Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista internacional de contaminación ambiental
versión impresa ISSN 0188-4999
Rev. Int. Contam. Ambient vol.23 no.2 Ciudad de México abr./jun. 2007
Ranking tail–pipe vehicle emissions from reformulated gasolines by Electre method
Ordenamiento de emisiones de gasolina por el método Electre
J. Luis JAIMES–LOPEZ1*, Julio SANDOVAL–FERNÁNDEZ2, Emmanuel GONZÁLEZ–ORTÍZ1, Ángel ZAMBRANO–GARCÍA1, Martín LLANOS–PLATA3 and Uriel GONZÁLEZ–MACÍAS1
1 Dirección Ejecutiva de Investigación y Posgrado, Instituto Mexicano del Petróleo, Eje Central Lázaro Cárdenas Norte No. 152, Col. San Bartolo Atepehuacan, CP 07730, México City, México. Tel. (55) 9175–6756, Fax: (55) 9175–6599, e–mail: jjaimes@imp.mx
2 Instituto Politécnico Nacional. ESIQIE–SEPI. U.P. Adolfo López Mateos, Edif. 8, 2o. piso.
3 General Motors de México, S. de R.L. de C.V. , Industria Minería No. 700, Col. Zona Industrial, Toluca, Edo. de México, C.P. 50000.
* Corresponding author:
Tel.: (55) 7195–6756;
fax: (55) 3003–6935.
E–mail address: jjaimes@imp.mx
Recibido enero 2007
Aceptado junio 2007
ABSTRACT
Even though gasoline reformulation has contributed in abating the high levels of some tropospheric pollutants in México City Metropolitan Area (MCMA), such as lead and sulfur dioxide, it is still being explored as practical alternative to alleviate other local air pollution problems, such as the high ozone levels. Because gasoline is a complex mixture of chemical compounds, the number of alternative reformulated gasoline fuels (RGF) to be tested is potentially very high. Thus, rapid and inexpensive methods are required to make such testing less costly. We report our experience while using a common ranking technique (Electre method) to select a subset of reformulated gasoline fuels (RGF) with low potential to form gaseous toxics and O3–forming compounds in vehicle tail–pipe emissions, so that further studies might focus on the most promising of them. Ten RGF differing in contents of sulfur, aromatics, olefins and oxygenated compounds were subjected to chassis dynamometer tests in two vehicles representing different engine technologies: Tier 1, as representative of the currently dominant technology in MCMA, and Euro 4 which will soon enter the local market. Emissions sampling was done at constant volume (CVS) using the Urban Cycle driving test, a standardized Mexican chassis dynamometer test that simulates the slow driving conditions at MCMA. All comparisons were based on the contents of regulated gaseous pollutants (THC, CO, CO2 and NOx), CH4 and VOC (HC and aldehydes and ketones) in the tail–pipe emissions. In tests with the local unleaded gasoline (n = 9), which was used as experimental reference (RF), the Tier 1 vehicle had significantly higher emissions of most measured pollutants than Euro 4. For selecting the fuels with lower emissions of toxics and O3 forming compounds, the emission data were normalized (0–100) and weighed by two external factors (w1, for O3 forming potential, and w2 for toxicity of each compound) and then subjected to Electre analysis. The best fuel options for data representing the average emissions from the two vehicle technologies were: F11 > F6 > F1 > F7 > F8 > F12, for w1; and F6 > F11 > F1> F5 > F8 > F12, for w2. Thus, by both weighing criteria, the Electre method selected F11, F6 and F1 as the best gasoline options. The ranks of the remaining fuels depended upon weighing. F11 was formulated with low concentrations of aromatics, olefins, benzene and sulfur, whereas F6 had the highest Reid vapor pressure (RVP) and 370 ppm sulfur. According to these results, the MCMA's air quality may be improved by substituting the currently used gasoline, which ranked as the last option, by another such as F11, F6 or F1.
Key words: CVS tests, fuel reformulation, air quality, Electre method.
RESUMEN
Mediante reformulación de las gasolinas se han podido disminuir contaminantes atmosféricos como el plomo y el azufre en la Zona Metropolitana de la Ciudad de México (ZMCM), por lo que la reformulación continúa siendo una alternativa práctica para abatir otros importantes contaminantes locales como el ozono. Pero dado que la gasolina es una mezcla compleja de componentes químicos, el número de mezclas alternativas a valorar antes de introducir una nueva gasolina en el mercado es muy alto. Esto plantea la necesidad de contar con métodos rápidos y de bajo costo para evaluar las alternativas. Aquí se reporta la aplicación de una técnica de jerarquización (método Electre) para preseleccionar un grupo de gasolinas reformuladas con bajo potencial de formación de compuestos gaseosos tóxicos y de precursores de ozono en las emisiones vehiculares de escape, para enfocar estudios más detallados en ese grupo. Se evaluaron diez gasolinas preparadas con diferentes cantidades de azufre, compuestos aromáticos, olefinas y compuestos oxigenados. Para ello se utilizaron dos vehículos con diferente tecnología: un Tier 1, que actualmente es la tecnología dominante en la ZMCM, y un Euro 4, que pronto entrará al mercado local. Las pruebas vehiculares se hicieron en un dinamómetro de chasis utilizando la prueba de manejo estandarizada conocida como Mex Urban, que simula las condiciones de manejo lento "típicas" de la ZMCM. Las muestras de emisiones de escape se obtuvieron con un muestreador a volumen constante (CVS). Las comparaciones entre combustibles y vehículos se basaron en los contenidos de contaminantes gaseosos regulados (hidrocarburos totales, THC; monóxido de carbono, CO; bióxido de carbono, CO2, y óxidos de nitrógeno, NOx); metano, CH4 y compuestos orgánicos volátiles (COV, HC y aldehídos) presentes en las emisiones de escape. Como referencia experimental se utilizó la gasolina sin plomo que se vende actualmente en la Ciudad de México. En las pruebas con esta última gasolina, el vehículo Tier 1 tuvo emisiones más altas de la mayoría de los contaminantes que el Euro 4. Para seleccionar los combustibles con las emisiones más bajas de compuestos tóxicos y precursores de ozono, los datos de la emisión fueron normalizados (0–100) y ponderados por dos factores externos (w1, para el potencial de formación de ozono y w2 para la toxicidad de cada componente) y posteriormente analizados mediante el método Electre. Las mejores opciones de gasolina reformulada (F, fuel) obtenidas con las emisiones promedio de los dos tipos de vehículo fueron: F11 > F6 > F1 > F7 > F8 > F12, con w1; y F6 > F11 > F1> F5 > F8 > F12, con w2. Con los dos criterios de ponderación, el método Electre seleccionó a las gasolinas F11, F6 y F1 como las mejores opciones. El ordenamiento de los demás combustibles dependió también, de los factores de ponderación. La gasolina F11 fue formulada con las menores concentraciones de aromáticos, olefinas, benceno y azufre; mientras que la F6 tuvo la más alta presión de vapor Reid (PVR) y 370 ppm de azufre. De acuerdo a estos resultados, la calidad del aire de la Ciudad de México podría mejorarse sustituyendo la gasolina usada actualmente, la cual quedó clasificada como la última opción, por alguna otra como las F11, F6 o F1, aunque se requieren todavía estudios de mayor detalle.
Palabras clave: Pruebas CVS, combustibles reformulados, calidad del aire, método Electre.
INTRODUCTION
Improving the quality of human life through and industrial development still involves a heavy use of petroleum energy, which implies emision of environmental pollutants. For instance, a petroleum–based transportation is a well–known source of air pollution. Although unwanted vehicle exhaust emissions have been lowered by improvements in engine technology and fuel quality, some highly populated regions, such as México City Metropolitan Area (MCMA), will remain affected by elevated levels of toxic compounds in the air; e.g., nitrogen oxides (NOx), carbon monoxide (CO) and hydrocarbons. These primary pollutants are also precursors of secondary, photochemical pollutants such as ozone (O3), which continues to be a critical air pollutant in the MCMA. As further improvements in the air quality of the MCMA are desirable, it is expected that local regulations limiting vehicle exhaust and industrial emissions will be increasingly stringent.
Air quality can be substantially improved via fuel reformulation since both evaporative and exhaust vehicle emissions are largely determined by the fuel chemical composition. For instance, lowering the olefin content in gasoline reduces the amount of total hydrocarbons (THC) and CO in the exhaust emissions, though some other toxics such as 1,3–butadiene, formaldehyde and acetaldehyde may increase (Schuetzle et al. 1994). New fuel formulations focus mainly on lowering sulfur, aromatics and olefins to reduce direct emissions from these pollutants. Some studies report decreased THC and NOx exhaust emissions by reducing sulfur levels in the fuel (Koehl et al. 1989 and Bensen et al. 1997), or decreased emissions of benzene, toluene, nitrophenols and peroxyacetyl nitrates by reducing aromatic contents (Whitten 1999, Graskow et al. 1998, Auto Oil 1993 and Marshal and Owen 1995). Decreases in HC, CO, and 1,3 butadiene tail–pipe emissions can be also achieved by increasing the contents of oxygenated compounds in the fuels; unfortunately, such benefit is counteracted by increased emissions of formaldehyde (Ragazzi et al. 1999), but this latter effect is minimal for new vehicles equipped with oxygen sensors. Increased RVP increases evaporative HC emissions (Koehl et al. 1989). Thus, a careful prior assessment of toxic emissions is required before releasing a new reformulated fuel to avoid or minimize potential adverse effects in air quality. For such purpose, controlled laboratory experiments are usually conducted as primary source of information.
This CVS study compared the vehicle tail–pipe emissions from 10 reformulated fuels and the regular unleaded gasoline currently in use in the MCMA. Two vehicles with different engine technology (Tier 1 and Euro 4) were used as an emission source. A rapid ranking method (Electre) commonly used in decision making was used to preselect a promising set of alternative reformulated fuels that would better reduce the vehicle tail–pipe emissions of toxic and O3 forming compounds in the MCMA.
MATERIALS AND METHODS
Ten reformulated gasoline fuels differing in contents of sulfur, oxygenated compounds (MTBE), aromatics, olefins and vapor pressure (Table I) were compared by their tail–pipe vehicle emissions in CVS tests. The unleaded gasoline most used in México City served as experimental reference fuel (RF). Originally 13 RFG were prepared, but F4, F9 and F10 were excluded from the final analysis because of measuring problems. All reformulated fuels were prepared from a single gasoline stock provided by the Miguel Hidalgo petroleum refinery (Tula, Hidalgo State, Mexico).
Two vehicles (Tier 1 and Euro 4) were used to test each gasoline. Tier 1 represents the vehicle technology currently in use in Mexico, whereas the Euro 4 cars are programmed to enter the Mexican market in 2008. The main properties of these vehicles are summarized in table II. Euro 4 is basically a car with higher displacement, power and total metal charge compared to Tier 1. Each fuel/vehicle couple was tested within two consecutive days: a reformulated fuel in day one and RF in day two. All tests were conducted between December 2001 and April 2002.
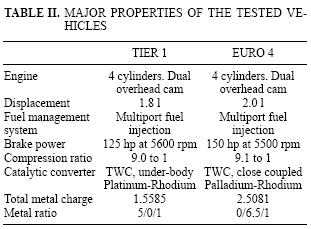
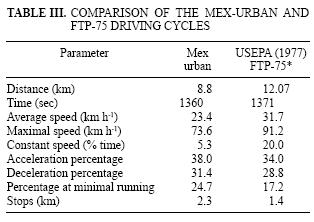
As the fuels are intended for use in the MCMA, the Mex Urban driving test that reproduces the typically slow driving conditions in México City (Díaz–Gutiérrez 2002) was used to generate the emissions. Table III compares Mex Urban parameters against the FTP–75 driving cycle (USEPA 1977). FTP–75 includes a greater distance (27 %), average speed (26 %), maximal speed (19 %) and constant speed (73.5 %); whereas the Mex Urban has higher acceleration (11.8 %), deceleration (9 %), minimal running (43 %) and stops (64 %). All tests were conducted on a 48 inches Horiba chassis dynamometer (Model LDV–48–86–125HP; Road Load Power, AC). Vehicles were previously conditioned according to the NMX–AA–11 –1993 SCFI standard method, which is used to evaluate gaseous exhaust emissions from new gasoline cars in plant (SCFI 993).
Emissions of regulated pollutants were measured for each test (THC, Horiba, model: FIA–236; CO, Horiba, Model: AIA–210; NOx, Horiba, Model: CLA–220; CO2, Horiba, Model: AIA–220 and methane, Horiba, Model: GFA–220). Hydrocarbons (C1–C9) and aldehydes and ketones were measured for samples collected in Tedlar bags by gas chro–matography coupled to ionization flame detector (Agilent technologies, model 6890), using method TO–14 (USEPA 1999a). Carbonyls were trapped with cartridges containing dinitrophenyl hydrazine and quantified by high performance liquid chroma–tography (HPLC, Agilent technologies, model 6890) according to method TO–11A (USEPA 1999b).
The vehicle technology tail–pipe emissions when using the reference fuel were compared by t–tests for unpaired samples (n = 9). In contrast, vehicle emission comparisons for the reformulated fuels were done by t–tests for paired samples because each fuel–vehicle combination had only one replicate.
Fuels were ordered by the Electre method, a ranking technique that compares decision alternatives using concordance and disconcordance concepts (Roy 1991). In this case, the pollutants emitted during each CVS test were used as criteria, whereas the fuel types were considered as alternatives. Prior to ranking, the measured amounts of each pollutant were normalized. Normalization proceeded by column (compound), assigning the maximum value (100) to that fuel showing the lowest emission rate. The remaining column values were obtained by linear interpolation. Each value was then weighed by an external factor (w1) representing a compound's potential to form O3, according to Carter (1994). A second weighing factor (w2) expressed the potential toxicity of each compound (Yin and Pratt 2001). The data matrices weighed by w1 and w2 were separately submitted to Electre analysis. The basic concepts underlying Electre calculations can be summarized as follows:
Concordance is defined as:
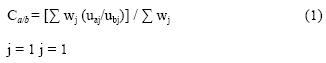
Where:
Ca/b = concordance between alternative a and alternative b
wj = external weighing factor for each criteria (pollutant)
uaj = value of alternative a for each criteria
ubj = value of alternative b for each criteria
Disconcordance is defined as:
Da/b = (Maximum opposition difference between alternatives a and b) / d (2)
Where:
Da/b = disconcordance of alternative a respect to alternative b
d = maximum opposition value
RESULTS AND DISCUSSION
Vehicle emissions comparisons
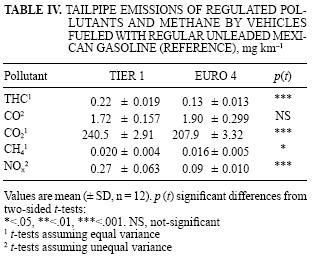
In tests with the regular gasoline currently in use in the MCMA (RF), the Tier 1 vehicle had higher emissions of most measured pollutants than the Euro 4 (Tables IV and V). Tier 1 emitted on average 15.5 % more total regulated gaseous pollutants (THC, CO, CO2 and NOx) than Euro 4 (Table IV). These technology differences were higher and more variable for the low abundance chemical compounds; e.g., Tier 1 produced on average 200 % more NOx (p < .001) and 69 % more THC (p < .001) than Euro 4. Tier 1 also emitted CH4 at slightly higher rates than Euro 4 (p < .05). Euro 4 tended to emit higher levels of CO, but such difference was no statistically significant. Table V summarizes VOC and total carbon emissions. Tier 1 had also significantly higher emissions of most measured VOC than Euro 4: formaldehyde (262 %, p < .001, t–test), acetaldehyde (98 %, p < .001), acetone (34 %, p < .05), methyl ethyl ketone (142 %, p < .001), olefins (65 %, p < .01), naphtenes (229 %, p < .05) and total C (75 %, p < .001). No significant differences between vehicles were detected for emissions of aromatics, paraffins and isoparafins.

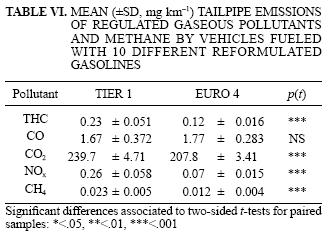
The vehicle emissions comparisons based on reformulated fuels were in general consistent with those observed for the reference fuel (Table VI and Table VII). Tier 1 emitted most regulated pollutants at higher rates than Euro 4: THC (95 %, p < .001), CO2 (15%, p < .001), NOx (253%, p < .001). However, Euro 4 emitted 92% more CH4 than Tier 1 (p < .001) and had slightly higher CO emissions, but this later difference was not statistically significant. In terms of VOC, Tier 1 had a higher mean production of formaldehyde (242 %, p < .001), acetaldehyde (85 %, p < .01), acetone (56 %, p <.05), methyl ethyl ketone (164 %, p < .001), aldehydes and ketones (128 %, p < .001) and olefins (47 %, p< .01). No significant differences were detected for aromatics, paraffins, isoparaffins, naphtenes and 1,3 butadiene emissions. Euro 4, however, emitted benzene at higher rates than Tier 1 (77 %, p < .01).

As indicated by simple correlation tests, the fuel composition related to tail–pipe emissions depended upon vehicle type. For Tier 1, increasing aromatics in the gasoline resulted in higher emissions of THC (r = .73, p = .01), aldehyde and cetones (r = .67, p = .02) and total aromatics (r = .78, p = .01), but it decreased the paraffin emissions slightly (r = –.59, p = .06). In contrast, for Euro 4, adding aromatics to the gasoline decreased the emissions of THC (r = –.63, p = .04), but increased benzene (r = .75, p = .01) and total VOC (r = .76, p = .01). The olefin content appeared associated to several compound increases and decreases in the tail–pipe emissions: for Tier 1, increases were observed for CO (r = .68, p = .02), naphtenes (r = .73, p = .01), benzene (r = .82, p < .01) and total VOC (r = .83, p =< .01), and decreases were observed for formaldehyde (r = –.58, p = .06), acetone (r = –.71, p = .02), methyl ethyl ketone (r = –.66,p = .03) and isoparaffins (r = –.80, p < .02). For Euro 4, the olefin gasoline content produced higher emissions of CH4 (r = .70, p = .02) and decreased emissions of 1,3 butadiene (r = –.64, p = .03). The used contents of MTBE in the reformulated fuels did not correlate with to any observed change in pollutants emission. Sulfur contents only increased the emission of acetone in Euro 4.
According to these results, the introduction of Euro 4 vehicles in the MCMA could be expected to have a positive impact on the local air quality, as it would lower considerably the concentrations of NOx and THC, and to a lower extent CO2 and most measured VOC, in the local air. The better performance of this vehicle can be partially explained by its improved capacity to control the air–fuel ratio precisely and the close coupled three way catalysts (TWC) technology for better light off catalyst performance that means lower THC emissions. Additionally, the Euro 4 vehicle uses Palladium–Rhodium catalytic converter with higher metal contents than Tier 2 vehicles generally focused in controlling emissions of NOx.
Fuel comparisons
Table VIII and table IX show the normalized emission data used for Electre analyses. The RGF preference rankings generated by Electre are shown in table X. Although fuel ranks depended on vehicle engine technology and weighing criterion, RF and F2 ranked consistently as the worst options in all cases; i.e., they had the highest potential to increase toxics and O3 forming compounds in exhaust vehicle emissions. This may be due to the comparatively high contents of aromatics, olefins and benzene in the composition of RF and F2.
Ranking by the average emissions from both vehicles, the best options by both weighting criteria were F11, F6 and F1, i.e., they had the lowest potential production of the studied unwanted compounds. In general, these fuels were formulated with relatively low contents of aromatics, olefins, benzene and sulfur. In particular, F11 contained the lowest olefins content, the second lowest content of sulfur (215 ppm), a low Reid's vapor pressure, and relatively low contents of benzene and aromatics . F11 produced the lowest emissions of aldehydes and ketones, olefins and methane, and it also produced low emission of benzene in Euro 4 (Table VIII). Tier 1 was not as efficient in burning up F1; however, this fuel also had low emissions of naphtenes, 1,3 butadiene and regulated gaseous pollutants (Table VIII). F6 produced the lowest amounts of aromatics, naphtenes and NOx in Tier 1, and it had the lowest production of benzene in Euro 4. F1 was the lowest producer of NOx and CH4, and it was a rather low producer of aromatics, including benzene, in Euro 4. This fuel was not as efficient in Tier 1, but it had relatively low production of aldehydes, NOx and other regulated pollutants.
Other RGF, such as F12 and F13, did well both for Tier 1 and Euro 4. In particular, F12 had the lowest production of both O3–forming and toxic compounds in Tier 1, and it was a low producer of toxics in Euro 4. F13 produced low amounts of O3 precursors in both cars. However, F12 would not be recommendable for use in MCMA because of its high sulfur concentration, and the environmental problems and negative effects of this pollutant on the performance catalytic emission controls (Carter 1994).
Thus, the Electre method was able to distinguish which fuels would potentially improve the air quality in the MCMA by considerabely lowering the concentration of O3 precursors and toxics in the local air. However, these results are not yet conclusive as the raw data set was limited by budgetary restrictions. They however allowed us to select a subset of promising fuels for further detailed studies.
CONCLUSIONS
According to the results, the Tier 1 vehicle emitted a higher volume of THC, NOx, CO2 and CH4 than the Euro 4. Some technological differences, such as a more efficient catalytic converter and two oxygen sensors, make the Euro 4 a vehicle with lower tail–pipe emissions. Three reformulated gasoline fuels (F11, F6 and F1) had the lowest emissions of both O3–forming and toxic compounds. Thus, these fuels are candidates for further studies as they are promising alternatives for improving the local air quality, the best to consider for incorporation into the local market. Fuel F11 was ranked in the first place for both technologies (Euro 4 and Tier 1). This fuel contains the lowest concentration of aromatics, olefins and benzene and it is a good alternative fuel and could be used in the México City metropolitan area.
REFERENCES
Auto Oil Air Quality Improvement Program, Phase 1, (1993). Final Report. Society of Automotive Engineers. [ Links ]
Bensen J.D., Burns V., Gorse R.A., Hochhausser A.M., Koehl W.J., Painter L.J. and Reuter R.M. (1997). Effects of Gasoline Sulfur Level on Mass Exhaust Emission–Auto Oil Quality Improvement Research Program. Report: 912323. USEPA. [ Links ]
Carter P.L.W. (1994). Development of ozone reactivity scales for volatile organic compound. J. Air & Waste Manage. Assoc. 44, 881–899. [ Links ]
Díaz–Gutiérrez L. (2002). Estimación de factores de emisión para vehículos automotores de gasolina. Tesis de Maestría. México. Instituto Politécnico Nacional, 135 p. [ Links ]
Graskow B.R., Kittelson D.B., Abdul–Khalek C.S., Ahmadi M.R. and Morris J.E. (1998). Characterization of exhaust particulate emissions from a spark ignition engine. Paper No. 980528. Society of Automotive Engineers. [ Links ]
Koehl W.J., Gorse R.A., Knepper J.C., Rapp L.A., Bensen J.D., Hochhausser A.M., Leppard W.R., Reuter R.M., Burns V.R., Painter L.J. and Rutherford J.A. (1989). Effect of Gasoline Sulfur Level on Exhaust Mass and Speciated Emission: The Question of Linearity–Auto/Oil Air Quality Improvement Program. Report: 932727. USEPA. [ Links ]
Marshal E.L. and Owen K. (Eds.) (1995). Motor gasoline. The Society of Chemical Industry, Oxford. [ Links ]
Ragazzi R.A., Nelson K. and Gallagher G. (1999). The Impact of a 10 % ethanol blended fuel on the exhaust emissions of TIER 0 and TIER 1, Light Duty Gasoline Vehicles Emissions at 35°F. 9thCoordinating Research Council. On–Road Vehicle Emissions Workshop, San Diego, California, April 19–21. [ Links ]
Roy B. (1991). The outranking approach and the foundation of ELECTRE methods. Theory and Decision, 31, 49–73 [ Links ]
Schuetzle D., Siegl W.O., Jensen T.E., Dearth M.A., Kaiser E.W., Gorse R., Krecher W. and Kulik E. (1994). The Relationship Between Gasoline Composition and Vehicle Hydrocarbon Emissions: A Review of Current Studies and Future Research Needs. Environ, Health Perspect, 102, Supplement 4. [ Links ]
SCFI (1993). NMX–AA–11–1993SCFI. Norma mexicana. Método de prueba para la evaluación de emisiones de gases del escape de los vehículos automotores nuevos en planta que usan gasolina como combustible. Secretaría de Comercio y Fomento Industrial, México. [ Links ]
USEPA (1977). FTP–75, Driving cycle. Federal test Procedure. Title 40. Protection of environment. [ Links ]
USEPA (1999a). Compendium of Methods for the Determination of Toxics Organics Compounds in Ambient Air. Method TO–14–Determination of Volatile Organic Compounds (VOCs) in Air Collected In Specially–Prepared Canisters and Analyzed by Gas Chromatography/Mass Spectrometry (GC/MS). 2nd ed. United States Environmental Protection Agency. [ Links ]
USEPA (1999b). Compendium of Methods for the Determination of Toxics Organics Compounds in Ambient Air. Compendium Method TO–11A–Determination of Formaldehyde in Ambient Air Using Adsorbent Cartridge Followed by High Liquid Chromatography (HPLC). 2nd ed. United States Environmental Protection Agency. [ Links ]
Whitten G. (1999). Potential Extra Air Quality Benefits from Oxygenate That Are Not Required to Meet Reformulates Specification. 9thCoordinating Research Council. On–Road Vehicle Emissions Workshop, San Diego, California, April 19–21. [ Links ]
Yin W.C. and Pratt G.C. (2001). Analysis of air toxics emission inventory: Inhalation toxicity–based ranking. J. Air & Waste Manage. Assoc. 51. 1129–1141. [ Links ]














