Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista internacional de contaminación ambiental
versión impresa ISSN 0188-4999
Rev. Int. Contam. Ambient vol.25 no.1 Ciudad de México feb. 2009
Petroleum hydrocarbon assessment in the sediments of the Northeastern Havana littoral, Cuba
Determinación de hidrocarburos del petróleo en sedimentos del litoral noreste de la Habana, Cuba
Eloy Yordad COMPANIONI DAMAS1, Ana Catalina NÚÑEZ CLEMENTE1, Miriam Odette CORA MEDINA1, Luís GONZÁLEZ BRAVO2, Rolando MARBOT RAMADA3, Rodny MONTES DE OCA PORTO4, Maikel ROSABAL RODRÍGUEZ5 and Miguel Ángel DÍAZ DÍAZ1
1 Laboratorio de Química Ambiental, Centro de Investigaciones del Petróleo, 169 Calle Washington # 169, Cerro, Habana, Cuba. Email: elocompa@yahoo.com
2 Centro de Aplicaciones Tecnológicas y Desarrollo Nuclear, 30 Aven., Playa, Habana, Cuba.
3 Laboratorio de Química Analítica, Centro de Investigaciones del Petróleo, 169 Calle Washington # 169, Cerro, Habana, Cuba.
4 Laboratorio Antidoping, Calle 100, Altahabana, Habana, Cuba.
5 Centro de Ingeniería y Manejo Ambiental de Bahías y Costas, Casablanca, Habana, Cuba.
Recibido enero 2008
Aceptado agosto 2008
ABSTRACT
As a part of a geochemical study, aliphatic and aromatic hydrocarbons were determined in surficial sediments, from a Cuban coastal zone located in the Northeastern Havana Littoral. Sediment samples were collected at 15 sites in this area, and then extracted and analyzed by gas chromatography with flame ionization and mass spectrometry detectors. Total concentration of both, aliphatic (AH) and aromatic (ArH) hydrocarbons, varied from 2.4 ± 0.2 to 105.1 ± 5.9 µg/g (dry weight) and from 1.1 ± 0.2 to 38.4 ± 7.6 µg/g (dry weight), respectively. The chromatography profile of AH was dominated by an unresolved complex mixture (UCM), and the presence of isoprenoid hydrocarbons, steranes and hopanes, indicated petroleum – related hydrocarbon inputs. The predominant concentration of phytoplanktonic molecular markers (pristane and nC17) in collected sediments, revealed the marine productivity in this sites. The anthropogenic contribution detected showed the impact of the petroleum exploration along this coastal area.
Keywords: petroleum hydrocarbons, coastal sediments, Havana, Cuba.
RESUMEN
Como parte de un estudio geoquímico se determinaron hidrocarburos alifáticos y aromáticos en sedimentos superficiales, de una zona costera situada en el Litoral Nordeste de La Habana. Las muestras de sedimento se colectaron en 15 estaciones de muestreo en esta área, y posteriormente se extrajeron y analizaron mediante cromatografía gaseosa con detectores de ionización a la llama y espectrometría de masas. Las concentraciones totales de hidrocarburos alifáticos (HA) e hidrocarburos aromáticos (HAr) variaron desde 2.4 ± 0.2 a 105.1 ± 5.9 µg/g (peso eco) y desde 1.1 ± 0.2 a 38.4 ± 7.6 µg/g (peso eco), respectivamente. El perfil cromatográfico de los hidrocarburos alifáticos estuvo dominado por una mezcla compleja no resuelta (MCNR), y la presencia de hidrocarburos isoprenoides, esteranos y hopanos, indicó el aporte de hidrocarburos derivados del petróleo. La concentración predominante de marcadores moleculares fitoplanctónicos (pristano y fitano) en los sedimentos colectados, reveló la productividad marina en estos sitios. La contribución antrópica detectada demostró el impacto que ejerce la exploración petrolera a lo largo de esta área costera.
Palabras clave: hidrocarburos del petróleo, sedimentos costeros, Habana, Cuba.
INTRODUCTION
Research on coastal environments has been recognized as critical for achieving a sustainable management and ecosystem preservation. Pollutants are often introduced in off shore areas by land–production activities and have significant impact on coastal ecosystem and public health. Petroleum hydrocarbons constitute one of the more frequently detected pollutants in marine ecosystems (Hostettler et al. 1999, Eganhouse et al. 2000, Burns et al. 2001, Dojiri et al. 2003, Wu et al. 2003, Ahrens et al. 2004). Extensive transportation of petroleum by ocean going tankers and oil exploration in coastal areas, make marine and coastal environments particularly vulnerable to pollution.
When crude oil enters the surface environment, it is immediately subject to a number of processes that are collective known as weathering (Wang et al. 1995). Some hydrocarbon compounds evaporate, some dissolve, some are dispersed, some are photooxided, some absorb onto suspended particulate materials, and the majority are eventually biodegraded.
Aside from anthropogenic sources, hydrocarbons have also several natural ones such as terrestrial plant waxes, marine phytoplankton and bacteria, biomass combustion, and diagenetic transformation of biogenic precursors (Parrish et al. 2000, Faure et al. 2000, Medeiros et al. 2004). As a result of the variety of their sources hydrocarbons occur as complex mixtures in environmental samples. The differentiation of hydrocarbons with anthropogenic origin from those which are derived from natural sources, or by natural processes, is quite necessary for the assessment of accumulation and biodegradation of these compouns in marine environment.
In aliphatic fraction many sources for hydrocarbons are possible, including oil pollution and natural petrogenic inputs from oil seeps and from the erosion of ancient rocks (Peters et al. 2005). The presence of hopanes, steranes, an unresolved hump, and the lack of odd /even carbon–number predominance of n–alkanes may indicate pollution (Wang et al. 2003). There are also biogenic alkanes and alkenes which are relatively specific to the biota which produces them. Planktonic and benthic organisms synthesize hydrocarbons clearly distinguishable from the hydrocarbons found in the surface waxes of higher plants (Zegouagh et al. 1998, Gomes et al. 2003).
Coastal ecosystem located in Northeastern Havana Littoral, among Bacunayagua inlet (23°08'31" N – 81°40'34" W) and Guanabo strand (23°10'19" N – 82°05'50" W), is one of the most important areas of Cuba used for oil exploration. This area receive domestic untreated wastes from several riverine discharges, however this has not become a drastic environmental quality problem. On the contrary, the oil extraction wells and pipelines located along this coast, has caused the input of petrogenic pollutants towards this marine environment. Previous studies (Nuñez 2002) reported total hydrocarbon concentration in sediments of 5.3 to 300 µg/g. Nevertheless, almost nothing is known in relation to the organic geochemistry of surface sediments of this zone. In this respect, the present paper focuses on the determination of composition, levels and sources of hydrocarbons in sediments of this area. The assessment of the various biogenic and anthropogenic sources was achieved by using molecular marker approach, characteristic compositional patterns and related indices (Simoneit et al. 1982, Simoneit 1984).
MATERIALS AND METHODS
Sampling
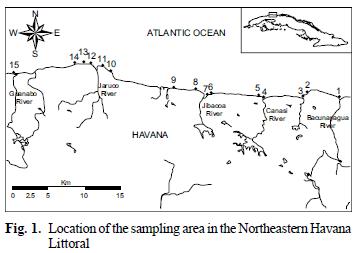
In order to evaluate hydrocarbon inputs from natural and anthropogenic sources to the Northeastern Havana Littoral, 15 sediments samples were collected in March 2006 at the sites shown in figure 1. Sediments were collected utilizing a Van–Veen grab. Only the top 5 cm of undisturbed surface sediment were sampled. Five replicated grabs were taken and homogenized in a bucket to provide a single composite sample for each station. Homogenized samples were placed in pre–cleaned aluminium boxes, and then stored in a freezer at –20 °C until laboratory analysis. Before extraction, sediments were defrosted, dried (45 °C) overnight and passed through a sieve (2000 µm) to remove gravel and detritus.
Extraction and fractionation
About 30 g of homogenized dry sediment were Soxhlet–extracted with 150 ml of dichloromethane for a period of 16 h. Activated powder copper (5 g) was added to the extraction balloons for elemental sulphur removal. The extract was dried in a column containing glass wool and 2 g of Na2SO4, and concentrated on a Kuderna Danish to a volume of approximately 2 ml. The extraction solvent was exchange to ciclohexane and the further volume reduction was achieved with a gentle stream of ultra pure nitrogen until 0.5 ml. The cleanup procedure used a column filled with 7 g of alumina (35 – 70 mesh, Merck) (top) and 7 g of silica (35 – 70 mesh, Merck) (bottom). Both adsorbents were previously activated at 240 °C for 4 h, afterwards 2 % and 5 % by weight of milli–Q water were added to alumina and silica, respectively, to obtain consistent activation. 0.5 g Na2SO4 was added to the top for removing trace amounts of water. Afterwards the extract was adsorbed on to the top of the column. The aliphatic fraction was recovered by elution with hexane (20 ml), and the aromatic fraction with 1:1 (v/v) hexane/dichloromethane mixture (20 ml).The eluates were concentrated with nitrogen until the required volume for analysis by GC/FID and GC/MS.
Instrumental analyses
Aliphatic and aromatic hydrocarbon analyses were conducted on a Konik HR 4000B gas chromatograph with flame ionization detector (GC/FID). The samples were analyzed using a DB–1 fused silica capillary column (30 m, 0.25 mm i.d., 0.25 µm film thickness, Tecknocroma, Spain) with hydrogen as carrier gas. The injector and detector were held at 300 °C. The column temperature program for aliphatic fraction analysis was 60 °C (2 min), followed by heating to 300 °C at 6 °C/min (isothermal hold for 20 min), and for aromatic fraction analysis was 80 °C (5 min), followed by heating to 300 °C at 6 °C/min (isothermal hold for 20 min).
Selected fractions were analyzed by gas chromatography–mass spectrometry (GC/MS) using a Hewlett Packard 6890 equipped with a 5973 MSD. The capillary column used was coated with DB–1 (12 m, 0.20 mm i.d., 0.33 µm film thickness, HP Ultra II, USA) with helium as carrier gas. The injector and detector were held at 280 °C and 300 °C, respectively. The ionization was carried out in the electron impact mode (70 eV). The electron multiplier voltage and automatic gain control target were set at 290 °C and 230 °C, respectively. The MS operated in a total ion current (TIC) mode, and the mass range scanned was from 50 to 550 amu. The column was held at 60 °C for 2 min, increased to 290 °C at a rate of 3 °C/min, and held for 10 min.
Compound identification was based on individual mass spectra and GC retention times in comparison to library data and authentic standards. Standards were injected and analyzed under the same conditions as the samples. GC/FID provides a baseline resolution of n–alkanes from nC14 to nC35 and nC17/pristane and nC18/phytane. Separate external standard calibration curves were established for the quantification of each n–alkane and PAH. Quantification of unresolved complex mixture (UCM) of hydrocarbons was performed using collective calibration curves formed by standard mixtures.
Quality assurance
In order to achieve the improved analytical precision and the accuracy, a number of measures were added to the processing of samples to monitor quality control. Spiked sediment samples with increasing concentration over a range between 33 and 200 ng/g were analyzed. The limit of detection (LOD) and the limit of quantification (LOQ) were determined as the concentration of analyte in a sample that produce a signal–noise ratio (S/N) of 3 and 10, respectively. The uncertainty of quantitative results was estimated using the recovery results according to Barwick and Ellison (1999, 2000). For every bath of 20 sediment samples, a procedure blank was run to check for interference and cross contamination.
RESULTS AND DISCUSSION
Table I illustrates the extraction recoveries and the relative standard deviations (RSD, %) obtained for the spiked levels of 66 and 116 ng/g. LOD and LOQ are also showed. The recoveries of n–alkanes and polycyclic aromatic hydrocarbons (PAH) ranged from 49.1 to 105.6 %. Only the more volatile analytes show recoveries below 40 %. PAH show RSD values slightly higher than n–alkanes. Similar extraction efficiency and precision were achieved by other authors at comparable concentration levels (Jaoen–Madoulet et al. 2000). LOD and LOQ values ranged from 19.26 to 35.97 ng/g and 29.40 to 65.75 ng/g, respectively for n–alkanes, and from 16.74 to 45.87 ng/g and 29.98 to 82.82 ng/g, respectively for PAH.
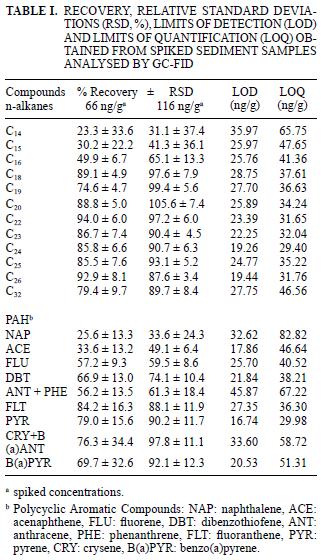
Table II gives the total concentrations of the aliphatic hydrocarbons and n–alkanes ranging from nC14 to nC32, as well as diagnostic criterias useful for the identification of natural and anthropogenic origins of hydrocarbons. The total aliphatic hydrocarbon concentrations varied from 2.4 ± 0.2 to 105.1 ± 5.9 µg/g (dry weight). Higher concentration occurred at sites 10 – 13. These sites are located in an area of intensive oil exploration. Lower concentrations were found for samples 6 and 7. These samples were collected at strands situated far–away from petroleum extraction installations.
The total n–alkane concentrations ranged from 0.10 ± 0.06 to 2.37 ± 0.19 µg/g (dry weight). Higher concentrations were found at sites 1, 9, 12 and 13, and lower concentrations at sites 6 and 7. Total n–alkanes found in the samples represented only a minor amount of the total aliphatic hydrocarbons. The majority of the compounds present in this fraction are molecules that can not be resolved by capillary GC columns and are termed unresolved complex mixture (UCM). The UCM appears in the GC trace as a hump area between the solvent baseline and the curve defining the base of resolvable peaks (Fig. 2). It consists of a complex mixture of branched alicyclic hydrocarbons (Peters et al. 2005), and has a well–known linkage to biodegraded petroleum residues (Wang and Fingas 1995, Wang et al. 1998 a,b, Grossi et al. 2002). The UCM shape depends on the hydrocarbon composition in these samples. Figure 2 shows chromatograms with bimodal envelopes in the elution ranges between nC17 – nC25 and nC25 – nC34.
The high abundance of UCM in all sampling sites (2.3 ± 0.2 – 102.7 ± 5.8 µg/g (dry weight); Table II), is a positive indication of chronic oil – pollution. Additional evidence is provided by the ratio of UCM to NA (UCM/NA) which at all sampling sites take values > 5 up to 61.8. This range characterizes hydrocarbon mixtures with significant contribution from degraded petroleum products (Simoneit and Mazuret 1982, Simoneit 1984). Higher UCM values which correspond to sites 10 – 13, indicates that more significant petroleum–related inputs occurred at this zone.
In the GC–chromatogram of aliphatic hydrocarbons (Fig. 2), the most prominent resolved components are represented by the homologous series of n–alkanes ranging in carbon number from nC14 to nC34. These compounds are shown more clearly in the m/z 85 fragmentograms (Fig. 3) which allowed the characterization of n–alkanes and isoprenoid hydrocarbons, with less interference of the UCM constituents. In the case of weathered hydrocarbons, the chromatograph profiles usually appears with a very different GC trace, in which the n–alkanes are almost completely lost, and only a large UCM is seen in the chromatogram (Charrié–Duhaut et al. 2000). By contrast, the kind of chromatographic features obtained is typically characteristic of a non–weathered and non–biodegraded oil (Wang et al. 1998b).
Carbon preference index (CPI) is used in quantifying plant wax contribution versus fossil fuel contamination (Eglinton and Hamilton 1967, Tulloch 1976). CPI values higher than 4 is commonly associated to biogenic n–alkanes (Jaffé et al. 1996, Wang et al. 2003). In this study, all CPI values in the two ranges (CPIC15 – C20 and CPIC21 – C34) were around 1, which is evidence of a major contribution of fossil fuel hydrocarbons. n–Alkane distributions does not showed any predominance of nC27, nC29 and nC31 (Fig. 4), usually associated to land–derived organic inputs transported toward coastal areas (Eglinton and Hamilton 1967). However, the n–alkane profiles of samples 12 and 13 accounted for the predominance of nC17 in the lower carbon number region (< nC20), which is indicative of the input of marine–derived hydrocarbons (Blumer et al. 1971).
The hydrocarbon composition of many aquatic and photosynthetic bacteria is dominated by the n–alkane nC17 (Giger et al. 1980). This compound has also been detected in many surface sediments, for example: in the Rhone and Ebro River, Spain (Dachs et al. 1999) and in the St. Marys River, Michigan, USA (Tenzer et al. 1999). However, the hydrocarbon nC15, which is usually found in algae (Giger et al. 1980), has not been detected in the samples. The selective degradation or solubilization of biogenic markers in the range nC15 – nC20, with respect to the higher molecular weight homologs has been noticed (Prahl and Carpenter 1984, Marty et al. 1994). Therefore, the low concentration of short–chain n–alkanes could be attributable to the labile character of these compounds.
In sample 13 two different profiles are superimposed. One of them characteristic of biogenic alkanes, with a slight odd carbon number predominance in the range nC19 – nC27 (Fig. 4), as well as the nC17 prevalence in the lower range indicative of autochthonous algal input (Gomes et al. 2003). The second dominant feature is the pronounced UCM hump (from ~ nC17 to nC34) representing fossil fuel contribution (Fig. 2). These characteristics suggest a mixed input of anthropogenic and marine organic matter, although the high UCM concentration (Table II) justifies that the petrogenic contribution is the main source.
The isoprenoid hydrocarbons, pristane (2, 6, 10, 14 – tetramethylpentadecane) and phytane (2, 6, 10, 14 – tetramethylhexadecane), are derived from the diagenetic transformation of phytol and other isoprenoidyl natural products, and are not primary constituents of most terrestrial biota (Peter et al. 2005). However, a higher predominance of pristane over phytane in sediments can be due to zooplanktonic contribution, particularly of calanoid copepods which produce it from the phytol in chlorophylla during their diet (Blumer et al. 1963, Volkman et al. 1992). Uncontaminated sediments frequently present pristane/phytane (Pr/Ph) ratios higher than 1, typically between 3 and 5 (Steinhauer and Boehm 1992). These isoprenoid hydrocarbons were present in almost all analyzed samples, and the Pr/Ph ratios were close to 1 (Table II). Only the samples 4 and 12 showed Pr/Ph ratios slightly higher (2.3 and 2.2, respectively), which could be caused by the mixture of anthropogenic and natural hydrocarbons. This result corroborates the impact of petroleum residues in the study area. Finally, the ratios of nC17/pristane and nC18/phytane (Table II), widely applied as indicators of oil biodegradation, where in the ranges 0.9 – 1.8 and 0.7 – 1.3, respectively. These values correspond to non–heavily degraded oil residues (Wang et al. 1998 a,b), as has been previously stated.
Table III shows the aliphatic hydrocarbons identified by GC–MS–TIC in the samples 1 and 13. Besides n–alkanes and branched alkanes, alkenes were also identified. Biogenic alkenes have been widely found in sediments and algae (Rowland et al. 1985, Belt et al. 2001), and are associated to the production of marine biogenic material in the aquatic environment. The hopanes and steranes detected constitute a further indication of the input of fossil fuel hydrocarbons at these sites (Faure et al. 2000), as already suggested by the existence of UCM and the biomarkers pristane and phytane. The hopane homologue with the thermodynamically more stable 17α(H), 21β(H) configuration was also identified. These hopanes are of the most recalcitrant organic compounds from petrogenic sources found in the environment (Schwarzbauer et al. 2000, Zakaria et al. 2000).
The total aromatic hydrocarbon concentrations are shown in Table II. These concentrations varied from 1.1 ± 0.2 to 38.4 ± 7.6 µg/g (dry weight). Higher levels occurred at sites 10, 12, and 13 located in the proximity of Jaruco River, where the major oil extraction operations are carried out. Lower concentrations were found for samples 2, 3, 5 and 7. Sites 2 and 3 are situated in the farthest portion of an area used for oil extraction. Site 5 is near of oil storage facilities. Site 7 constitutes a strand zone which is more distant from petrogenic sources.
CONCLUSIONS
The anthropogenic tracers, such as UCM, isoprenoid hydrocarbons, steranes and hopanes determined in this study, revealed that this coastal zone from the Northeastern Havana Littoral received fossil fuel inputs. The Pr/Ph, CPI and UCM/NA ratios obtained, lead to the same conclusion. Higher concentrations were detected in sampling sites surrounded the Jaruco River (Sites 10 – 14) (Fig. 1). This is a direct consequence of the intensive petroleum exploration occurring at this area.
On the other hand, the aliphatic hydrocarbon analysis revealed aquatic input in this region. This biogenic contribution was clearly determined by the predominant presence of marine – derived hydrocarbon such as nC17 and the isoprenoid hydrocarbon pristane. Anthropogenic and biogenic hydrocarbons mixture was commonly detected in sediments, with the prevalence of petroleum–derived compounds. The influence of land material inputs (higher plants) were not detected in the study area.
REFERENCES
Ahrens M.J. and Depree C.V. (2004). Inhomogeneous distribution of polycyclic aromatic hydrocarbons in different size and density fractions of contaminated sediment from Auckland Harbour, New Zealand: an opportunity for mitigation. Mar. Pollut. Bull. 48, 341–350. [ Links ]
Barwick V.J. and Ellinson S.L.R. (1999). Measurement uncertainty: approaches to the evaluation of uncertainties associated with recovery. Analyst 124, 981–990. [ Links ]
Barwick V.J. and Ellinson S.R.L. (2000). VAM Project 3.2.1. Development and harmonisation of measurement uncertainty principles. Part D: Protocol for uncertainty evaluation from validation data. Report No: LGC/VAM/1998/088. LGC (Teddington) Ltd., UK. [ Links ]
Belt S.T., Massé G., Allard W.D., Robert J–M. and Rowland S.J. (2001). Identification of a C25 highly branched isoprenoid triene in the freshwater diatom Navicula sclesvicensis. Org. Geochem. 32, 1169–1172. [ Links ]
Blumer M., Guillard R.R.L. and Chase T. (1971). Hydrocarbons of marine phytoplankton. Mar. Biol. 8, 183–189. [ Links ]
Blumer M., Mullin M.M. and Thomas D.W. (1963). Pristane in zooplankton. Science 140, 974–986. [ Links ]
Burns K.A., Greenwood P.F., Summons R.E. and Brunskill G.J. (2001). Vertical fluxes of hydrocarbons on the Northwest Shelf of Australia as estimated by a sediment trap study. Org. Geochem. 32, 1241–1255. [ Links ]
Charrié–Duhaut A., Lemoine S., Adam P., Connan J. and Albrecht P. (2000). Abiotic oxidation of petroleum bitumens under natural conditions. Org. Geochem. 31, 977–1003. [ Links ]
Dachs J., Bayona J.M., Fillaux J., Saliot A. and Albaigés J. (1999). Evaluation of anthropogenic and biogenic inputs into the western Mediterranean using molecular markers. Mar. Chem. 65, 195–210. [ Links ]
Doriji M., Yamaguchi M., Weisberg S.B., Lee H. J. (2003). Changing anthropogenic influence on the Santa Monica Bay watershed. Mar. Environ. Res. 56, 1–14. [ Links ]
Eglinton G. and Hamilton R.J. (1967). Leaf epicuticular waxes. Science 156, 1322–335. [ Links ]
Enganhouse R.P. and Pontonillo J. (2000). Depositional history of organic contaminants on the Palos Verdes Shelf, California. Mar. Chem. 70, 317–338. [ Links ]
Ezra S., Feinstein S., Pelly I., Bauman D. and Miloslavsky I. (2000). Weathering of fuel oil on the east Mediterranean coast, Ashdod, Israel. Org. Geochem. 31, 1733–1741. [ Links ]
Faure P. and Landais P. (2000). Natural and anthropic organic contributions in the sediment of the Kruth–Wildenstein Lake (Hault–Rhin, France). Géochim./Geochem. 330, 39–46. [ Links ]
Giger W., Schaffner C. and Wakeham S.G. (1980). Aliphatic and olefinic hydrocarbons in recent sediments of Greifensee, Switzerland. Geochim. Cosmochim. Acta. 44, 119–129. [ Links ]
Gomes A.O. and Azevedo D.A.(2003). Aliphatic and aromatic hydrocarbons in tropical recent sediments of Campos dos Goytacazes, RJ, Brazil. J. Braz. Chem. Soc. 14, 358–368. [ Links ]
Grossi V., Massias D., Stora G. and Bertrand J.C. (2002). Burial, exportation and degradation of acyclic petroleum hydrocarbons following a simulated oil spill in bioturbated Mediterranean coastal sediments. Chemosphere 48, 947–954. [ Links ]
Hostettler F. D., Pereira W. E., Kvenvolden K. A., van Genn A., Luoma S. N., Fuller C. C. and Anima R. (1999). A record of hydrocarbon input to San Francisco Bay traced by biomarker profiles in surface sediment and sediment cores. Mar. Chem. 64, 115–127. [ Links ]
Jaffé R., Elismé T., Cabrera A. C. (1996). Organic geochemistry of seasonally flooded rain forest soils: molecular composition and early diagenesis of lipid components. Org. Geochem. 25, 9–17. [ Links ]
Jaouen–Madoulet A., Abarnou A., Le Guellec A.–M., Loizeau V. and Leboulenger F. (2000). Validation of fan analytical procedure for polychlorinated biphenyls, coplanar polychlorinated biphenyls and polycyclic aromatic hydrocarbons in environmental samples. J. Chromatogr. A. 886, 153–173. [ Links ]
Marty J.C., Nicolas E., Miquel J.C. and Fowler S.W. (1994). Particulate fluxes of organic compounds and their relationship to zooplankton fecal pellets in the northwestern Mediterranean Sea. Mar. Chem. 46, 387–405. [ Links ]
Medeiros P.M. and Bícego M.C. (2004). Investigation of natural and anthropogenic hydrocarbon inputs in sediments using geochemical markers. I. Santos, SP–Brasil. Mar. Pollut. Bull. 49, 761–769. [ Links ]
Nuñez A. (2002). Study of Petroleum pollution in cuban marine ecosystems. MSc Thesis in Environmental Impact. Instituto de Ciencias y Tecnologías Aplicadas, Habana, Cuba., Havana, Cuba. [ Links ]
Parrish C.C., Abrajano T.A., Budge S.M., Helleur R.J., Hudson E.D. Pulchan K. and Ramos C. (2000). Lipid and phenolic biomarkers in marine ecosystems: analysis and applications. In: The Handbook of Environmental Chemistry: Marine Chemistry (P. Wangersky ed.), Springer–Verlag, Berlin, Vol. 5, pp. 194–223. [ Links ]
Peters K.E., Walters C.C. and Moldowan J.M. (2005). The biomarker guide, Second Edition, Vol. I, Biomarkers and isotopes in the environment and human history. Cabridge University Press, New York, 471 p. [ Links ]
Prahl F.G. and Carpenter R. (1984). Hydrocarbons in Washington coastal sediments. Estuarine Coast. Shelf Sci. 18, 703–720. [ Links ]
Rowland S.J., Yon D.A., Lewis C.A. and Maxwell J.R. (1985). Occurrence of 2,6,10–trimethyl–7–(3–methylbutyl)–dodecane and related hydrocarbons in the green alga Enteromorpha prolifera and sediments. Org. Geochem. 8, 207–213. [ Links ]
Schwarzbauer J., Littke R. and Weigelt V. (2000). Identification of specific organic contaminants for estimating the contribution of the Elbe River to the pollution of the German Bight. Org. Geochem. 31, 1713–1731. [ Links ]
Simoneit B.R.T. and Mazuret M.A. (1982). Organic matter in the troposphere – II. Natural background of biogenic lipid matter in aerosols over the rural western United States. Atmos. Environ. 16, 2139–2159. [ Links ]
Simoneit, B.R.T. (1984). Organic matter of the troposphere: III. Characterization and sources of petroleum and pyrogenic residues in aerosols over the western United States. Atmos. Environ. 18, 51–67. [ Links ]
Steinhauer M.S. and Boehm P.D. (1992). The composition and distribution of saturated and aromatic hydrocarbons in nearshore sediments, river sediments, and coastal peat of Alaskan Beaufort Sea: implication for detecting anthropogenic hydrocarbon inputs. Mar. Environ. Res. 33, 223–253. [ Links ]
Tenzer G.E., Meyers P.A., Robbins J.A., Eadi B.J., Morehead N–R. and Lansing M.B. (1999). Sedimentary organic matter record of recent environmental changes in the St. Marys River ecosystem, Michigan–Ontario border. Org. Geochem. 30, 133–146. [ Links ]
Tulloch, A.P. (1976): Chemistry of waxes of higher plants. In: Chemistry and biochemistry of natural waxes, (Kolattukudy, P.E. Ed.),Elsevier, Amsterdam, 236 pp. [ Links ]
Volkman J.K., Holdworth D.G., Neill G.P. and Bavor Jr. H. J. (1992). Identification of natural, anthropogenic and petroleum hydrocarbons in aquatic sediments. Sci. Total Environ. 112, 203–219. [ Links ]
Wang Z. and Fingas M. (1995). Differentiation of the source of spilled oil and monitoring of the oil weathering process using gas chromatography – mass spectrometry. J. Chromatogr. A. 712, 312–343. [ Links ]
Wang Z. and Fingas M.F. (2003). Development of oil hydrocarbon fingerprinting and identification techniques. Mar. Pollut. Bull. 47, 423–452. [ Links ]
Wang Z., Fingas M., Blenkinsopp S., Sergy G., Landriault M., Sigouin L., Foght J., Semple K. and Westlake D.W.S. (1998a). Comparison of oil changes due to biodegradation and physical weathering in different oils. J. Chromatogr. A. 809, 89–107. [ Links ]
Wang Z., Fingas M., Blenkinsopp S., Sergy G., Landriault M., Sigouin L. and Lambert P. (1998b). Study of the 25–year–old nipisi oil spill: persistence of oil residues and comparisons between surface and subsurface sediments. Environ. Sci. Technol. 32, 2222–2232. [ Links ]
Wu Y., Zhang J. and Zhu Z. (2003). Polycyclic aromatic hydrocarbons in the sediments of the Yalujiang Estuary, North China. Mar. Pollut. Bull. 46, 619–625. [ Links ]
Zakaria M.P., Horinouchi A., Tsutsumi S., Takada H., Tanabe S. and Ismail A. (2000). Oil Pollution in the Straits of Malacca, Malaysia: Application of molecular markers for source identification. Environ. Sci. Technol. 34, 1189–1196. [ Links ]
Zegouagh Y., Derenne S., Largeau C., Bardoux G. and Mariotti A. (1998). Organic matter sources and early diagenetic in Arctic surface sediments (Lena River delta and Laptev Sea, Eastern Siberia) II. Molecular and isotopic studies of hydrocarbons. Org. Geochem. 28, 571–583. [ Links ]














