universities’ online repositories, research centers’ websites, and scientific (Scopus) and academic (Google Scholar) databases were consulted. Search keywords were: “contamination”, “heavy metals”, and “Gulf of Guayaquil”, in English and Spanish.
Search results were not filtered by year. Information about heavy metal accumulation in plants was not included, as it was out of the scope of this study. In cases where only an abstract was available online, libraries were visited in person to access full-text documents. If a study was available as a thesis and peer-reviewed article, both documents were considered in the analysis.
Information retrieved from these documents includes: sampling location coordinates, sampling design description, lists of test materials and metals, analytical techniques, supplies and reagents used for preservation of samples and extraction of metals, analytical quality assurance procedures (AQA) (Taverniers et al. 2004), reported indicators of data quality and heavy-metal concentration values.
Analytical quality assurance assessment
Information regarding AQA procedures and data quality indicators were used to assess the robustness of the available evidence for water, sediments and seafood metal contamination in the GG. Reported AQA practices were classified as being at the intra-, inter- or external-laboratory levels of quality control.
Use of certified reference material was interpreted as an accuracy indicator. Coefficient of variation (CV) was calculated for all studies as a common indicator of precision (Taverniers et al. 2004).
Qualitative diagnosis of contamination risk
A qualitative diagnosis of heavy metal contamination levels in the GG was elaborated in three steps using quantitative indicators according to the following steps:
The design of a classification system for contamination levels consisted in adapting the “traffic-light” method (Baigún 2013), developed for artisanal fisheries management in the Paraná river basin in Argentina (Table I). Measured concentrations (Cm) were compared with their correspondent permitted levels (Csl) defined in international regulations, following EC (2016) for seafood, CCME (2014) for water and sediments, and USEPA (1980) for Pb in water. Qualitative indicators resembled the colors of a traffic-light, green for ideal conditions (Cm < Csl × 10-1), yellow for alert situations that require precautionary measures (Csl × 10-1 ≤ Cm ≤ Csl × 10), or red for dangerous situations that must be reverted (Cm > Csl × 10).
TABLE I LEVELS OF POTENTIAL RISK BY HEAVY METAL CONTAMINATION
| Level | Level description | Quantitative definition | Reference color |
| Ideal conditions | Ideal/desired conditions associated to practices that promote ecological integrity conservation and long-term fisheries sustainability | Heavy-metal concentration below safety level’s order of magnitude. Cm < (Csl e × 10-1) | Green |
| Alert | Situations or practices that might threaten ecosystem functioning and fisheries status, and require precautionary measures | Heavy-metal concentration in the same order of magnitude as safety level. Cm ≤ (Csl × 10) and Cm ≥ (Csl × 10-1) | Yellow |
| Danger | Dangerous or inviable situations or scenarios that must be reverted for they seriously compromise riverine ecosystem functioning, resources and fishing communities | Heavy-metal concentration above safety level’s order of magnitude. Cm > (Csl × 10) | Red |
The framework is an adaptation of the “traffic-light” system described in Baigún (2013). Cm: measured concentration, Csl; safety level concentration
The classification system (Table I) was applied to available data (i.e., reported average concentration per sampling site, per study) for the most frequently analyzed elements, and obtained a matrix of samples versus test elements, containing traffic-light colors in each cell.
The geographic coordinates of sampling sites were added to the database to map results using the QGIS 2.8 application (QGIS Development Team 2015). Each element was represented in a separate point vector layer, with symbol color defined by cells’ values.
Spatial analysis
Boxplots were elaborated to show the variability of heavy metal concentrations in three sectors of the GG for available water, sediments and seafood data. Sectors were delimited around clusters of sampling sites.
All data available for each sector were included in the graph. (Geo-) statistical analyses were not conducted with data because of the large variation in sampling designs (spatio-temporal scales, focus species), which resulted in several confounding factors without the sufficient sample size to account for them. Therefore, results were interpreted qualitatively.
RESULTS
Twenty-four published studies performed between 2010 and 2017 were found, of which only one (Fernández-Cadena et al. 2014) was indexed in the scientific database Scopus (Table II). No studies conducted prior to 2010 were found.
TABLE II DETAILS OF THE METHODS USED TO MEASURE HEAVY METAL CONCENTRATIONS IN ORGANISMS ACROSS THE GULF OF GUAYAQUIL
| Species | Test elements | Analytical method* | Body part analyzed (textual translations) | Preservation prior to extraction | Extraction reagents | References |
| Crustacea | ||||||
| Callinectes arcuatus | Cr, Ni, Cd, Hg, Pb | FAAS, GH-AAS | Soft tissues and hepatopancreas | In cool box (4°C) | HNO3 | Franco-Solórzano (2015) |
| Ucides occidentalis | Cd, Pb | AAS | Exoskeleton | Room temperature | HNO3 | Feys-Espinoza (2013) |
| Ucides occidentalis | Cd, Hg, Pb, | FAAS, GH-AAS | Hepatopancreas tissue and claw muscle | In cool box | HNO3 | Chuquimarca-Montesdeoca (2015) |
| Ucides occidentalis | Cd, Pb | AAS | Soft tissues and digestive gland | Live to the lab | HNO3 | Siavichay-Lalangui (2013) |
| Ucides occidentalis | Cr, As, Hg, Pb | AAS | Soft tissues | Not specified | Hg: HNO3, H2SO4; Cr, As, Pb: HCl, HNO3 | Ayala-Armijos et al. (2015) |
| Mollusca | ||||||
| Anadara tuberculosa | Cd, Hg, Pb | AAS | Soft tissues | In cool box | HNO3 | Ordóñez-Lucin (2015) |
| Anadara tuberculosa, Anadara similis | Cr, Mn, Co, Ni, Cu, Zn As, Cd, Hg, Pb | FAAS, CVAA | Soft tissues | In cool box | HNO3, HclO4 | Tobar-Ordóñez (2013) |
| Anadara tuberculosa, Mytella guyanensis, Leukoma asperrima | Cr, Cu, As, Cd, Hg, Pb | AAS, Polaro- graphy | Soft tissues | Freezer (-20°C) | HNO3 | Carrasco-Peña and Webster-Coello (2016) |
| Crassostrea columbiensis | Cr, Ni, Cd, Pb | AAS | Soft tissues | In cool box (4°C) | HNO3 | Jiménez-Verdesoto (2012) |
| Crassostrea columbiensis | Cd, Hg, Pb, | FAAS, GH-AAS | Soft tissues | In cool box | HNO3 | Castro-Infante (2015) |
| Mytella guyanensis | Ni, Cd, Pb | FAAS | Soft tissues | In cool box | HNO3 | Banguera-Rodríguez (2013) |
| Mytella strigata | Cr, Cd, Pb | FAAS | Soft tissues | In cool box | HNO3 | Kuffó-García (2013) |
| Mytella strigata, Ostrea columbiensis | Cd, Pb | FAAS | Soft tissues | In cool box | HNO3 | Mero-Valarezo (2010) |
| Pomacea canaliculata | Cd | AAS | Soft tissues | In cool box | HNO3 | Ramírez-Prado et al. (2016), Ramírez-Prado (2016) |
| Teleostei | ||||||
| Diapterus peruvians, Sardinops sagax | Mn, Ni, Cu, Zn Cd, Hg, Pb | FAAS, CVAA | Soft tissues | Freezer | HNO3, HclO4 | Ortega-Pereira (2015) |
*Methods used to quantify the concentration of test elements present in test materials: AAS: atomic absorption spectrometry, CVAA: cold vapor, AAS, FAAS: flame AAS, GH-AAS; hydride generation AAS
Three types of materials were tested for contaminants: water, sediments and seafood. The most common test elements were Pb (n = 117), Cd (n = 114) and Hg (n = 58), so our analyses focused on them. Seafood species represented in these studies comprised eight mollusks, two crustaceans and two fish species (Tables II and V).
Other test elements analyzed less frequently in studies included chromium (Cr), zinc (Zn), arsenic (As), copper (Cu), nickel (Ni), cobalt (Co) and manganese (Mn), among others (Appendix II).
Summary of analytical methods
All studies found in this review used spectrometry methods to measure the concentrations of heavy metals in samples (Tables II-IV). However, there were some differences among the methods used prior to the analysis. For instance, most studies focusing in organisms analyzed what authors generally called “soft tissues”, and only one study (Feys-Espinoza 2013) was carried out using hard parts (crab exoskeleton).
The extraction of metals from organisms was done using HNO3 in all cases, but some authors added other acids in this process: Ortega-Pereira (2015) also used HClO4, while Ayala-Armijos et al. (2015) added H2SO4 for Hg measurements, and HCl for Cr, As and Pb.
Water samples were reportedly fixed with HNO3 in the field and transported in low temperature conditions to the lab in all cases (Table III). Water was filtered using papers of varied pore sizes, ranging from 0.2 to 8 µm, while this information was missing in half the studies.
TABLE III DETAILS OF THE METHODS USED TO MEASURE HEAVY METAL CONCENTRATIONS IN WATER SAMPLES ACROSS THE GULF OF GUAYAQUIL
| Test elements | Analytical method* | Pore-size of water filtration paper | Preservation prior to extraction | Extraction | References |
| As, Cd, Hg, Pb | AAS | Not specified | In cool box (< 10 °C); HNO3 | HCl | Alcívar-Tenorio and Mosquera-Armijo (2011) |
| Cr, Ni, Cu, Zn, Cd, Hg, Pb | AAS | 0.45µm | In cool box; HNO3 | Not specified | Banguera-Rodríguez (2013) |
| Cd | AAS | 8 µm (Whatman No. 40) | 4°C; HNO3 | Not specified | Carpio-Rivera (2016) |
| Cd, Hg, Pb, | AAS | Not specified | In cool box (< 10 °C); HNO3 | Not specified | Del Pezo-Quijije and Ruíz-Chiriguaya (2016) |
| Cd, Pb | FAAS | Not specified | In cool box (< 10 °C) | PDAAAC** | Huayamave (2013) |
| Cd, Pb | AAS | Not specified | HNO3 | Not specified | Jiménez-Verdesoto (2012) |
| Cr, Cd, Pb | AAS | 0.2 µm | In cool box (< 10 °C); HNO3 | PDAAAC** | Kuffó-García (2013) |
| Cr, Ni, Cd, Pb | AAS | 0.47 µm | In cool box; HNO3 | PDAAAC** | Mero-Valarezo (2010) |
| Ni, Cd, Pb | FAAS | Not specified | In cool box; HNO3 | PDAAAC** | Proaño-Alvarado (2016) |
| Cd, Cr, Pb | FAAS | 8 µm (Whatman No. 40) | HNO3 | PDAAAC** | Ramírez-Prado et al. (2016); Ramírez-Prado (2016) |
| Cr, Cd, Pb | FAAS | Not specified | HNO3 | Not specified | Rivera-Pizarro (2016) |
*Methods used to quantify the concentration of test elements present in test materials: AAS: atomic absorption spectrometry, FAAS: flame atomic absorption spectrometry
**Pyrrolidine dithiocarbonate ammonium, ammonium acetate, and chloroform
The most common method to extract the metals from the sediments used pyrrolidine dithiocarbonate ammonium, ammonium acetate and chloroform (PDAAAC), but this information was often not explicit. The authors simply cited the “standard methods” guidelines (APHA 2005) in the methods’ section, leaving uncertainty about the exact procedures conducted in each case.
Finally, sediment samples were sieved to collect particles below 71 or 63 µm (Table IV). The extraction was done using HNO3 in all but two cases: Fernández-Cadena et al. (2014) used HNO3 and H2O2, and Rivera-Pizarro (2016) used HCl.
TABLE IV DETAILS OF THE METHODS USED TO MEASURE HEAVY METAL CONCENTRATIONS IN SEDIMENT SAMPLES ACROSS THE GULF OF GUAYAQUIL
| Test elements | Analytical method | Grain fraction analyzed | Preservation prior to extraction | Extraction | References |
| Cr, Cd, Pb | AAS | < 71 µm | In cool box (<10°C) | HNO3 | Alcívar-Tenorio and Mosquera-Armijo (2011) |
| Ni, Cd, Pb | FAAS | < 71 µm | In cool box | “ “ | Banguera-Rodríguez (2013) |
| Cd, Pb | AAS | < 71 µm | In cool box | “ “ | Carpio-Rivera (2016) |
| Li, Be, Al, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, As, Se, Mo, Ag, Cd, Sn, Pb | ICP-MS | Not specified | Not specified | HNO3, H2O2 | Fernández-Cadena et al. (2014) |
| Cr, Ni, Cd, Hg, Pb | FAAS, GH-AAS | Not specified | In cool box | HNO3 | Franco-Solórzano (2015) |
| Cr, Ni, Cd, Pb | AAS | < 71 µm | In cool box | “ “ | Jiménez-Verdesoto (2012) |
| Cd, Pb | AAS | < 71 µm | In cool box | “ “ | Jiménez-Verdesoto (2017)** |
| Cd, Pb | FAAS | < 63 µm | Not specified | “ “ | Mero-Valarezo (2010) |
| Cd, Cr, Pb | FAAS | < 63 µm | Not specified | “ “ | Proaño-Alvarado (2016) |
| Cd | AAS | < 71 µm | In cool box | “ “ | Ramírez-Prado et al. (2016), Ramírez-Prado (2016) |
| As, Cd, Hg, Pb | AAS | Not specified | Not specified | HCl | Rivera-Pizarro (2016) |
| Cd, Pb | AAS | < 71 µm | In cool box | HNO3 | Siavichay-Lalangui (2013) |
*Methods used to quantify the concentration of test elements present in test materials: AAS: atomic absorption spectrometry, FAAS: flame AAS, GH-AAS: hydride generation AAS, ICP-MS: inductively coupled plasma mass spectropmetry
**This study also analized organisms (fish) but those data were not included in the present review because, according to the author, the specimens were bought on a nearby fish market, and thus lacked evidence of the geographic location where they were collected. Also, the species was not determined
Summary of analytical quality assurance procedures
Implicit references to AQA procedures in the methods section were found in most (88 %) studies (Appendix I). References to methods used by other authors, and at least one intra-laboratory procedure were reported in 71 % of the studies found. Certified reference materials (mainly sediment) were reportedly used in 38 %.
Inter-laboratory AQA procedures or participatory proficiency testing schemes were not mentioned in any of the studies. The laboratories from Universidad de Guayaquil and Universidad Técnica de Machala together hosted 88 % of published studies, and both hold the certification ISO/IEC 17025 - general requirements for the competence of testing and calibration laboratories.
Of the six (25 %) studies mentioning the ISO certification to support their data reliability, only two reported the use of reference materials, and none reported the resulting percentage of recovery.
Rapid risk assessment
Risk levels were assessed for Cd, Hg and Pb. Reported concentrations equal to or higher than the permitted levels were found in 53 % of samples for Cd, 27 % for Hg, and 33 % for Pb. Most samples were classified in the alert level (Fig. 2). Water analysis showed high percentages (> 70 %) of samples indicating dangerous concentrations of Cd and Hg.

Fig. 2 Percentages of samples with distinct levels of heavy metal contamination across the Gulf of Guayaquil. Original data were published between 2010 and 2016. Risk levels are relative to safe levels defined in international regulations
In sediments, Cd and Pb ranged from ideal to alert conditions, while for Hg from alert to dangerous. In seafood, the three elements ranged from ideal to dangerous.
Spatial distribution of contaminants
Only one study (Proaño-Alvarado 2016) explicitly used a spatial analysis approach. It was conducted inside the Manglares Churute Reserve (a national marine protected area within the GG) and did not find significant differences in Cd, Cr or Pb concentrations among the four sampling stations established.
Most studies were conducted in the inner estuary, north of Puna island (Fig. 1). Three sectors were studied in more detail: Salado (n = 49), Churute (n = 32) and Jambeli (n = 28) (Figs. 3-5).
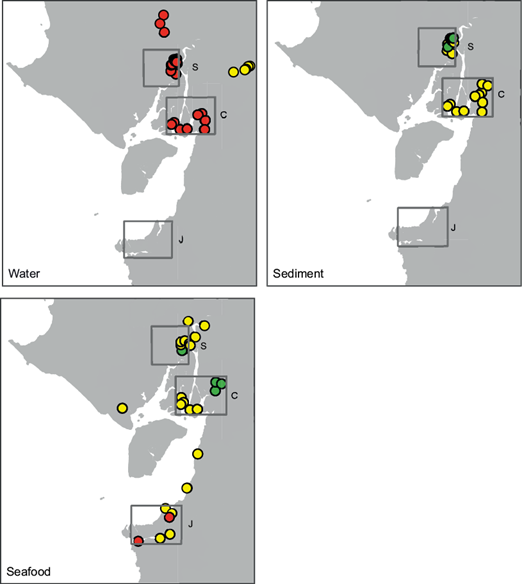
Fig. 3 Cadmium contamination risk levels inferred from in local studies across the Gulf of Guayaquil. Dot colors represent the risk level as defined in the Methods’ section: green is “ideal conditions”; yellow is “alert”; and red is “danger”. Rectangles represent sectors referred to in the text: (S) Salado, (C) Churute, (J) Jambeli
Salado includes an estuarine branch that runs through the city of Guayaquil and the Manglares Estero Salado Reserve, south of the city. Churute (as defined in this study) includes Manglares Churute Reserve and nearby islands from the Guayas river estuary. The Jambeli archipelago is located south of Puna island.
Cadmium
Cd data was mainly available for Churute and Salado (Figs. 3 and 6a-c). In water, sediments, and organisms, Cd concentrations reached higher values in Churute than in Salado.
In Jambeli (only seafood data was available), Cd levels were similar to those found in Churute, and included the only Cd outlier: the oyster Crassostrea columbiensis with ten times the safe level of Cd for mollusks meat (location: Puerto Hualtaco) (Castro-Infante 2015).
Mercury
Hg data was the scarcest, and was not available for Churute (Figs. 4 and 6d-f). Concentration values covered large ranges, as reflected in the vertical-axis’ breaks in figure 6f in water, data was only available for Salado and all values were above the permitted level. In sediments, all values from Jambeli were above the permitted level, while all values from Salado were below.
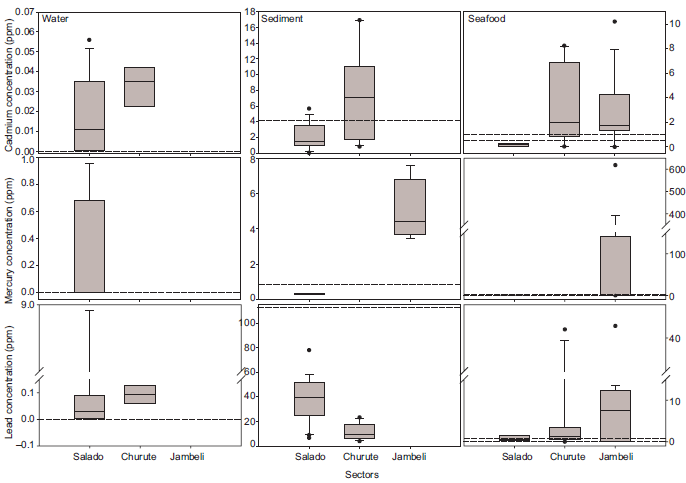
Fig. 6 Box plots showing the variability of concentrations of heavy metals found in water, sediment and seafood and sectors of Gulf of Guayaquil. Box plots represent median, 10th, 25th, 75th and 90th percentiles. Dashed lines represent the permitted levels as defined in international regulations. Boxplots C and I contain two dashed lines corresponding to the highest and lowest permitted levels for the species involved. Black dots represent outliers
In seafood, Jambeli had the highest values, more than half of which passed the permitted level. Also in Jambeli, two studies (Tobar-Ordóñez 2013, Ayala-Armijos et al. 2015) found Hg concentrations > 200 times higher than the permitted level. The outlier in the seafood graph belongs to the black ark Anadara similis, with 618 ppm (location: Puerto Bolívar, Tobar-Ordóñez 2013). Organisms from Salado were represented by a single species (Callinectes arcuatus) and a single study (Franco-Solórzano 2015).
Lead
Pb data was mainly available for Churute and Salado (Figs. 5 and 6g-i). Again, vertical-axis breaks were needed to represent the large variability across the data. In water, most values were above the permitted level.
Two studies of water in the exact same sampling sites inside Salado found very different Pb concentrations: Kuffó-García (2013) found values three times the permitted level during the rainy season, while del Pezo-Quijije and Ruiz-Chiriguaya (2016) found values 200-1000 times the permitted level during the dry season.
Sediments from Salado showed higher concentrations and larger variability than from Churute, but both were below the permitted level.
In seafood, Jambeli showed the highest concentrations of Pb. Extreme concentrations were found in the crab Ucides occidentalis, in Churute and Jambeli, supported by only one study (Feys-Espinoza 2013). This study included the three outliers for seafood (> 30 ppm, Fig. 6i), plus one value from a different sector (location: Puerto El Morro, Fig. 1, 44.19 ppm).
Other studies of Pb on this species (Siavichay-Lalangui 2013, Chuquimarca-Montesdeoca 2015) did not find such extreme values, but they worked with soft tissues (i.e., clam meat and hepatopancreas), while Feys-Espinoza (2013) analyzed exoskeletons.
DISCUSSION
Heavy metal contamination of the estuarine environment in the GG has proven to be a topic of interest at the local level. Environmental degradation of the system could lead to the loss of ecosystem services and threaten the health of people from nearby cities who consume seafood products harvested from the area.
Two patterns persisted in the analysis despite the variability in reported heavy metal concentrations. First, water was highly contaminated by the three focus elements (Cd, Hg, Pb) at the regional level. Second, extreme levels of seafood contamination were repeatedly found in samples from Jambelí, an estuarine area located at the outlet of a gold-mining dominated watershed.
The generalized alert signal indicates that the current practices are threatening artisanal fisheries and, in some cases, the danger signal demands immediate action to mitigate the current situation (Baigún 2013).
Influence of analytical methods on heavy metal concentration values
There were large variations among the heavy metal concentrations reported by different authors studying similar materials, which could be related with the methods used to extract the metals from the samples.
For instance, three authors measured heavy metal concentrations in “soft tissue” of the crab U. occidentalis (Siavichay-Lalangui 2013, Ayala-Armijos et al. 2015, Chuquimarca-Montesdeoca 2015). All these authors used HNO3 to extract the metals, but only Ayala-Armijos et al. (2015) used additional acids in this process (Table II).
Interestingly, the Pb and Hg concentrations reported by Ayala-Armijos et al. (2015) are one and two orders of magnitude above the other two studies, respectively (Table V). The same happened in studies of the bivalve genus Anadara (A. tuberculosa and A. similis), where the only author that reported using additional acids during extraction (Tobar-Ordóñez 2013) obtained values far above those found by authors using HNO3 alone (Ordóñez-Lucin 2015, Carrasco-Peña and Webster-Coello 2016).
TABLE V HEAVY METAL CONCENTRATION RANGES IN SEAFOOD ORGANISMS COLLECTED IN THE GULF OF GUAYAQUIL
| Species (Spanish/English common names) | Concentration* (ppm) | References | ||
| Cd | Hg | Pb | ||
| Crustacea | 0.5 | 0.5 | 0.5 | EC (2016) |
| Callinectes arcuatus (jaiba azul/ arched swimming crab) | 0.01-0.03 | BDL | 0.05-0.57 | Franco-Solórzano (2015) |
| Ucides occidentalis (cangrejo rojo/mangrove crab) | 2.97-3.67 | 37.26-43.10 | Feys-Espinoza (2013) | |
| Ucides occidentalis (cangrejo rojo/mangrove crab) | 0.08-0.10 | 1.47 | 0.96-1.59 | Chuquimarca-Montesdeoca (2015) |
| Ucides occidentalis (cangrejo rojo/mangrove crab) | 0.03-0.04 | 0.01-0.32 | Siavichay-Lalangui (2013) | |
| Ucides occidentalis (cangrejo rojo/mangrove crab) | 137.83-142.54 | 10.26-13.50 | Ayala-Armijos et al. (2015) | |
| Mollusca | 1 | 0.5 | 1.5 | EC (2016) |
| Anadara tuberculosa (concha prieta, pustulose ark) | 1.40-3.97 | 0.242-1.743 | BDL | Ordóñez-Lucin (2015) |
| Anadara tuberculosa (concha prieta, pustulose ark) | 1.68 | 364.38 | 7.52 | Tobar-Ordóñez (2013) |
| Anadara tuberculosa (concha prieta, pustulose ark) | 2.14 | 0.21 | 0.11 | Carrasco-Peña and Webster-Coello (2016) |
| Anadara similis (concha, brown ark) | 1.21 | 618.70 | 8.51 | Tobar-Ordóñez (2013) |
| Crassostrea columbiensis (ostión de mangle, columbia black oyster) | 0.07-0.30 | 0.14-1.5 | Jiménez-Verdesoto (2012) | |
| Crassostrea columbiensis (ostión de mangle, columbia black oyster) | 5.8-17.4 | 0.32-0.96 | BDL | Castro-Infante (2015) |
| Mollusca | 1 | 0.5 | 1.5 | EC (2016) |
| Mytella guyanensis (mejillón/guyana swamp mussel) | 0.09-0.28 | 0.61-2.89 | Banguera-Rodríguez (2013) | |
| Mytella guyanensis (mejillón/guyana swamp mussel) | 0.08 | BDL | BDL | Carrasco-Peña and Webster-Coello (2016) |
| Mytella strigata | 1.7-1.95 | 1.04-1.98 | Mero-Valarezo (2010) | |
| Ostrea columbiensis | 6.25-8.24 | 1.02-1.96 | Mero-Valarezo (2010) | |
| Pomacea canaliculata (caracol manzana/applesnail) | 0.738-1.221 | Ramírez-Prado et al. (2016); Ramírez-Prado (2016) | ||
| Leukoma asperrima | 0.08-2.14 | BDL-0.21 | BDL-0.4 | Carrasco-Peña and Webster-Coello (2016) |
| Teleostei (fishes) | 0.5 | 0.5 | 0.3 | EC 2016 |
| Diapterus peruvians (chaparra/Peruvian mojarra) | 1.55 | 0.80 | 12.30 | Ortega-Pereira 2015 |
| Sardinops sagax (sardina/sardine) | 1.51 | 0.36 | 12.03 | “ “ |
*Heavy metal concentration ranges in seafood organisms collected in the Gulf of Guayaquil, regulated by the European Commission (2016). BDL: below detection level, EU: European Commission. Figures in bold: samples exceeding the EC limits. Empty cells indicate that the metal was not tested. Scientific names follow WoRMS Editorial Board (2016)
Thus, it is possible that many values presented in this study are underestimations of the actual concentrations. However, even studies that did not use additional acids during extraction reported values above those required by the European Commission (EC 2016).
Moreover, studies that used additional acids and found extreme levels (Tobar-Ordóñez 2013, Ayala-Armijos et al. 2015) were both located in the southern sector of the Jambeli archipelago (Fig. 1), raising the question of whether the extreme values are due to the analytical methods, the influence of goldmining, or a combination of both. Therefore, the present literature review suggests that the variability in methods used could have influenced the quantitative values, but overall, there is a high heavy metal contamination in the Gulf of Guayaquil.
Dealing with uncertainty in a precautionary approach
Uncertainty is part of environmental decision-making (Kriebel et al. 2001). As is common in research projects conducted in tropical environments, most local information is written in the native language (Spanish), produced by academic institutions, and not published in peer-review journals (Corlett 2011).
Evidence of known accuracy (i.e., supported by certified reference materials) indicates metal contamination in the fishes Diapterus peruvianus and Sardinops sagax (Ortega-Pereira 2015), arks Anadara tuberculosa and A. similis (Tobar-Ordóñez 2013) and sediments from Estero Salado (Fernández-Cadena et al. 2014).
Sediment samples had the relatively largest amount of data supported through AQA procedures. The rest of the data had unknown accuracy, but often known precision. Since most samples were analyzed in ISO/IEC-17025 certified laboratories, the question of whether reference materials were not used, or not reported by authors, remains.
Insufficient descriptions of analytical quality assurance have been reported by Lewis et al. (2011) in a global review of contamination by anthropogenic chemicals in mangroves, and the GG is not the exception. Implementation of quality assurance and control procedures and improvement of reporting practices in academic theses are needed in further studies to enhance confidence on metal concentrations documented for the GG.
One way to deal with uncertainty is to look for preponderant evidence rather than details (Kriebel et al. 2001). Therefore, a flexible rule was used to define potential risk levels, and the “alert” signal was recurrent. This conclusion is confirmed by published international classifications (NOAA 1990), because the concentration ranges found in the GG fall in different categories of contamination/pollution (Table VI).
TABLE VI EXAMPLES OF CLASSIFICATION SCHEMES OF HEAVY METAL CONTAMINATION PUBLISHED BY NOAA (1990)
| Classification schemes | Concentration (ppm) | ||
| Cd | Hg | Pb | |
| Rotterdam Harbor sediment quality1 | |||
| 1. Slightly contaminated | < 6* | < 1.5* | < 460* |
| 2. Moderately contaminated | 6-9* | 1.5-9* | 110-460 |
| 3. Contaminated | 9-32* | 9-16 | 460-660 |
| 4. Heavily contaminated | > 32 | > 16 | > 660 |
| Great Lakes harbors2 | |||
| Non-polluted | 6 | < 1 * | < 40* |
| Moderately polluted | - | - | 40-60* |
| Heavily polluted | - | > 1* | > 60* |
| Ranges in the GG (this study) | BDL-16.93 | BDL- 7.61 | BDL- 78 |
1Jansen (1987) cited in Long and Morgan (1990); 2 Bahnick et al. (1984) cited in Long and Morgan (1990)
*Levels of contamination in sediments found in the Gulf of Guayaquil, BDL: below detection level
Unless actions are taken to control and limit the sources of contamination, heavy metal accumulation will continue.
Potential threats to public health
Seafood consumption has been identified as the major pathway of human exposure to heavy metals, especially when it represents a large percentage of food intake in a population (Storelli et al. 2005, Martí-Cid et al. 2007, Zheng et al. 2007). Cd, Hg and Pb are produced by different economic activities taking place in GG the basin (Table VII); they are known as non-essential metals, with no demonstrated biological requirement in humans and have the potential to seriously harm human health (Goyer 1997).
TABLE VII BACKGROUND INFORMATION ABOUT HEAVY METALS FOUND IN THE GULF OF GUAYAQUIL*
| Heavy metal sources | Potential effects on human health | References |
| Cadmium (Cd) | ||
| - Soils, rocks, coal and mineral fertilizers - Batteries, pigments, metal coatings, plastics - Electroplating | - Cancer - Lung damage in smokers - Stomach irritation - Kidney disease - Fragile bones | Suwazono et al. (2006), Bernard (2008), Martin and Griswold (2009), Lane et al. (2015) |
| Mercury (Hg) | ||
| - Chlorine gas and caustic soda production - Thermometers, dental fillings, switches, light bulbs, batteries - Coal-burning power plants, biocides, pesticides and household antiseptics - Gold, mercury, aluminum, copper mining - Cement production | - Immune system suppression - Neurodevelopmental problems in prenatal and postnatal periods - Cardiovascular, lung, kidney problems in adults - DNA damage and mitosis impairment | Tchounwou et al. (2003), Castilhos et al. (2006), Mozaffarian and Rimm (2006), Bebianno et al. (2007), Dórea (2008), Feng et al. (2008), Holmes et al. (2009), Martin and Griswold (2009), Selin et al. (2010), Li et al. (2014), Antoszczyszyn and Michalska (2016), Salazar-Camacho et al. (2017) |
| Lead (Pb) | ||
| - Fossil fuel burning - Mining - Manufacturing - Battery production - Ammunition - Metal products like solder and pipes - X-ray shielding devices | - Cancer - Weakness in fingers, wrists or ankles - Increased blood pressure, anemia - Nervous system malfunctioning - Kidney damage - Miscarriage in pregnant women; damage to reproductive organs in men | Martin and Griswold (2009), Almeida et al. (2013), Turner and Solman (2016), Rosen et al. (2017) |
*Summary of sources and effects of heavy metals on human health reported in scientific literature
Seafood consumption has benefits and drawbacks. Fish and shellfish are known for supplying important nutrients associated with a decreased risk for cardiovascular disease (Han et al. 2000, Storelli 2008). Even though thousands of American children are born each year with blood methylmercury levels exceeding the EPA reference dose, evidence shows that benefits of modest fish consumption (1-2 servings per week) outweigh the risks of toxicity by heavy metals (Mozaffarian and Rimm 2006, Chen et al. 2008, Selin et al. 2010, FAO 2016).
Avoidance of fish consumption in a population caused by misleading information may result in excess coronary heart disease deaths and suboptimal neurodevelopment in children (Mozaffarian and Rimm 2006). Thus, the message of this review should not be confused with an advice against seafood consumption, rather, a call for precaution among consumers, and an alert signal for decision makers to focus on the causes of the problem.
Emergent spatial patterns
The spatial information presented in this review (Figs. 3-4) reveals that research efforts have focused on a few sectors of the estuary, leaving large portions of the GG unexplored in terms of heavy metal contamination. Similar patterns of heavy metal contamination in water were found across the three identified sectors.
The influence of agriculture, through the use of fertilizers and pesticides, is reflected in the Daule river’s contamination (Huayamave 2013). However, watershed contaminants are likely to be accumulated in downstream deposition areas (Mackey and Hodgkinson 1995). This is the case of Churute, a protected area located downstream the Guayas river system and used by more than 1400 artisanal fishermen and crab collectors (El Telégrafo 2017), which shows high levels of Cd.
Another recurrent result was the contamination of seafood from Jambeli. This sector is also less studied in terms of water and sediments contamination. Seafood from Jambeli had the highest levels of the three heavy metals focused on in this review. Of these, Hg represents the contaminant of most concern. Jambeli is in El Oro province, where small-scale mining is abundant. This is the most likely source of Hg contamination, as it is used to recover gold from mines through a process of amalgamation (IIED 2001, CPPS 2012).
CONCLUSIONS AND FURTHER RESEARCH
This review is the first peer-reviewed, comprehensive study focusing on heavy metal contamination in the GG. Robust studies included here (Tobar-Ordóñez 2013, Fernández-Cadena et al. 2014, Ortega-Pereira 2015) confirm the general estimations of contamination.
Samples from different sectors of the GG often contain Cd, Hg and Pb concentrations above the international permitted levels, including those found in seafood products. These are non-essential toxic elements that can cause severe health problems to consumers, depending on the level of exposure.
Further research in heavy metal contamination in the region should address topics such as the concentration in traditional seafood products, consumers’ behavior, and levels of exposure among the local population. For instance, crab and black ark collectors spend extended periods in contact with mangrove muds, and anecdotal information indicates they may suffer more health issues than fishermen using boats (Almeida and Kan 2016).
Limited information and uncertainty of available data about metal concentration in the estuary currently allows only a qualitative assessment of the situation. Local researchers have made important contributions through field and laboratory studies, but large information gaps and uncertainty persist. These gaps undermine the promotion of science-based decision-making and systematic conservation planning for the GG.
Collaborative inter-institutional studies could help address these gaps and benefit local laboratories, if performed as part of participatory proficiency testing schemes (Taverniers et al. 2004). Moreover, much of this uncertainty could be eliminated through the adoption of standardized procedures for the testing of environmental contaminant levels.
Finally, regional studies are urgently needed to provide valuable information about spatial trends of metal contamination in the GG, their relation to industry, and their effects on local communities. This is important, because as extractive industries grow in the economy of a developing country (such as mining), the negative spillovers in local communities become more relevant than the social benefits they provide.
These externalities are not only associated with the increase of contamination and its effect in human health and the environment, but also with the upsurge of disputes and violent confrontations between residents and industry representatives (Bebbington et al. 2008).
As a response, the alert signal from the GG demands not only a better management of wastewaters at the catchment level (i.e., agricultural, industrial, urban, mining), but the active involvement of stakeholders and decision makers at different levels of governance, in order to combat the causes of thiscontamination.











 nueva página del texto (beta)
nueva página del texto (beta)




