Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista mexicana de micología
versão impressa ISSN 0187-3180
Rev. Mex. Mic vol.35 Xalapa Jun. 2012
Contribuciones
Elaphomyces appalachiensis and E. verruculosus sp. nov. (Ascomycota Eurotiales, Elaphomycetaceae) from eastern North America
Elaphomyces appalachiensis y E. verruculosus sp. nov. (Ascomycota, Eurotiales, Elaphomycetaceae) del este de Norteamérica
Michael A. Castellano1, Gonzalo Guevara Guerrero2, Jesús García Jiménez2, James M. Trappe3
1U.S. Department of Agriculture, Forest Service, Northern Research Station, 3200 Jefferson Way, Corvallis, Oregon 97331 USA.
2Instituto Tecnológico de Cd. Victoria, Av. Portes Gil 1301 Pte. C.P. 87010, A.P. 175 Cd. Victoria, Tamaulipas, México.
3Department of Forest Ecosystems and Society, Oregon State University, Corvallis, Oregon, 97331 USA
*Autor para correspondencia:
Gonzalo Guevara guevaragg@hotmail.com
Received 28 September 2011;
Accepted 10 February 2012.
Resumen
Se describe Elaphomyces verruculosus como una nueva especie del este de Norteamérica con un rango de distribución que va desde Quebec, Canadá hacia el sur a lo largo del este de E.U.A. y costa del Golfo hasta el noreste de México. Elaphomyces verruculosus es similar en morfología general a E. granulatus de Europa. Además se redescribe E. appalachiensis y se reporta su presencia en Florida, Iowa, Tennessee, oeste de Virginia y México.
Palabras clave: Pinus, Quercus, secotiode, ectomicorriza.
Abstract
We describe Elaphomyces verruculosus as new species from eastern North America, ranging from Quebec, Canada south along the eastern USA and along the Gulf Coast to northeastern México. E. verruculosus is similar in overall morphology to E. granulatus of Europe In addition we re-describe E. appalachiensis and report it as occurring in Florida, Iowa, Tennessee, West Virginia and Mexico.
Keywords: Pinus, Quercus, sequestrate, ectomycorrhiza.
Introduction
Elaphomyces Ness is characterized by having a single, large, empty chamber in youth that soon becomes stuffed with cottony hyphae. At maturity, the chamber is filled with yellow to brown, olive-brown to black or blue to black powdery spore mass (Castellano et al., 1989; Trappe et al., 2009). Elaphomyces species are ecologically important because they form part of the diet of animal mycophagists and are widespread ectomycorrhizal partners to a vast variety of large woody plants (Maser et al., 2008; Trappe et al., 2009).
In Mexico, a variety of species from various sequestrate genera such as Elaphomyces, Gautieria, Genea, Geopora, Glomus, Hydnangium, Hydnobolites, Hymenogaster, Hysterangium, Leucogaster, Macowanites, Melanogaster, Octaviania, Pachyphloeus, Radiigera, Rhizopogon, and Tuber have been reported for the states of Nuevo León, Coahuila, Durango, Tamaulipas and Mexico (Guzmán, 1971; Trappe and Guzmán, 1971; Trappe et al.. 1979; Hosford and Trappe, 1980; Guzmán, 1983; Cázares et al., 1992, 2008; Guevara et al., 2008a, 2008b, 2011; Healy et al., 2009). Recently, we examined Elaphomyces collections from northern Mexico deposited at Oregon State University herbarium (OSC) and fresh material and discovered an undescribed species and one Elaphomyces species previously described from a single locality in the eastern United States. This paper is a contribution to the knowledge of the sequestrate mycota of North America.
Materials and methods
Methods of collection and macroscopic and microscopic study were generally those of Castellano et al. (1999, 2003). Colors of fresh fruiting bodies are in general terms by the authors. Dried specimens were hand-cut and mounted in 5% KOH or water for microscopic observation. Twenty spores were measured to estimate range and average size. Spore dimensions are with ornamentation. Herbaria are abbreviated according to Index Herbariorum (2011).
Results and discussion
Elaphomyces appalachiensis Linder, J. Elisha Mitchell Sci. Soc. 55: 133. 1939. Figure 1
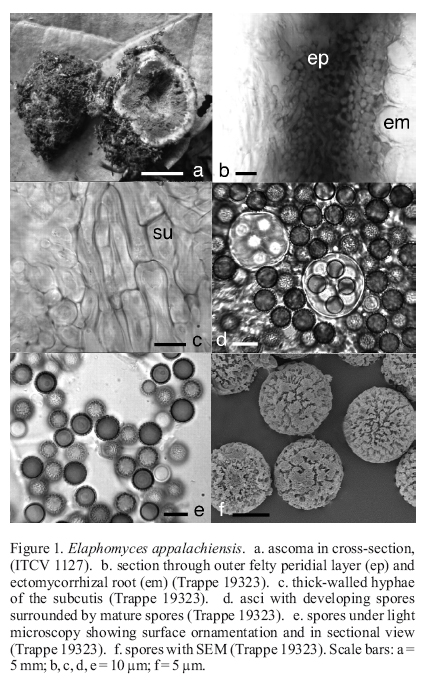
Ascomata up to 5-8 x 6-10 mm, subglobose to irregular, completely embedded in a lilac to red mycelial mat which forms a husk around individual ascomata and incorporating much soil, ectomycorrhizal roots, and debris; mycelium not staining. Peridium ±1 mm broad, outer surface 200-250 μm broad, a felty layer of pale purple hyphae with embedded ectomycorrhizal roots scattered within, the mesocutis and subcutis pale gray to pale purple. Glebal spore mass, powdery, blue-green, with white, spider-web-like hyphae. Odor not recorded. Taste not recorded.
Peridium three-layered, outer felty layer, 200-250 μm broad, of pale purple to violet, septate, loosely interwoven, thin-walled hyphae 3-5 μm broad, embedded within this layer are scattered ectomycorrhizae; mesocutis ±350 μm broad, of hyaline, septate, compact, thin-walled, interwoven hyphae 8-10 μm broad in bundles or strands; subcutis 150-300 μm broad, of pale brown to pale green-brown, septate, thick-walled (±2 μm broad), compact cells, 10-15 μm broad. Gleba constituted by spores and thin-walled, hyaline, septate, somewhat branched, sinuous hyphae, 3-4 μm broad. Asci globose, hyaline, thick-walled (±1 μm broad), 20-25 μm broad, 8-spored, arising from knots of short, irregular hyphae. Spores globose, (8-) 10-13 μm broad (mean = 10.95 μm); walls ±1 μm broad, in KOH pale olive to yellow-brown singly and in mass when mature, ornamentation of compact rods and short ridges or clumps, ±1 μm tall.
Etymology: "appalachiensis" appalach -Appalachia, ensis - pertaining to origin or place, in reference to the locality of the original collection from the Appalachian Mountains of eastern United States of America.
Habit, habitat and season: Known from Iowa south to northern Florida and northeastern Mexico under Carya glabra, C. ovata, Pinus pungens, P. virginiana, Quercus ovata, or Q. prinus; July through early October.
Collections examined: UNITED STATES OF AMERICA, Tennessee, Great Smoky Mountains National Park, Cades Cove, 18 Aug. 1938, A.H. Smith 10334 (holotype FH; isotype MICH); Florida, Alachua Co., Austin Carey Memorial Forest, northeast of Gainesville, July 1983, J. Gibson (FLAS 58700); Leon Co., Apalachicola National Forest, Lost Lake, 17 Jan. 2008, D. Mitchell DMFL08-8 (OSC 130871); Iowa, Lucas Co., Stephens State Forest, 8 Oct. 1999, R. Healy (OSC 130873); same data, White Breast unit, 27 Sept. 2000, R. Healy 820 (OSC 130872); West Virginia, Pendleton Co., Round Knob, 30 Sept. 1996, D. Mitchell Trappe 19323 (OSC 130870); MEXICO, Nuevo León, El Barro, road to paraje del oso, 6 Aug. 2007, J. Quiroga (ITCV 900, OSC 130874, DUKE); Tamaulipas, Las Mulas, Victoria, 18 Aug. 2010, G. Guevara (ITCV 1127), same data J. García (ITCV 18245).
Discussion: Overall the specimens in the holotype collection appear to be somewhat immature. Linder's (1939) specimens were parasitized with Cordyceps intermedia S. Imai which would be in keeping with the young age of specimens. Linder also reports the spores as 7.5-9 μm broad. Our spore measurements of the holotype material are (8-) 1013 μm broad (mean = 10.95 μm) with ornamentation, 7-9 μm broad without ornamentation.
Elaphomyces appalachiensis resembles E. atropurpureus Vitt. in the enveloping, purple-colored mycelium surrounding the sporocarp. The slightly larger-sized spores of E. atropurpureus have a much less dense ornamentation that is readily apparent in surface-view using a light microscope with oil immersion at 1000x.
Elaphomyces verruculosus Castellano sp. nov. Figure 2 Mycobank - 563163.

Ascomata are irregularly subglobose to reniform, up to 22 x 27 mm. Peridial outer surface of low, semi-rounded to angular or elongate warts, up to 500 μm wide and 300 μm tall, rounded to acute or even flattened at apex, contiguous with each other at base. Pale yellow-brown when young then mottled brown and yellow-brown when mature with much pale brown, brown or yellow-brown hyphae, soil and debris covering the warts. Warts often obscured by interwart hyphal structures to make the appearance of the surface nearly smooth or papillate; in section warts are outlined with a layer ±140 μm thick of red-brown to dark red-brown cells underlain with paler yellow-brown to off-white tissue, 300-350 μm thick. Below at the basis of the warts there is an off-white layer sometimes tinged pale gray-blue; subcutis up to 2000 μm broad, off-white to pale gray, sometimes zoned off-white above and pale gray or pale gray-tan below, this is more readily apparent on mature specimens; often a distinct, brown layer, ±100 μm thick at interface with gleba. This layer is contiguous and concolorous with the dissepiments that envaginate into the gleba. Gleba off-white and cottony when immature then spore mass powdery, dark brown to nearly black when mature, numerous pale gray to brown mycelial strands (dissepiments) arising from inner peridial wall and traversing gleba. Odor indistinct, mild. Taste not recorded. Peridium interwart spaces filled with stacked parallel, hyaline, pale yellow to pale yellow-brown, thick-walled (2-3 μm broad), septate hyphae 3-8 μm broad, also at times covering wart apex; Overall the peridium is two-layered, epicutis is a warty layer, ±450 μm broad, with a red to redbrown layer near wart surface then yellow-brown then grading to off-white near wart base, composed of compact, interwoven hyphae 3-8 μm broad, similar in structure to interwart hyphae and sometimes contiguous in organization but not in color; subcutis up to ±2000 μm broad, also similar in structure to outer interwart layer in structure except hyphae are hyaline with amorphous, hyaline granules or pigments interspersed across this layer, these granules scattered are much less dense near gleba and are dark gray or dark redbrown. Gleba constituited by spores and thick-walled (1-1.5 μm broad), hyaline, septate, smooth or slightly encrusted, sinuous, loosely, interwoven hyphae, up to 5 μm broad. Asci globose, hyaline, thick-walled (±2 μm broad), 42-50 μm broad, (2-) 4-8-spored, arising from ascogenous hyphae of clustered knots of hyaline, thick-walled (±1 μm broad), shortsegmented hyphae, ±3 μm broad. Spores globose, (35-) 36-45 (-46) μm broad (mean = 41.1 μm), aborted spores present and are much darker and 23-28 μm broad, mature spores are brown to red-brown to dark red-brown in KOH singly and in mass, ornamentation of tall, hyaline spines or rods when immature (in asci), of dense rods and tufts of rods when mature, 2-3 x 2-3 μm, giving the spore surface a more or less coarse appearance, appearing fuzzy in section, as spores mature they darken and the tufts become darker and more distinct, when mature spores are dark red-brown they appear much more coarse.
Similar to Elaphomyces granulatus Fries of Europe is overall macromorphology with a brown, leathery peridium ornamented with low warts, an inner peridium of off-white to gray-colored tissue and spores ornamented with dense rods and tufts of rods. Elaphomyces granulatus has larger, more coarsely ornamented spores and a uniform, gray inner peridium.
Holotypus: Florida, Walton Co., Eglin Air Force Base, 1 mile west of junction of rd. 403 and rd. 382, 30 m along unnumbered road, 8 Aug. 2007, M. Castellano, Trappe 32038 (OSC 130866).
Etymology: "verruculosus" verrucu – warty, losus – low, in reference to the low warts of the peridial surface.
Habit: habitat and season: Known from Quebec, Canada south to Florida and northeastern México; hypogeous under Picea abies, P. rubens, Pinus elliotii, P. palustris, P. pungens, P. resinosa, P. strobus, P. taeda, P. virginiana, and Tsuga canadensis: June through October.
Other collections examined: UNITED STATES OF AMERICA, Connecticut, no locality, 23 June 1997, J. Floberg, Trappe 19272 (OSC 60116); Georgia, Dougherty Co., Chehaw State Park, 21 Aug. 1977, J. Trappe 5074 (OSC 38947); Louisiana, St. Tammany Parish, Covington, Delta Primate Research Center, 5 June 1976, J. Trappe 4602 (OSC 38724); Massachusetts, Franklin Co., near Conway, Women's Federation Park, Aug. 1986, J. Trappe 9663 (OSC 48877); Mississippi, Harrison Co., DeSoto National Forest, near Brooklyn, 8 Aug. 1985, J. Trappe 8759 (OSC 46775); New York, Thompkins Co., Danby, Michigan Hollow, 1 Oct. 1980, J. Trappe 5971 (OSC 40220); North Carolina, Scotland Co., Scotland Lake, 16 Oct. 1994, M. Castellano, Trappe 11422 (OSC 49671); Vermont, Windsor Co., Ascutney, 30 Aug. 1996, D. Mitchell 119-96 (OSC 59255); Virginia, Augusta Co., near Sherando, 28 Sept. 1984, S. Miller 0807 (RMS); West Virginia, Randolph Co., Bear Heaven, 7 Sept. 1995, D. Mitchell (OSC 59071); Canada, Quebec Province, Montreal, 15 Sept. 1992, F. Marzitelli, Trappe 12707 (OSC 51036); MEXICO, Hidalgo State, La Mojonera, Mpio. Zacualtipan, 23 Sept. 2007, G. Guevara, (ITCV 937, ITCV 938); same data, J. García 17213 (ITCV 17213); Veracruz, Parque Nacional de Perote, Los Gallos, 4 Oct. 1985, J. Trappe 8705 (ITCV, OSC 130867); 1 km south of Tembladeras, 12 Sept. 1985, V. Bandala-Muñoz (ITCV, OSC 130868); México State, Parque Nacional Zoquiapan, along México-Puebla Hwy, 7 Aug. 1972, J. Trappe 3360 (ITCV, OSC 130869).
Discussion: Probably the Elaphomyces granulatus of Trappe & Guzmán (1971) and Cázares et al., (1992) is Elaphomyces verruculosus. Elaphomyces verruculosus resembles E. granulatus of Europe in sporocarp characteristics but differs by its smaller spore size (36-45 μm broad, mean = 41.1 μm with spines 2-3 μm) and less coarse ornamentation compared to E. granulatus and by the uniform gray inner peridium of E. granulatus. Elaphomyces granulatus described by Montecchi & Sarasini, (2000) is actually E. leucocarpus Vitt. characterized by the uniform, white inner peridium and smaller, spiny-reticulate spores they describe and illustrate.
Key to the studied and related Elaphomyces species (spore size includes ornamentation)
1a. Ascoma completely embedded in a lilac to red mycelial mat, spores (8) 10-13 (means 10.95 μm), parasitized by

Acknowledgements
The authors thank DGEST (Dirección General de Educación Superior Tecnológica and ANUIES) for financial support. This work was also partially supported by NSF grant 0641297. We appreciate the field assistance of Greg Bonito, Efrén Cázares, Rosanne Healy, and Rytas Vilgalys. We appreciate the loan of collections from the following herbarium: DUKE, FH, FLAS, ITCV, and RMS.
References
Castellano, M.A., E. Cázares, B. Fondrick, T. Dreisbach, 2003. Handbook to additional fungal species of special concern in the Northwest Forest Plan. Gen. Tech. Rep. PNW-GTR-572. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Station. [ Links ]
Castellano, M.A., J.E. Smith, T. O'Dell, E. Cázares, 1999. Handbook to strategy 1 fungal taxa from the Northwest Forest Plan. Gen. Tech. Rep. PNW-GTR-476. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Station. [ Links ]
Castellano, M.A., J.M. Trappe, Z. Maser, C. Maser, 1989. Keys to spores of the genera of hypogeous fungi of North Temperate forests with special reference to animal mycophagy. Mad River Press, Eureka. [ Links ]
Cázares, E., J. García, J. Castillo, J.M. Trappe, 1992. Hypogeous fungi from northern México. Mycologia 84: 341-359. [ Links ]
Cázares, E., G. Guevara, J. García, J.M. Trappe, 2008. Melanogaster minysporus sp. nov. un nuevo miembro secuestrado de los Boletales de México. Revista Mexicana de Micología 28: 67-69. [ Links ]
Guevara, G., Bonito, E. Cázares, J. Rodríguez, R. Vilgalys, J.M. Trappe, 2008a. Tuber regimontanum, a new species of truffle from México. Revista Mexicana de Micología 26: 17-20. [ Links ]
Guevara, G., M.A. Castellano, J. García, E. Cázares, J.M. Trappe, 2008b. Hysterangium (Hysterangiales, Hysterangiaceae) from Northern México. Revista Mexicana de Micología 28: 95–100 [ Links ]
Guevara, G., B. Stielow, H. Tamm, E. Cázares-González, M. Göker, 2011. Genea mexicana, sp. nov., and Geopora tolucana, sp. nov., new hypogeous Pyronemataceae from México, and the taxonomy of Geopora reevaluated. Mycological Progress (DOI10.1007/s11557-011-0781-y). [ Links ]
Guzmán, G. 1971. Notas sobre los géneros Radiigera y Mesphelliopsis en México. Boletín de la Sociedad Mexicana de Micología 5: 7-11. [ Links ]
Guzmán, G. 1983. Los hongos de la Península de Yucatán II. Biótica 8: 71-100. [ Links ]
Healy, R., G. Bonito, G. Guevara-Guerrero, 2009. The truffle genus Pachyphloeus in the USA and Mexico: phylogenetic analysis and a new species. Mycotaxon 107:61–71. [ Links ]
Hosford, D.R., J.M. Trappe, 1980. Taxonomic studies on the genus Rhizopogon. II. Notes and new records of species from Mexico and Caribbean countries. Boletín de la Sociedad Mexicana de Micología. 14: 3–15. [ Links ]
Index Herbariorum 2011, http://sciweb.nybg.org/science2/Index Herbariorum.asp [accessed 10 June 2011] [ Links ].
Maser, C., A.W. Claridge, J.M. Trappe, 2008. Trees, truffles, and beasts, how forest function. New Jersey, USA, Rutgers University Press. [ Links ]
Montecchi, A., M. Sarasini, 2000. Funghi ipogei d'Europa. Associazione Micologica Bresadola, Fondazione Centro Studi Micologici. Vicenza. [ Links ]
Trappe, J.M., G. Guzmán, 1971. Notes on some hypogeous fungi from México. Mycologia 63: 317-345. [ Links ]
Trappe, J.M., G. Guzmán, C. Vazquez-Salinas, 1979. Observaciones sobre la identificación, distribución y uso de los hongos del género Elaphomyces en México. Boletín de la Sociedad Mexicana de Micología 13: 145-150. [ Links ]
Trappe, J.M., R. Molina, D.L. Luoma, E. Cázares, D. Pilz, J.E. Smith, M.A. Castellano, L. Miller, M. J. Trappe, 2009. Diversity, ecology and conservation of the truffle fungi in forests of the pacific northwest. US Dept. of Agriculture, Forest Service General Technical Report, PNW-GTR-772. [ Links ]














