Foreword
AD is a clinical entity frequently sub-diagnosed and not extensively treated; in fact, a review of current literature shows that AD is only briefly and superficially considered on preventive cardiology and lipid specialized clinical guidelines.1
AD is characterized by an increase in triglycerides-rich lipoproteins (TGRLP) and other atherogenic particles in plasma. Scientific interest in these TGRLP has fluctuated during last years from being an important cause of cardiovascular atherosclerotic disease (CVAD) to discard them as an important cardiovascular risk factor (CVRF). Recent epidemiological and Mendelian randomization studies have made reconsider the importance these particles have as a cause for CVAD and their role on residual risk creating a general agreement on the necessity for their optimal treatment as a way to reduce the incidence of new cardiovascular events.2
LA is a subcontinent on its own with ethnic, socioeconomic and cultural characteristics that differentiate it from other regions of the world, and at present time it is undergoing an epidemiological transition and entering in a new age of development that exposes its population to adverse variables such as a substantial increase in consumption of ultra-processed foods (UPF) and food with high caloric density (as fried flour and beverages with a high sugar content), that together with a sedentary lifestyle related to the process of migration from rural areas to cities, are causing a significant increase in obesity/overweight and cardiometabolic diseases (CMD) higher than the ones registered in other regions of the world.3
These circumstances and some clinical isolated observations have made us suppose that LA countries may have a superior prevalence of AD that is not prevented, diagnosed and treated properly.
To review the current evidences and obtain conclusions over this topic, a group of experts named Latin American Association for the study of Lipids (ALALIP) was created. This group had the mission to create a document for showing the prevalence of AD in LA and for making specific recommendations to optimize its prevention, diagnosis and treatment. These actions could derive in elaboration of public policies to reduce cardiovascular morbidity and mortality in our region.
Methodology
A modification of the Delphi method4 was used to obtain among the authors of this document a systematic and structured communication and draw conclusions of consensus derived from ample discussions of the available evidences of AD in LA.
A panel of academic experts from different Latin American countries (Mexico, Guatemala, Dominican Republic, Venezuela, Colombia, Ecuador, Peru, Chile, Argentina, Paraguay and Brazil) were selected on the basis of their expertise in lipidology, nutrition and CMD. The whole panel was organized in small task force groups to answer specific questions previously suggested by the coordinators. Each group answered its corresponding questions and later on all the groups met for discussing extensively all questions and answers and to elaborate a final document. The group considered as «unanimous» all those recommendations that reached a 100% of acceptance; as «of consensus» those with at least an 80% of acceptance; and as «discrepant» those with less than 80%. All recommendations and conclusions were in the final document.
To answer each question a systematic review of available publications was made using as key words atherogenic dyslipidemia, triglycerides, triglyceride rich lipoproteins, high density lipoprotein cholesterol, cardiovascular risk, obesity, metabolic syndrome, cardiometabolic diseases and Latin America. The search was made using the bibliographic bases of MEDLINE, SciELO, Revencyt, BIREME, ScienTI, LIVECS and PERIÓDICA. Non-indexed medical literature as well as official publications and those from international organizations were also selected if the methodology used for its elaboration was considered appropriate. Priority was given to publications with data derived from epidemiological and clinical research in LA and/or with conclusions applicable for this region.
Definition of atherogenic dyslipidemia
AD is classically defined as a group of lipid phenotypic alterations characterized by:
High concentrations of TGRLP.
Normal or slightly elevated serum levels of low density lipoprotein cholesterol (LDL-C) mass but with a higher number of particles due to a predominant amount of small and dense LDL-C particles.
Elevated non-high density lipoprotein cholesterol (Non-HDL-C), obtained by subtracting the value of HDL-C from the value of total cholesterol. This Non-HDL-C includes cholesterol from all atherogenic lipoproteins: LDL-C, quilomicron remnants, very low density lipoprotein cholesterol (VLDL) and its remnants, intermediate density lipoprotein cholesterol (IDL-C), and lipoprotein «a» [Lp(a)].
Low levels of HDL-C.
This altered lipid profile favors the initiation and accelerated development of atherosclerosis,5 it is clinically associated to CVAD, and poorly reflected in the traditional scales of risk.6,7
Epidemiology of atherogenic dyslipidemia in Latin America
Cardiovascular diseases are the first cause of death in LA, being myocardial infarction (MI) and stroke responsible for 70% of these deaths; in both conditions atherosclerosis plays the major role influenced by the demographic, socioeconomic and cultural changes seen in LA during last few decades. Facts like increases in life expectancy, migration from rural to urban areas, modification of feeding patterns, increases in tobacco use, in obesity and sedentary lifestyle, together with a limited access to a proper health care, have definitely influenced the growing incidence of CVAD in LA.8-11
What’s the prevalence of atherogenic dyslipidemia in Latin America?
To determine the prevalence of AD in LA a search of all studies done in LA was made using representative samples of the general or regional population that included a serum lipid profile (see Table I on the appendix).
The 1992-1993 Health Survey from Mexico included 2,256 adults from 20 to 69 years of age with blood samples taken after 9-12 hours of fasting. In this urban sample, selected from 417 cities with populations higher to 2,500 inhabitants, it was reported a prevalence of HDL-C < 35 mg/dL in 46.2% of men and in 28.7% of women; there were no differences in prevalence between different age ranges. A level of triglycerides (TRG) > 150 mg/dL was found in 49.7% of men and in 30.8% of women, with an increase across years of age. A combination of low HDL-C and moderately high TRG (values > 200 mg/dL) was observed in 12.9% of the total sample, but being more prevalent in men than women (20.9% vs. 7.2%, respectively).12
This same survey was repeated in 2006 in a sample of 4,040 individuals with a similar age range finding a higher frequency of this combination of low HDL and high TRG (18.3% in total, meaning a 41.8% of increment as compared with the previous study).13
The Chilean National Health Survey from years 2010-2011 used a national randomized sample of 4,965 subjects from 18 to 74 years of age. The prevalence of TRG ≥ 150 mg/dL measured after 9 hours or more of fasting was 31.2%, being more frequent in men than in women (35.6% vs. 27.1%, also after a fasting of 9 or more hours, was 45.4% (more frequent in women than in men: 52.8% vs. 37.6%, respectively).14
A national study on CRFS, with a nationwide population sampling made in Dominican Republic: EFRICARD,15 which included 4,976 adults between 18 and 75 years old reported: a prevalence of HDL-C < 40 mg/dL of 30.7 being significantly higher in men than in women (40% vs. 26.4%. p < 0.0001), and a prevalence of TRG > 150 mg/dL of 21%, also being more prevalent in men (26.3 vs. 18.3% p < 0.004).
In the Latin American Consortium of Studies in Obesity (LASO)16 an analysis of the results obtained from 11 cross sectional independent population studies was made. This study was performed in eight LA countries (Argentina, Chile, Colombia, Costa Rica, Dominican Republic, Peru, Puerto Rico and Venezuela) on 31,009 participants and showed a 53.2% in the prevalence of low HDL-C (defined as < 40 mg/dL in men and < 50 mg/dL in women), and a prevalence of 25% in high TRG level (≥ 150 mg/dL). There was no central laboratory for sample processing.
The study CESCAS I,17 a prospective cohort study made from 2013 to 2014 in 4 cities from the southern cone of America (2 cities from Argentina, 1 from Chile and 1 from Uruguay), included 7,524 subjects from 35 to 74 years of age and reported a prevalence of 34.1% in low HDL-C (< 40 mg/dL) and of 26.5% of TRG ≥ 200 mg/dL. There are some reports from different studies made using randomized general population samples coming from different LA countries: In Peru, the studies PREVENCION18 made in Arequipa; CRONICAS,19 made in Lima, Puno and Tumbes; MIGRANT,20 made in Ayacucho and Lima; studies made on Sao Paulo, Brasil;21,22 on the city of Talca, in Chile,23 and in Mexico City (see results on Table I).24
In Venezuela, a study made in 3,108 subjects with 20 or more years of age from the city of Maracaibo25 showed an age-adjusted prevalence of AD was 24.1% with a low HDL-C frequency of 65%. Data from 4 other more recent studies, also made in Venezuela, has suggested that the prevalence of AD in this country is 24.7%.26
It could be concluded, despite of all this scattered data, that there is an urgent need to design a global LA epidemiological study in order to find out the real prevalence of AD in our region.
Are the lipid alterations found in LA similar than the ones present in the rest of the world?
When comparing HDL-C and TRG data from LA with the one from other countries, a higher prevalence of these alterations is found in LA.
In USA, the National Survey on Nutrition and Health (NHANES 2009-2010) showed a low HDL-C prevalence (30.1%, 95% CI: 29.9-33.2%) which is lower than the prevalence found in LA.27 In Spain, a study made in the province of Murcia showed a prevalence of low HDL-C of 27.3% (95% CI: 25.1-29.4%).28 Both studies, the American and the Spanish ones used as a cut point a level of HDL-C < 40 mg/dL in men and < 50 mg/dL in women. These results sugest that in LA the reported prevalence of low HDL-C is higher than the one reported in some other parts of the world, being the highest prevalence in women as compared with men.
If we consider as high a TRG level ≥ 150 mg/dL, the prevalence of hypertriglyceridemia showed in the LA studies -with the only exception of LASO study16- is, in general, higher than the one found in the NHANES study were it was 24.3% (95% CI: 21.6-26.9%).27 Same happens in the above mentioned Spanish study were it was of 22.8% (95% CI: 25.1-29.4%).28 In LA the hypertriglyceridemia prevalence is higher in men than in women.
Finally, it must be mentioned one study made in Venezuela that showed a lower prevalence of low HDL-C in the Amerindian population as compared with whites and African-descendants.26
In LA there are no studies that have quantitated the levels of small and dense LDL-C (sdLDL-C).
Which could be the causes for the prevalence of these lipid abnormalities in LA: socioeconomic and cultural, feeding patterns, genetic/epigenetic?
Socioeconomic and cultural causes
LA has a series of ethnic, economic and cultural characteristics that are unique of it and help determine the high prevalence of CMD in this region. Some data show differences in the mechanisms and importance of each of the risk factors on the LA population.29 Also, in LA psychosocial stress must be considered as an additional risk factor for cardiovascular disease.30
Feeding
Patterns of feeding can modify the prevalence of CMD.
a) Fats: in 2014 the study NutriCoDE from The Global Burden of Diseases Nutrition and Chronic Diseases Expert Group31 made a systematic analysis of 266 national surveys of adult nutrition looking at the consumption of saturated fats, and omega 6 Fatty Acids (FA), omega 3 FA from fish, omega 3 FA from plants, trans fats and cholesterol from diet.
This study showed that there is a high consumption of cholesterol and trans fats in LA, particularly in Mexico where it can be as high as 4.5 g/day. Saturated fats are consumed in a similar way to other underdeveloped regions of the world but unsaturated FA are less consumed, specially omega 3 FA obtained from fish; there are countries like Argentina, Bolivia, Mexico and Paraguay where its consumption could be less than 50 g/day.
Most important sources of fats in LA are lard and fried foods, specially fried carbohydrates preparations.
b) Sugars and ultra-processed foods: consumption of food with a high caloric content is related to AD and it is also a causal agent for obesity, diabetes and CVAD.
Refined sugars, soft beverages with sugar added and ultra-processed foods (UPF), that are preparations ready to eat or drink with a high content of calories, sugars, fats and sodium, are extensively consumed in LA since they are practical, ubiquitous, very well publicized, with an intensive flavor and able to generate habit.32,33
UPF are associated to an increase of body mass index (BMI) in adults at all levels of consumption after covariate adjustment (R2 = 0.79; p < 0.0001). Per capita sells of UPF in Kg is an independent predictor of increases in BMI across time.32
The Pan-American Health Organization made a study to estimate UPF consumption tendencies using selling information coming from the data base of Euromonitor International 2014.34 This study did a time series analysis using national surveys from 12 countries across the years 1999 and 2013 and looked for the association between changes in per capita sells of UPF (in kg) and average standardized changes in adults BMI.
BMI data was obtained from the Global data Base of the World Health Organization and data for co-variables was obtained from the World Bank data base. This study showed that the LA market is third on the world after Asia and Canada with a 50% increase between years 2000 and 2013. On this same year 2013, the LA sells of soft beverages was higher than the ones on the USA market.33
There are differences in the consumption of UPF in LA: Mexico, Argentina, Chile, Bolivia and Paraguay are, in this order, the 5 LA countries with the higher per capita UPF consumption and, at the same time, they are the 5 countries with the highest BMI.33
Educating the population and stablishing public policies and rules to reduce consumption of those products is an unfulfilled opportunity to reduce obesity and CMD in LA.
Genetics/epigenetics
Epidemiology studies have shown that the most frequent lipid abnormality in LA is a low level of HDL-C. A recent study has suggested that there is a genetic factor associated to hipo alpha-lipoproteinemia in populations of native ascent in LA.35 This study, that examined the impact of genetic factors related to the frequency of AD in native populations of LA, confirmed that low HDL-C is the most common lipid abnormality with a prevalence ranging between 40-50%. Contribution of this lipid abnormality for the metabolic syndrome is higher in persons with Amerindian ancestors than in other ethnic groups.
R230C variant for the « ATP-binding cassette transporter » (ABC-A1), associated to low levels of HDL-C is common in mestizos with native ascent (10% in Mexican mestizos). This variant seems to be specific for Amerindian populations where the frequency of the allele is 0.28 in Mayas, 0.214 in Purepechas, 0.23 in Yaquis, and 0.179 in Tenek; on the contrary, R230C allele has not been found in African, European, Chinese or south Asian populations.
Genetic code is not fix and invariable and today it is totally accepted that ambient changes can induce alterations like methylation and acetylation on the chromatin that alters the expression of this genetic code modifying the phenotype. These changes are collectively called epigenetics.
Chromatin epigenetic modifications are related to the level of activation of some cell functions and could be transferred from a generation of cells to the following one. Epigenetic modifications could generate changes on genetic information transcription (transcriptomic), on the quality and quantity of proteins produced (proteinomic) and in the metabolic pathways regulated by these proteins (metabolomic).36
Fetal and neonatal periods are critical for development and growing. The increase in CMD as it is the case for AD in LA today could be the result of epigenetic changes due to the discrepancy between the nutritional milieu during fetal period and early life, the so called « first one thousand days » and the adult environment; this discrepancy produces a mismatch between fetal programing and changes induced by new styles of living and alters the expression and intensity of the impact of risk factors in populations.37
A major international study on CVRF for a first MI9 demonstrated that in LA, as different as in other regions, the factor with the highest population attributable risk was abdominal obesity. This could be due to epigenetic changes induced by the modifications on the nutritional environment across life.
LA population shows a higher susceptibility to the development of insulin-resistance and low grade inflammation at lower levels of abdominal obesity that could be due to a fetal adaptive response to a deficient nutritional millieu resulting in a loss of structural units like nephrons, cardio-myocites and pancreatic β-cells. These adaptations could become deleterious if in extra-uterine life feeding becomes abundant.29 In LA due to the high frequency of maternal and fetal malnutrition these adaptations could contribute to a higher risk of CMD.
Pathophysiology of atherogenic dyslipidemia and its relationship with cardiovascular atherosclerotic disease
What mechanisms can cause atherogenic dyslipidemia?
Insulin resistance seems to be the main mechanism for AD production. TRG and cholesterol esters are the two most important lipids on circulation; since they are hydrophobic, most combine each one with apoproteins to form lipoproteins and be transported in plasma. Cholesterol is transported by all lipoproteins but mostly by HDL-C and LDL-C.
In general TRG are transported by VLDL, quilomicrons and its remnants; because of its high TRG content all these lipoproteins are collectively called TGRLP. Also, the lipoproteins, besides its content of TRG or C, its size and its density, can be characterized by the kind of apoproteins they have. So, apoprotein B100 (apoB100) is found into VLDL, IDL and LDL-C, and this group is collectively called Non High Density Lipoprotein-Cholesterol (Non-HDL-C). On the contrary, HDL-C has apoprotein A1 (apoA1). Patients with AD have excess of TRG in plasma and hypertriglyceridemia is frequently and independently associated with some metabolic conditions as diabetes mellitus type II, obesity and the metabolic syndrome.38,39
Under normal conditions TRG lipolysis on adipocytes is suppressed by insulin (Figure 1) but in the states of insulin resistance associated with abdominal obesity and overweight this phenomenon does not occur (step 1 on Figure 1) resulting in an augmented production of free FA (FFA) on the portal circulation. These FFA arrive to the liver to be used for an augmented synthesis of TRG by the hepatocyte, being its immediate consequence (step 2 on Figure 1) an overproduction of particles of VLDL-C big and rich in TRG, with apoprotein CIII (apoCIII) that have a longer time of permanence in plasma. Lipolysis of this excess of TRG from VLDL produces remnants of VLDL, IDL and, finally, small and dense particles of LDL-C (step 3 in Figure 1).40,41
Simultaneously, high TRG content on VLDL favors an exaggerated interchange of it for cholesterol between VLDL and HDL-C due to the action of the enzyme cholesterol-esters transfer protein (CETP) (step 4 on Figure 1) producing HDL-C particles abnormally charged with TRG; these particles are later depleted of its TRG content by the enzymes hormone sensible lipoprotein lipase and hepatic lipase (step 5 on Figure 1) producing small and dense HDL-C particles that are less efficient for the reverse cholesterol transport mechanism and are eliminated more rapidly from circulation and consequently reducing its blood concentration.42 In patients with visceral obesity low concentrations of plasmatic HDL-C and apoA1 are related to lower levels of adiponectin.43
May excess of triglycerides be considered as the father of atherogenic dyslipidemia?
Due to their higher size and high content of TRG, on states of insulin-resistance the half-life of TGRLP is prolonged and consequently on the post-prandial period of these patients the plasma TRG are elevated for a longer time than in normal people. This phenomenon is known as post-prandial hyperlipidemia.44
When there is an excess of plasma TRG, cholesterol is transported on a higher proportion by TGRLP and so, hypertriglyceridemia must be considered as a marker of the increase on atherogenic particles like the sdLDL-C (Table I).44
Patients with moderately elevated TGRLP are identified by TRG levels > 200 mg/dL, from these concentrations onward plasma VLDL are partially metabolized particles called VLDL remnants that have a higher cholesterol esters content; in fact, with TRG concentrations > 200 mg/dL or when TRG/HDL-C ratio is > 3.8, more than 40% of plasma cholesterol is bound to the TGRLP. Also, on these patients half-life of quilomicrons and TRG remnants is > 12-24 hours.39
VLDL and its remnants are equal or more atherogenic than LDL-C and although its size is relatively higher than the one of the LDL-C, they have the capacity to penetrate the sub-endothelial space and be phagocyted by macrophages without previous oxidation.
In these patients LDL-C measurement sub-estimates the total charge of atherogenic particles and for this reason on patients with a high level of TRG, the measurement of Non-HDL cholesterol gives us a better estimation of cardiovascular risk since Non-HDL-C includes the cholesterol present on the LDL-C + the one present on the TGRLP.
Which is the role of insulin-resistance? What is first?
The above mentioned alterations on metabolism and lipid kinetics coexist because they have a common base and are frequently found in patients with insulin-resistance and type II diabetes mellitus. Insulin-resistance seems to be a fundamental step on the sequence of abnormalities driving to AD as it is suggested by the strong correlation between insulin secretion response to a glucose oral charge and TRG plasma levels in which a higher resistance to insulin implies higher hypertriglyceridemia.
How is atherogenic dyslipidemia related to associate metabolic conditions?
Insulin-resistance, obesity and type II diabetes mellitus produce increase accumulation of TRG as fat on the liver and this is known as non-alcoholic steatohepatitis. Non esterified or free FA represent the main source of the lipid excess that is accumulated in the liver particularly on states of insulin-resistance (Figure 2). The mechanisms involved in the production of these lipid abnormalities are basically two: 1) excess of hepatic lipids favors an increased incorporation of TRG to the VLDL-C particles that are later secreted contributing to the increase of apoB100 and apoCIII in plasma. 2) There is an over-stimulation of the endothelial hepatic cell lipase that degrades HDL-C particles.45,46
It is not know the mechanism by which the hepatic lipid excess stimulates the synthesis of the lipoprotein regulators but the most accepted hypothesis states that FA oxidation increase their production as well as some coagulation factors. Under physiologic conditions, insulin stimulates phosphorilation of a transcription factor known as «fork-head box protein 01» (FOX01); this protein belongs to a family that plays an important role on the regulation of genes implied in cell growing and neo-glycogenesis.47 On the states of insulin-resistance this pathway is not activated and neo-glycogenesis and de novo lipogenesis are stimulated.48
Chronic hyperinsulinemia also induce de novo lipogenesis by inducing the stimulation of the regulatory element of sterols link to protein 1-c that slows intrahepatic degradation of apoB100,49 and diminishes expression of LDL-C receptors and consequently the clearance of LDL-C.
Which is the role of apolipoprotein CIII?
The apoCIII is a small apolipoprotein that is mainly synthesized in the liver and circulates in plasma associated to TGRLP. It is related to a higher cardiovascular risk probably by inhibiting the union of the apoB100 to its hepatic receptor, reducing in this way the clearance of VLDL by the liver. Also, it has been suggested that apoCIII could inhibit endothelial lipoprotein lipase.50
There are some epidemiological evidences that suggest a relationship between apoCIII and AD:
1. In case and controls studies with clinical and angiographic end points and in observational prospective studies, the apoCIII plasmatic concentrations are strong independent risk factors for cardiovascular disease (CVD).
2. Humans with genetic deficiency of apoCIII have low levels of TGRLP and low incidence of AD.50
Is atherogenic dyslipidemia a cause of cardiovascular disease? Epidemiologic evidences
Plasmatic elevation of LDL-C is one of the most important risk factors for coronary heart disease (CHD) and cerebrovascular disease. Intervention studies with lipid lowering drugs, mainly with statins have shown that reductions in LDL-C produce substantial drops in cardiovascular morbidity and mortality; however, despite of these benefits still persist an important level of residual risk.51 Multiple causes explain the persistence of this residual risk: coexistence of other risk factors, level of individual basal risk, familial and genetic factors, and presence of AD.
It has been proposed that high TRG levels, as an important element of AD, have a predictive capacity for cardiovascular events but a good deal of epidemiological data (see Table II) shows that the Non-fasting TRG levels are most robust predictors of cardiovascular events than the fasting TRG, even after reaching LDL-C goals.
In the Women’s Health Study52 made in 26,509 female subjects, the association between fasting TRG levels and risk of cardiovascular events was not significant (p = 0.90) but when data from non-fasting subjects was considered there was a significant association (p = 0.006) even after an analysis of covariance for total cholesterol, HDL-C and indicators of insulin resistance.
Similarly, the Copenhagen Heart Study53 showed that total mortality and accumulated incidence of cardiovascular events (ischemic ictus, MI and ischemic heart disease) were proportionally related to the increase in non-fasting TRG levels (p = 0.001). These associations were not adjusted for other lipid parameters.
In patients with IHD treated with statins this association was also demonstrated: In the study PROVE IT, TIMI 22, made in patients with an acute coronary syndrome treated with statins to optimal levels of LDL-C, a value of fasting TRG > 200 mg/dL was associated to a higher number of events even after adjustment for HDL-C and LDL-C levels (HR: 0.8; p = 0.025).54 Other studies have not found this positive association between TRG levels and cardiovascular events.55
Based on the disparities found between fasting and non-fasting TRG, it has been suggested that TGRLP levels could be a better marker of risk instead of values of TRG.
A recent Mendelian randomization study,56 an approach that minimizes inverse causality problems and avoid confusion factors, has been made in 73,513 individuals from the cohort of the Copenhagen Heart Study (11,984 developed an ischemic event during the study). In this study a genotype analysis for gen variants that could affect levels of cholesterol, LDL-C, non-fasting cholesterol remnants and HDL-C was made to all participants looking for a causal association between lipoproteins and ischemic heart disease (IHD). The results shown that an increase in 39 mg/dL in non-fasting cholesterol remnants was proportionally associated with a 2.8 times increased risk of IHD independently of low levels of HDL-C. This finding implies that the cholesterol content transported in TGRLP is related to IHD.
Other studies57 with similar methodology have shown a positive association between TGRLP and IHD, low grade inflammation, and total mortality; so, it is fair to conclude in the association of AD with CVAD.
Can atherogenic dyslipidemia produce cardiovascular disease? Pathophysiologic evidences
Atherogenesis is the result of a certain accumulation of plasmatic lipid particles and of an endothelial dysfunction process that allows them to enter the arterial intima. There, processes of inflammation, oxidation and cell migration originate an atherosclerotic plaque and its different clinical manifestations.
Former epidemiologic studies have shown that there is a lineal and continuous relationship between plasma cholesterol levels and atherosclerosis58 and is this relationship the one that has allowed to affirm that, in principle, high plasma cholesterol is the single, independent and totally necessary factor for the genesis of atherosclerosis. However, total value of plasma cholesterol does not allow for a clear difference between healthy and sick individuals in a certain population because the distribution curves of both groups for this continuous variable overlap.59 For this reason, and others derived from basic research, it has been established that the pathogenetic relationship between cholesterol and atherosclerosis does exist but with all cholesterol sub-fractions considered as atherogenic (VLDL and its remnants, IDL, LDL-C and Lp(a); all these molecules share an apoB100 in their composition to facilitate their union to cellular receptors allowing them to give cholesterol esters to the tissues.60
Under normal conditions, approximately 90% of particles with apoB100 are LDL-C molecules61 and represent a strong support for the relationship between this sub-fraction and coronary morbidity and mortality; however, in patients with AD this proportion changes and the percentage of cholesterol transported by LDL-C diminishes to two thirds due to the increase in cholesterol transported by VLDL and its remnants, and IDL.
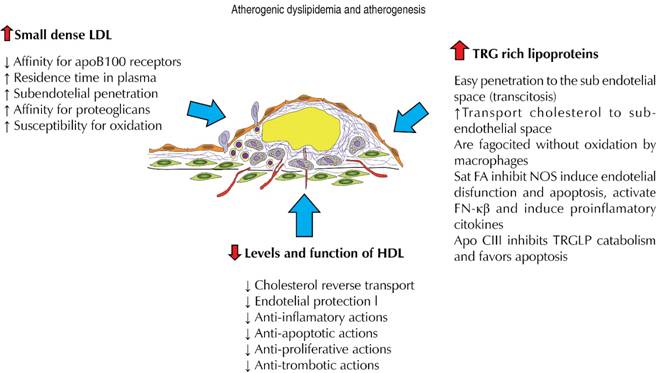
Why LDL small and dense particles are more atherogenic?
Patients with insulin-resistance and AD have an increase in TGRLP with a longer residence time in plasma, facilitating the interchange of TRG and cholesterol esters between TGRLP and LDL-C due to the action of CETP. This produces a structural modification in the LDL-C particles that generates sdLDL-C.
There are some reasons why these sdLDL-C could be more atherogenic than their higher and less dense counterparts:
sdLDL-C have less affinity for the LDL receptor and have a longer residence time in plasma increasing the interaction with the endothelial barrier.
sdLDL-C can penetrate 1.5-1.9 times more the endothelial barrier.
sdLDL-C have a higher affinity for sub-endothelial matrix proteoglycans and this increases their residence time in the intimal space.
sdLDL-C have a higher susceptibility for oxidation and glycosilation).62-66
Between 40 and 50% of all patients with coronary heart disease (CHD) have significant levels of sdLDL-C.67 Some cross sectional and prospective studies of cases and controls have shown the relationship between LDL-C particle size and CHD incidence: on the Quebec Cardiovascular Study, men with LDL-C particle size less than 25.6 nm had a 2.2 times increase on the coronary events incidence in comparison to their counterparts with particles higher than 25.6 nm. The coronary ischemic event predictive value of these small size particles was independent from other plasmatic lipid fractions of these patients.68
Also, on the Cardiovascular Health Study,69 patients with a MI or angina had higher LDL-C concentration and a higher number and smaller size of these particles in comparison to healthy participants.
The Diabetes and Atherosclerosis Intervention Study (ADIS)70 made in type II diabetes mellitus patients treated with lipid lowering drugs, showed that LDL-C final size was inversely correlated with the porcentual increase of the coronary stenosis of these patients.
The ARIC study71 made in 11,419 subjects followed across approximately 11 years showed that the higher quartile of sdLDL-C as compared with the lower one was associated to an increased risk of coronary events (HR: 1.51; 95% CI: 1.21-1.85); even more, sdLDL-C had a higher predictive power (HR: 1.61; 95% CI: 1.04-2.0) on individuals considered of low risk for coronary events according to their LDL-C levels.
Recently, the prospective study of cohorts Ludwigshafen72 showed in 1,643 patients without lipid lowering drugs, who were sent for a coronary angiography and followed for 9.9 years, that LDL-C particles of smaller size (< 16.5 nm) were associated to a higher risk of total mortality (HR: 1.24; 95% CI: 0.95-1.63) and cardiovascular mortality (HR: 1.54; 95% CI: 1.06-2.12) than particles of intermediate size (16.5-16.8 nm). These results were considered as robust even after adjustment for sex, age and other CVRF.
Can triglyceride rich lipoproteins generate atherosclerosis?
As we have already said, TGRLP are a group of particles with varying sizes, densities, and lipoprotein content that have in common a significant proportion of TRG on their composition. This group comprises quilomicrons, VLDL type 1 (higher and less dense), VLD type 2 (of a lower size), quilomicrons remnants, VLDL remnants and IDL.
The TGRLP value is obtained by withdrawing HDL-C and LDL-C values from the total cholesterol (this is also called Non-HDL/Non-LDL cholesterol). From a pathophysiologic point of view these particles could also have an atherogenic potential due to different reasons (Figure 3):73
Due to their small size VLDL type 2 and its remnants can use the transcytosis mechanism, with the protein clatrin covered vesicles, to cross the vascular endothelium and reach the arterial intima.74
Remnant particles have approximately 40 times more cholesterol than the LDL-C, and so they are able to bring more cholesterol to the intima.75
Once on the sub-endothelium, the remnant particles could be phagocited by macrophages without oxidation to produce foam cells.76
TGRLP have been found on human atherosclerotic plaques and on animal preparations suggesting their contribution to plaque formation and to the progress of CVAD.77-79
Another fact implied on the pro-atherogenic role of TGRLP is mediated by lipoprotein lipase (LpL) expressed on the endothelial luminal surface. This enzyme hydrolyses TGRLP and liberates FFA that penetrate by simple diffusion into the endothelial cells where are partially transformed in the hydrophilic Acil-CoAs required for multiple cellular functions. However, when there is a significant proportion of saturated FFA, they can develop pro-apoptotic effects and a direct inhibition of the endothelial nitric oxide synthase, inducing toxicity and endothelial dysfunction in a direct way.
Saturated FFA are also implied on the activation of the kappa-beta nuclear factor and, consequently, on the production of pro-atherogenic cytokines and chemokines as well as adhesion molecules that increase recruitment of monocytes to the arterial wall.80 These effects are not produced by mono or poli-unsaturated FFA, being this the reason for the suggested prevention and/or reduction of these deleterious effects with the ingestion of unsaturated omega-3 FFA.
Apart from the apoB100, TGRLP have as a regular constituent the apoCIII that develops multiple pro-atherogenic actions.81 ApoCIII modulates the activities of different enzymes implied on lipoprotein metabolism and so it can elevate plasmatic concentration of TGRLP by inhibiting its degradation by endothelial LpL, and for reducing its capture by the lipoprotein remnants hepatic receptor. Also, apoCIII can reduce some protective actions of HDL-C, increase endothelial cells apoptosis and produce some direct anti-inflammatory effects as it has being suggested by some experimental studies.82-84
Finally, TGRLP could favor pro-thrombosis by stimulation of tissue factor production from endothelial cells and monocytes,85 by promoting thrombin generation86 and reducing fibrinolytic activity.87
Is atherosclerosis development stimulated by low levels of HDL-C? Is there an effect of TGRLP on HDL-C?
There are multiple anti-atherosclerotic actions related to the HDL-C particles that can be summarized as: 1. reverse cholesterol transport,88 2. endothelial protection,89 3. anti-inflammatory,90-92 4. anti-apoptotic,93 5. anti-oxidative,94 6. anti-thrombotic actions.39,94 All these properties have contributed to the elaboration of a functional athero-thrombotic protection hypothesis that could explain a beneficial effect of HDL-C increase. Some data from human research using infusions of synthetic recombinant HDL or recombinant apoA1/phospholipidic complex have shown the restauration of endothelial function in hypercholesterolemia patients and the reduction of atherosclerotic plaques in patients with acute coronary syndromes;95,96 similar results have also been obtained in other patients with coronary or peripheral atherosclerotic lesions but further confirmation is being required.39
On the contrary, low levels of HDL-C have been associated to the increased production of cardiovascular events.97 In the meta-analysis of the Collaboration Group of Emergent Risk Factors98 made in 302,430 participants from 68 prospective studies, it was shown that for an increment of 15 mg/dL of HDL-C the Hazard Ratio for coronary events, adjusted for lipid factors and clinical data, was 0.71 (95% CI: 0.68-0.71).
Low levels of HDL-C are inversely associated to lipoproteins with apoB100 and for this reason have been considered as biomarkers of atherogenic lipoproteins. This circumstance has been underlined for the lack of association between levels of HDL-C and risk of coronary events in the AFCAPS/TexCAPS study,99 in trials with high intensity statins therapy,100 in the lack of reduction of cardiovascular events in niacin trials,101,102 and in trials made with CETP inhibitors.100,103
It must be mentioned that the use of high intensity statins could increase the expression of micro RNA-33 and hence reduce the expression of ABCA-1 with the consequence of a reduction in the reverse cholesterol transport which could theoretically diminish the beneficial effect of treatments that increase HDL-C.104 Finally, studies of Mendelian randomization have also failed in demonstrate the relationship of HDL-C with production of cardiovascular events.105
HDL-C is a group of particles differing in physical and functional properties, so there are some bigger and lighter (buoyant) known as HDL-C2, and other smaller and dense also known as HDL-C3; these differences are related to their function106 and, although there has been some controversy over its prognostic value, some recent studies have shown a protection effect with the HDL-C3 but not with HDL-C2 or HDL-C total.107
Finally, it must be considered that HDL-C particles especially HDL-C3 can be made dysfunctional and with less anti-atherogenic capacity under increases in oxidative stress and inflammation.108,109 Also, high plasmatic levels of TGRLP generate higher interchange of TRG, from these TGRLP, for cholesterol coming from HDL-C due to the action of CETP; this interchange produces smaller and dysfunctional HDL-C 3 rich in TRG that are more rapidly metabolized, liberating apoA1 that is eliminated by the kidney. Consequently, this causes a significant reduction on the pools of apoA1 and HDL-C with normal function.42
Population and individual measures to prevent and treat AD
How can the development of AD on a population and individual scale be prevented?
The measures targeted to improve eating habits and lifestyle help prevent and minimize complications associated with dyslipidemia. In LA, as described in the epidemiology section, some of the most important causes of AD are malnutrition (in which high intake of sugar and refined carbohydrates with a high caloric density are common), an inadequate relationship between saturated and poli-unsaturated fat intake, and a sedentary lifestyle and obesity.3,31 The physician is responsible for knowing how to modify these variables and for the reduction of cardiovascular risk in our continent.
Multiple interventions have demonstrated their effectiveness at improving the different parameters of AD. Their isolated, short-term impact in associated cardiovascular morbidity and mortality is controversial; still, what is really important is that a global, complete, and long-lasting change is necessary to obtain a significant improvement of cardiovascular risk (Table III).
Table III: Therapeutic objectives based on basal level of triglycerides.

*GCVR = global cardiovascular risk.
TRG are the lipid parameter that best and faster respond to improvements in lifestyle (eating habits, working out, weight loss, abdominal fat reduction). The amount of the TRG response to these changes is directly proportional to their basal level.
The key to prevent AD is nourishment with a proper caloric content, adapted to daily energetic consumption, offering a balance of the macro and micronutrients needed to an appropriate corporal function. The consumption of UPF (carbohydrate-enriched foods that can be easily and rapidly absorbed), as well as a higher consumption of saturated fat and processed products, low on fiber and nutrients, are the principal issues to fix because of their connection to AD.
Classical clinical studies, like the Lyon Heart study and the recent PREDIMED111 based on a Mediterranean diet have demonstrated a lower cardiovascular risk, with benefits to some cardiometabolic parameters. This diet is characterized for its high consumption of olive oil, unprocessed fruits, nuts, vegetables and integral cereals; includes a moderate consumption of fish and poultry and a low consumption of dairy products, and red and processed meat and sweets. European guidelines 2016 recommend the consumption of a Mediterranean diet and they specify the following:112
Saturated FFA must be less than 10% of the total energetic load, and ideally must be substituted by poli-unsaturated FA (PUFA).
Avoid trans fats on processed foods.
Daily ingestion of less than 5 g of Table salt.
30 to 45 g of daily ingestion of fiber, whole grain products, fruits and vegetables.
200 g of fruits per day (2-3 portions).
200 g of vegetables per day (2-3 portions).
Fish at least two times per week, one of which must be oily.
Alcoholic beverage daily consumption limited to two cups (20 g of alcohol) in men, and one cup per day (10 g of alcohol) in women.
Specific nutrients
-
Fats. In June 2008, as requested by the Pan-American Health Organization, a document titled Declaration of Rio de Janeiro «The Americas Free from Trans Fat» was issued. Created as part of a public policy to reduce cardiovascular risk, this document emphatically states that trans fatty acid levels should not be more than 2% in oil and margarine and 5% of total fat in processed foods.113 Trans FA are not nutrients, and their intake reduces HDL-C, increases the levels of LDL-C and the risk of suffering from CHD.114
Saturated FA also increase plasmatic levels of LDL-C, from which the myristic and palmitic acids are the most deleterious. PUFA omega 3 and 6 can be found, at different proportions, in corn, canola, soy and sunflower oil, and mono-unsaturated FA, which are more abundant in olive oil, reduce the plasmatic levels of total cholesterol, LDL-C and TRG. At the same time, they have an impact on the sdLDL and HDL-C. Acceptable amounts of omega 6 are consumed in the traditional western diet; on the contrary, omega 3 must be obtained from an animal source, mostly fish.115
The risk of CHD is reduced in a 2 to 3% for every 1% reduction of TDCI coming from saturated FA and their replacement by PUFA. Adopting these nutritional recommendations can reduce the incidence of cardiovascular events up to a 16% and cardiovascular mortality to a 9%.
The main dietary sources of omega 3 are cold-water fish, fish oils, and some varieties of grains and vegetable oils such as canola, soy, flax, avocado and nuts (Table IV).114-116
Table IV: Mechanisms of action and effects of drugs that reduce TRG levels.
Abbreviations: PPARα = peroxisome proliferator activated receptor α. CETP = cholesterol esters transport protein. DGAT-2 = diacyl-glyceril-acyl transferase. EPA = eicosapentaenoic acid. DHA = docosahexaenoic acid. LpPLA2 = lipoprotein lipase PL2.
Alcohol. Alcohol consumption in amounts and frequency above the recommended limits increases TRG levels significantly and even more than the consumption of simple carbohydrates. Therefore, its intake must always be controlled and verified in patients with AD.
Carbohydrates and UPF. High consumption of carbohydrates that can be found in sugar, sweeteners, flour, and high caloric density foods accumulates and forms functionally inactive adipose tissue. The main sources of high caloric density foods in LA are fried flours (pastries, fry-ups, and others) and UPF.3,31,117 Simple sugar, saturated FA and trans fats are the main components of UPF.
As explained in the epidemiology section, the consumption of these products has been exponentially increasing in the LA market where they are believed to be a major cause of increases in weight and in morbidity and mortality.118 Since UPF are the main cause of obesity and overweight risk, and probably contribute to the prevalence of AD in LA, lowering their ingestion at an individual and population scale has to be the primary objective in the fight against obesity and CMD.3 Physicians must stimulate the consumption of less processed and more natural products. Moreover, it is also important to encourage the creation of public and educational policies to halt the damaging effect of these foods.
Similarly, eating whole fruits or natural non-starchy vegetables on a daily basis reduce in a 4 to 5% the risk of CHD or stroke. It has been demonstrated that people who eat 3 to 5 portions of fruits and vegetables minimize the threat of stroke by 11%, and those who eat more than 5 portions lower the risk by 26%.119
Physicians and scientific societies must encourage some public policies to reduce the consumption of foods with a null nutritional value and consequently to reduce obesity and associated CMD.
Recommended public policies:
To reduce availability and accessibility of UPF: adding taxes and/or limiting their sale in school cafeterias and in stores that do not sell food. (In Mexico, as an initiative of the scientific societies, a tax was assigned to beverages that contained sugar).
To educate the population and to change the perception towards UPF: Demanding a detailed labeling of products and clearly showing the effect of UPF on obesity, and avoiding and restricting publicity of these products.
These actions must be accompanied by educational programs for medical specialists such as pediatricians, cardiologists, specialists in internal medicine and general and family doctors on the effects of these products on health.
What specific diets must be recommended to patients in order to prevent or treat AD?
Adjustment of caloric intake to the patient’s ideal weight with a lower consumption of simple carbohydrates and saturated FA, completely avoiding trans fats, and having a higher consumption of PUFA, as well as fruits rich in fibers and vegetables, are the desired modifications for the diet in the patient with AD or cardiovascular risk.
A meta-analysis of 15 controlled clinical trials showed that the consumption of omega 3 obtained exclusively from fish reduces weight, body fat composition and waist circumference dramatically.116
PREDIMED (PREvention with MEDiterranean Diet111 evaluated the effects of two nutritional interventions on the primary prevention of cardiovascular diseases in individuals with a high cardiovascular risk. The patients were randomized in three groups: Mediterranean diet with an olive oil supplement, Mediterranean diet supplemented with nuts and a control diet in which a low-fat diet was advised. PREDIMED’s Mediterranean diet reduced total mortality by 28% and the risk of cardiovascular mortality by 5%.
The effects of the PREDIMED study on cardiovascular morbidity and mortality demanded adjustments in the classic DASH diet which became HF-DASH diet (modified) allowing a higher consumption of calories derived from fatty foods with cholesterol (e. g.: eggs), increasing from 8 to 14% the amount of calories coming from saturated fats, and reducing the amount of energy from carbohydrates, essentially sugar and juices, by 12% (see appendix).120 These changes improved adherence, and significantly reduced the plasmatic levels of TRG.121
What kind of physical exercise should be recommended to patients in order to prevent and treat AD?
Regular physical activity can reduce cardiovascular risk and mortality. Its clinical benefits are related to the improvement of the values of blood pressure, positive effects on weight control, sensitivity to insulin and glycemic control, lipid profile, and coagulation cascade. There is also an inverse relationship between the level of physical activity and the risk of CHD, having reported reductions of cardiovascular events up to 23%. It is suggested performing aerobic dynamic exercises 5 to 7 times a week for at least 30 minutes (between 2.5 and 5 hours a week), ideally complemented by adding resistance exercises on 2 to 3 days per week, with a total weekly charge of 1 to 1.5 hours. There is evidence that suggests that performing repetitive physical exercises daily for ten minutes could be more beneficial to senior citizens or in patients with a disability than long exhausting sessions.122
Physical activity is usually measured in METS/week. A MET is a unit of measurement of basal metabolism and it is defined as the energy consumed while being seated resting, which is usually 1 Kcal/minute. The expected energetic consumption in an adult is 1,200 to 1,500 kcal/day, depending on the sex, age, physical activity and others.
The most suitable activities for training are those that include exercising many muscles in a continuous and rhythmic form and with a moderate intensity and duration (walking, jogging, swimming, and cycling). The physical activity to develop must be moderate, that is, with a 40 to 59% from the maximal volume of oxygen consumption or from the heart rate reserve (which corresponds to an absolute energy consumption between 4.8 and 7.1 METS in young individuals; 4.0 to 5.9 METS in middle-aged subjects; 3.2 to 4.7 in senior citizens and 2.0 to 2.9 in very old patients.110
How can AD be diagnosed and treated?
Should the lipid profile be done while fasting or not?
This is one of the questions that continue to be controversial. Some recent publications56,123 state that, comparatively, performing the lipid profile without fasting is more useful and more informative to the prediction of cardiovascular risk than the one made on fasting. Moreover, Friedewald124 has assured that the endothelium tends to be more exposed to postprandial lipids.
The European Atherosclerosis Society and the European Federation of Clinical Chemistry and Laboratory Medicine123 recently issued a joint consensus statement in which they affirm that fasting in not routinely required in order to determine the lipid profile. This statement recommends to do the lipid profile without fasting. Abnormal values and measurements must be reported, these being complementary but not exclusive. For this reason, in this document we do not recommend measuring while fasting.
Which of these must be the therapeutic goal in AD: TRG, HDL, apoB100, Non-HDL Cholesterol, or remnants?
Without any doubt the LDL-C must continue to be the primary goal in the treatment of the dislipidemic patient since this is the lipoprotein with the highest amount of evidence that proves its direct association with the risk of CVAD, both in observational epidemiologic studies and in randomized studies with pharmacological therapy which have demonstrated an effective reduction of cardiovascular events when lowering the LDL-C levels51,125,126 That is why, traditionally, guidelines and agreements from multiple scientific societies consider that using LDL-C therapeutic goals can be useful in treatment and follow-up of patients with dyslipidemia.127
However, after the last ACC/AHA 2013 Joint Guidelines did not consider therapeutic goals but only indication of statins depending on their intensity and the individual risk, a controversy in the academic world shows up: does it make sense to consider therapeutic goals or not?128 Regarding this issue, our working group unanimously believes that therapeutic goals are necessary to both the physician and the patient since these allow calculating the intensity of the pharmacological treatment and stimulating the patient to know their risk and keep their therapy. Multiple national guidelines in LA share this view and agree on the need to employ LDL-C goals.129,130
It is true that randomized studies have not had statin dose-adjustments in their design or other drugs to reach a therapeutic goal. However, when analyzing results there is no doubt that the achieved LDL-C levels are associated to a specific CV risk.131 An argument to support our view can be found in the study IMPROVE-IT, in which obtaining LDL-C low values (70 vs. 50 mg/dL) while using statins + ezetimibe, was accompanied by a higher reduction of cardiovascular events despite receiving both arms similar doses of statin.132
In patients with AD, once the LDL-C goal is reached, we must consider as a second therapeutic target the Non-HDL-C levels; this value comprises all TGRLP plus LDL-C and gives us more information on the atherogenic potential of plasma from patients with AD.133,134 We consider that in patients with AD, the sole evaluation and treatment of LDL-C underestimates the residual risk given by the LPRTG.
Determining the level of Non-DHL-C is easy and inexpensive and offers and excellent correlation with plasmatic levels of apoB100.135 It must be remembered that apoB100 is the most precise sign of lipoproteic atherogenic risk since it can be found in all potentially atherogenic particles, including the underestimated Lp (a).136,137 However, its analysis is very expensive and it cannot be easily found everywhere. Although the relationship between apoB100 and apoA1 was analyzed and a great risk among its high tertiles was seen in the study INTERHEART,9 classical studies with statins have reported neither information about the levels of apoB100 nor their therapeutic objectives. As a result, and considering that there is a close relationship between plasmatic concentration of apoB100 and Non-HDL-C, the most reasonable thing to do is to evaluate the latter.
Which drugs have been effective in the treatment of AD and what is their effect on the cardiovascular mortality and morbidity?
We have said that the ultimate goal of lipid-reducing therapy must be LDL-C, but those patients that continue to show high levels of TRG and Non-HDL-C after optimal treatment with TCSL and statins must undergo pharmacological treatment in order to reduce residual risk associated to TGRLP.
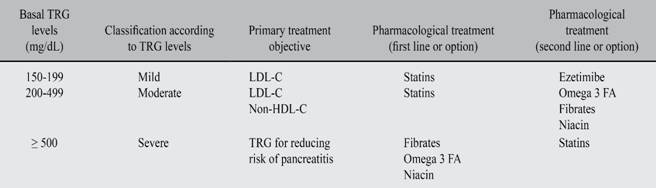
Although the clinical definition of severe HTG varies among guidelines and studies, most of them define it as a TRG level > 500 mg/dL.39,129,137-139 In this case, the reduction of TRG is targeted to reduce the risk of pancreatitis, which is greater that the risk of ischemic cardiomyopathy at those values. This belief is shared in this document, and this is why the following therapeutic recommendations are intended exclusively for patients with a TRG level > 200 mg/dL and < 500 mg/dL; in other words, to patients in which the target is to reduce the risk of CVAD.
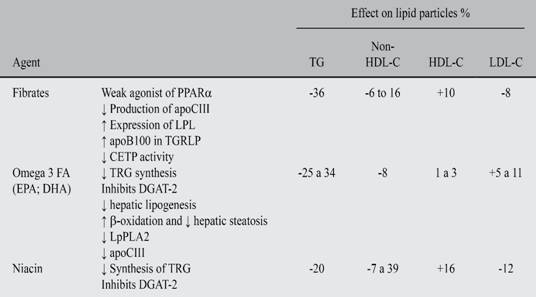
The following drugs have an effect on TRG levels: fibrates, omega 3 FA eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) and niacin (Table III). All of these drugs have metabolic effects on the TGRLP, they reduce effectively TRG and Non-HDL-C and increase HDL-C, although their strength varies: omega-3 FA and fibrates have a lower effect on HDL-C partially dependent on their ability to reduce TRG and the basal levels of HDL-C. Results in clinical tests with these drugs have varied. Yet, when analyzing their use in patients with AD, there is a coincident reduction on cardiovascular risk.
Statins remain to be the foundations in the treatment of dyslipidemia and its associated risk, but since their objective is the reduction of LDL-C, these will not be studied in this document.
Fibrates
Fibrates lower TRG levels in a 36%, levels of Non-HDL-C between 6% and 16% and increase HDL-C in a 10%, and the levels of LDL-C drop to 8%. In patients with severe HTG, a slight increase of LDL-C induced by fibrates can be seen.
So far, RCT intended to study the effect of fibrates on cardiovascular morbidity and mortality have shown some disparities:140-143 some studies suggest a moderate benefit, especially when there are other risk factors besides HTG, which is the case of low HDL-C or metabolic syndrome patients. On the other hand, there are negative results in other RCT.
Meta-analysis of RCT have also suggested benefits with the use of fibrates in HTG. Jun’s meta-analysis made with 18 RCT that used fibrates to demonstrate their effect on cardiovascular risk, which included 45,058 individuals, showed a significant reduction of CVAD (RR 10%; p = 0.048), but with no effect in the total mortality.144 Another meta-analysis, with 5,068 patients with TRG levels of > 200 mg/dL and HDL-C < 40 mg/dL showed a 29% lowering in the number of cardiovascular events. Possibly, this patient profile is to present date the one that has obtained the greatest benefit in the reduction of cardiovascular morbidity and mortality in the RCT made with fibrates.145
Fibrates are drugs that have to be used with precaution when being combined with statins since there is risk of suffering from severe myopathy, rhabdomyolysis and damage to the liver; for this reason, although there is no clear contraindication of their combined use, except with gemfibrozil which cannot be combined with statins, patients undergoing this combination of drugs must be monitored in order to detect muscular symptoms and muscular and hepatic enzyme changes.
Omega-3 FA
The preparations of omega 3 FA that have been defined as pharmacological products by the Food and Drugs Administration of the United States of America, and which have been clinically tested, are purified formulations of EPA and/or DHA presented as mixtures of highly purified ethylic esters (> 90%) of EPA and DHA, or as sole presentations of carboxylic acids of them.
Free formulation presentations that are sold as supplements contain variable concentrations of fish oil, they are not purified and may contain other FA. Also, they could have a variable concentration of toxins and must not be use as treatment drugs for HTG. Omega 3 FA on doses of 2-4 g/day have shown a reduction on TRG levels between 25 and 34%, a drop of 20-42% on VLDL-C levels and increases of 1-3% on HDL-C levels and a slight increase of 5-11% on LDL-C levels.146 Overall, the higher the basal TRG levels, the greater the benefit.
Just like fibrates, RCT with omega 3 FA have shown controversial results.147,148 One meta-analysis made with these studies149 included 63,030 patients and showed a significant reduction in cardiovascular mortality (RR: 0.86; 95% CI: 0.75-0.99; p = 0.03), but it was not beneficial to the total mortality or to a combined cardiovascular end point of MI, stroke and cardiovascular death (p = 0.24 and p = 0.28, respectively). It is important to say that in an analysis of subgroups from this last meta-analysis made on patients with TRG basal levels > 150 mg/dL in contrast to those with < 150 mg/dL, a benefit on the compound end point was shown (RR: 0.82; IC 95%: 0.74-0.91; p = 0.006).
Results of the GISSI-Prevenzione (Gruppo Italiano per lo Studio della Sopravvivenza)150 proved that the use of the combination of highly purified ethilic esters of EPA and DHA administered to post-MI patients achieved TRG reductions of 16-20%, and reduced greatly the risk of re-infarction and death after a 3-year follow-up period.
1.8 g of EPA were used as treatment in JELIS (Japanese Study of Lipid Intervention) showing a reduction of 18% in major coronary events. The effects were more notorious in patients with TRG > 200 mg/dL and HDL-C < 40 mg/dL (with AD) who showed a 53% risk reduction as compared with the effect obtained with statin monotherapy.151
The ORIGIN clinical trial152 with a 2 x 2 factorial design was made with the intention to study the cardiovascular protective effects of Insulin Glargine or Omega 3 FA vs. placebo on patients with type II diabetes mellitus or disglycemia. Omega 3 FA reduced TRG levels on an average of 23% but did not show effect on cardiovascular events reduction; this fact, that seems paradoxical, could be due that placebo group received olive oil that has mono unsaturated FA with some cardiovascular protection properties.
Heterogeneity on the results of RCT may be related to the inclusion of subjects with normal basal levels of TRG (< 150 mg/dL), which is why new studies with omega 3 FA are required to include patients with high cardiovascular risk and TRG levels between 200 and 500 mg/dL.153,154 The most common gastrointestinal adverse effects associated to omega 3 FA are nausea and diarrhea, although these effects are very limited with highly purified preparations. The observed rate of treatment stopping on RCT is similar between omega 3 FA and placebo, and there is no alteration on hepatic function.155,156 Finally, it must be said that there is no interaction whatsoever with statins or other lipid lowering drugs, and for this reason omega 3 FA can be safely used in combination with statins, fibrates or in triple therapy.
8.3. Niacin. Niacin reduces TRG levels in a 20%, LDL-C in a 12%, Non-HDL-C between 7% and 39% and increases the levels of HDL-C in a 16%. But, so far big RCT with niacin added to statins have not shown reductions on cardiovascular risk.101,157-160 A post-hoc analysis have suggested that in patients with TRG > 200 mg/dL and HDL-C < 32 mg/dL (with AD), niacin seems to reduce CVAD events in a 37% (p 0.05).161
Frequent adverse effects limit the use of niacin: the most common is cutaneous vasodilatation or «flushing» which, according to some reports, can be observed in up to 70% of the patients.161-163 Other adverse effects to consider are hyperglycemia, gastrointestinal effects and myopathy.
To evaluate the possible reduction on adverse effects and increases on clinical benefits with the use of niacin, the study HPS-THRIVE was performed on patients with atherosclerosis that were receiving statins.157 This study used, against placebo, a combination of extended release niacin with laropiprant, a selective inhibitor of prostaglandin D which is responsible for the cutaneous adverse effects of niacin. HPS-THRIVE did not show reductions on cardiovascular events on these patients and, contrary to expectations, with the combination of niacin and laropiprant there was an excess of cutaneous, muscular, gastrointestinal and metabolic adverse effects. These results together with the ones obtained on the AIM-HIGH Study161 raise a significant doubt on the use of niacin as a combination therapy on hyperlipidemic patients.
Pharmacologic treatment of AD
Why should AD be treated?
So far, we have exposed, discussed and detailed, epidemiological and pathophysiological evidences that prove that:
AD is a key cardiovascular risk factor in LA, with a higher prevalence and population impact that that the one recorded in other regions of the world.
AD causes CVAD and it is responsible for residual risk.
There is clinical evidence that suggests that AD treatment may alter the course of atherosclerosis, lipid cardiovascular risk (both in primary and secondary prevention) and cardiovascular residual risk.
Having said that, this group concludes unanimously that in LA does exist the necessity to perform a big multicentric international study to evaluate the usefulness of treatment on patients with AD with Omega 3 FA and/or fibrates. Also, a consensus was reached to change current treatment paradigms on patients with AD in LA where, after an optimal statins treatment, Non-HDL-C must be used as a surrogate of TGRLP and treated with the aim to reduce cardiovascular residual risk, both at an individual and a general level.164
How can AD be treated?
The treatment of AD should, at least conceptually, modify underlying metabolic alterations: reduce TRG and sdLDL-C, increase HDL-C and accelerate the clearance of all TGRLP.
To this date, none of the big clinical trials have specifically tested the treatment of AD, therefore the therapeutic approach for patients with this lipid condition is derived from post-hoc analysis of subgroups and from meta-analysis. It is important to say that there are no single drugs that can produce by themselves all the required lipid changes; therefore, combinations of drugs (omega 3 FA and/or fibrates) are suggested, especially on secondary prevention and in patients with cardiovascular high risk and AD that persist with HTG despite optimal treatment with statins.
Maki and collaborators in a recent meta-analysis165 done on a group of RCT made with fibrates, niacin, or omega 3 FA, alone or combined with statins, demonstrate that even though there is no global reduction of cardiovascular risk in all individuals, on the subgroup with elevated TRG and low HDL-C (constitutive elements of AD), there was an important statistical reduction of cardiovascular risk.
Which levels and from which lipid particle must be chosen to treat patients with atherogenic dyslipidemia?
As was previously mentioned, the metabolic alteration that best defines AD is HTG; therefore, the basal level of TRG must be considered as the starting point for establishing therapeutic objectives and for choosing the most appropriate drug therapy166,167 after correction of LDL-C. Also, Non-HDL-C, for its higher capacity to predict cardiovascular risk must be considered as a secondary objective.168
As shown in Table V, TRG levels are used to define the primary therapeutic objective. So, TRG levels > 500 mg/dL demand as a primary goal the risk reduction of pancreatitis, being secondary to this the reduction of cardiovascular risk.169 On the contrary, TRG levels between 200 and 499 mg/dL demand as a primary objective the LDL-C reduction; and secondarily the Non-HDL-C reduction as a combined approach to reduce cardiovascular risk.
Low HDL-C is associated to an increase in cardiovascular risk; however, pharmacologic interventions aimed to increase low HDL-C have not consistently shown a risk reduction of clinical events or mortality. Therapeutic changes in lifestyle and particularly exercise, more than drugs, are the most efficient way to increase HDL-C.127
What is the treatment algorithm for the patient with AD? (Figure 4)

Figure 4: Proposed treatment algorithm for high or moderate risk patients with atherogenic dyslipidemia.
The following algorithm is suggested for the treatment of patients with AD:
Suggesting therapeutic changes on lifestyle (TCL) as an accurate, initial and vital step (Table II).
Evaluate the GCVR of the patient.
Identify the therapeutic objectives (LDL-C and Non-HDL-C) and the goal according to the level of the risk (Table V). On every successive consultation the patient must be inquired on the adherence of treatment. It would be ideal to establish a team work with a nutritionist, a psychologist and a physical therapy technician. Use of electronic applications to help with this work is suggested.
Calculate the LDL-C and Non-HDL-C percentage of reduction required to reach the goal.
Statins are the first therapeutic choice in most patients with AD. Choose the average statin dose that can reach the desired LDL-C goal. In case of not achieving the LDL-C goal, intensify treatment, add ezetimibe or chose a more powerful statin.
Once reached the LDL-C goal, must be seen if Non-HDL goal was also reached; if not use a combination therapy with omega 3 FA or fibrates.
General principles
Traditionally, lipid treatment in the reduction of CVAD is based on therapeutic intervention in high risk patients (acute coronary syndrome patients, stable ischemic heart disease, cerebrovascular disease, peripheral artery disease, diabetes, hypertensive patients with severe dyslipidemia, and patients with multiple uncontrolled risk factors despite receiving effective treatment). However, most deaths related to CVAD occur in low to intermediate risk individuals just because they belong to a larger and more numerous group.170 Therefore, the compulsory initial step to begin the treatment of the patient is to calculate the global cardiovascular risk for estimate the chances of a cardiovascular event in the following 5 to 10 years. This approach has some benefits: first, it allows the classification of the patient level of risk; second, patients can be more involved in the treatment if they are informed of their risk; third, it allows us to better define the therapeutic goals for each individual.171
In order to define the global risk for every patient, multiples formulas and risk Tables can be used; nevertheless, it is not possible to precisely recommend one of them to LA patients since they have not been proved on the region. Framingham score is probably the most used;6 this score predicts coronary disease risk but there are a number of reasonable doubts about the applicability on the Latin population since it can under or overestimate the risk.172 SCORE (Systematic Coronary Risk Evaluation),7 made by the European Society of Cardiology is also widely used: it is the product of the analysis of 12 national surveys from different European countries. However, it is not completely validated in LA.
A potentially trustworthy score that could be used in the region is the product of the INTERHEART Study.173 This tool has been validated in LA population; however, in order to apply it, it is necessary to measure apoB100 and apoA1, which would increase costs and applicability in our region. Finally, the American College of Cardiology and the American Heart Association recently proposed the equation: ACC/AHA-ASCV Risk Estimator Pooled Cohort Equations174 that allows to calculate «life risk» (or in the long term: 20-30 years) which can be useful when evaluating young patients (20-40 years old), with CVAD family history or risk factors. The records used on its design only included a scarce LA population but still it can be used keeping on mind this limitation.
On our group there was some disagreement on which risk scale should be recommended but we did have a total agreement on the suggestion for estimating the global risk of every patient and in always using the same risk assessment tool. Also, this group suggests the urgent need to design and validate a regional tool for the calculation of risk in LA.
Besides the risk calculation, the systematic search of subclinical atherosclerosis must be done exhaustively in order to diagnose the existence of this disease during its preclinical stage (ankle-arm index, coronary calcium score, presence of atherosclerotic plaques in carotids or femoral arteries) and target organ damage (left ventricular hypertrophy, microalbuminuria). Finally, it is important to mention that in LA, psychosocial stress is a very important factor that should be taken into account in every risk calculation.
Identifying therapeutic risk and goal depending on risk level
We have already indicated the need to set therapeutic goals depending on the levels of lipid fractions. Our group unanimously continues to believe that it is imperative to define and reach goals that can be useful in the orientation of therapy, optimize and individualize risk reduction and monitor adherence to therapy. Moreover, we suggest reaching goals according to risk (Table V).
As a rule, Non-HDL-C goals are higher than the LDL-C ones in 30 mg/dL. Consequently, the majority of patients with AD must have a goal of < 100 and < 130 mg/dL on LDL-C and Non-HDL-C respectively. The exception would be patients with established cardiovascular disease (secondary prevention) and patients with diabetes and a cardiovascular risk > 7.5% in 10 years. In this case, the goal would be < 70 and < 100 mg/dL, respectively.
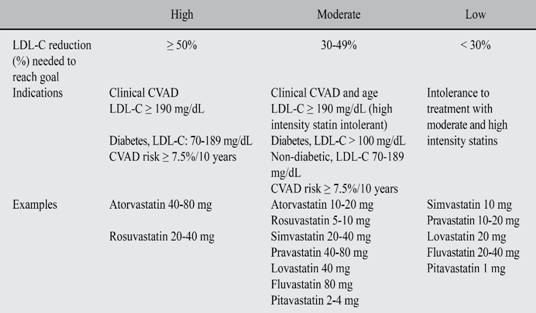
What kind of statin should be chosen?
We adopted the recommendations of the Guidelines to control the high cholesterol levels in adults to reduce cardiovascular risk so as to choose the correct statin and dosage to achieve the LDL-C goal (Table VI).131
Therapy to reach Non-HDL-C goal
Besides its known effect on LDL-C, statins reduce plasmatic levels of TRG in 15-50% and increase HDL-C up to a 15%. These last effects are proportional to the basal level of TRG. As a result, statins are the first therapeutic option to treat patients with dyslipidemia. If Non-HDL-C levels remain high after having reach LDL-C level goals, a combined therapy must be chosen in order to reduce them (Table VII).
Patients with AD that can make the most of the combined therapy are those with TRG > 200 and HDL-C < 40 mg/dL. The major intervention studies with statins (post hoc analysis of > 4,000 patients) support the concept of an added cardiovascular benefit when combining statins with omega 3 FA or fibrates in these patients.145,149,175,176 The new generation of fibrates such as fenofibrate and ciprofibrate have been proved to be safer in combination therapy with statins.
Treatment algorithm
The suggest algorithm designed for patients with a cardiovascular moderate or high risk and/or diabetes mellitus demands the need to always try to reach the LDL-C goal as a primary objective. The Non-HDL-C goal must be evaluated after at least 3 months of optimal fulfillment of the therapy and TCSL and if its goal has not been attained a combination therapy must be used (Figure 4).
Conclusions
In LA the lipid abnormalities related to AD have a high prevalence, probably higher to the one observed in other regions of the world. We have reach to this conclusion after the systematic analysis of national surveys of health and regional cohort studies with sampling population large enough to be considered representative. It is important to clarify that this analysis, even if based on a large number of studies and with a significant population, only allows to set a hypothesis that should motivate a prospective study of global design representative of our entire population (urban, rural, indigenous, African-descendants, mestizos, people from the plains, from the coast and the high mountains) made with a single methodology to study the real prevalence of AD in LA.
A study of this kind could help us to better understand the causes of this presumed high prevalence of AD in LA and to determine its human and economic impact; additionally, this study should be pivotal for the design and implementation of public policies and medical actions that would reduce the prevalence and consequences of AD in LA.
The causes of this high prevalence of AD in LA seem to be multiple; we postulate that they are the result of the interaction, on different grades, of the genetic load and of their epigenetic modifications, combined with a sedentary lifestyle and an inadequate nutrition, mostly driven by a high consumption of UPF, trans fats and cholesterol, and sweets beverages. All these factors are related to the psicocultural and socio-economic variables typical of our region.
Cardiovascular diseases are the leading cause of mortality and disability in our subcontinent, and AD is cause of ACVD. The lipid alterations that define AD increase the risk of cardiovascular and cerebrovascular events and are important factors on the residual risk of these patients; therefore, AD should be diagnosed and treated on an effective and systematic manner.
The primary therapeutic goal in the treatment of AD is still the LDL-C level, defined according to the individual risk of each patient. After achieving this goal with the effective application of the lifestyle changes, the optimal use of statins and eventually with the addition of ezetimibe, it is necessary to reach the Non-HDL-C goal based on the addition Omega 3 FA and/or Fibrates.
Finally we want to state that LA is a region with great economic and social inequities that impose to the population variable difficulties for the access to proper diagnosis and treatment. This is one of the reasons why in LA countries the prevention of cardiovascular diseases, based on a better understanding of our risk factors and our social and cultural characteristics, should be the number one priority with the additional advantage of its high cost-effectiveness. In LA cardiovascular prevention should not be an option but an obligation.
As a corollary, it is necessary to perform suitable epidemiologic research in order to determine the actual prevalence of AD in our region, its true causes, and medical and economic repercussions so as to design and to apply public policies and to take medical actions to reduce its incidence and impact.
Authors members of ALALIP by country
Argentina
Alberto Lorenzatti
Alfredo Lozada
Daniel Piskorz
Daniel J Pineiro
Centro América
Fernando Wyss
Chile
Leonardo Cobos
Raúl Villar
Fernando Lamas
Rodrigo Alonso
Paula Varleta
Mónica Acevedo
Colombia
Alonzo Merchan Villamizar
Juan Manuel Arteaga D
Patricio López-Jaramillo
Ecuador
Ernesto Peña-Herrera
Vladimir Ullairi
Joffre Lara
México
Enrique Gómez Álvarez
Enrique Morales
Paraguay
María Paniagua
Perú
Alfonzo Bryce
Félix Medina
Josefina Medina Lozada
Julio Chirinos
Venezuela
Carlos I Ponte N
Carlos Carrera
Jesús E Isea P
José R Gómez Mancebo
Dra. Livia T Machado
Dr. Juan Colan P











 nueva página del texto (beta)
nueva página del texto (beta)