Introduction
Given the deterioration of fisheries worldwide, aquaculture has been the alternative with the highest economic viability to solve the demand for food (FAO 2019). In Mexico, aquaculture has made significant progress in recent decades, particularly in the northwest region, where biological productivity, which includes fish, mollusks, and crustaceans, is the highest in the country. Oysters of the genus Crassostrea and the whiteleg shrimp, Litopenaeus vannamei (Boone, 1931), represent one of the highest marine aquaculture values in Mexico (SAGARPA 2017). Increased national and export marketing of products of aquaculture origin has favored the introduction, spread, and transmission of diseases (OIE 2019). In oyster farms Perkinsus spp. have threatened oyster productions (Cáceres-Martínez et al. 2008, Enríquez-Espinoza et al. 2010). Shrimp farms have been severely affected by mortality episodes associated with diseases caused by bacteria belonging to the genus Vibrio (Soto-Rodriguez et al. 2010) and the white spot syndrome virus (WSSV) (Hasson et al. 2006). Adequate disease control in aquatic farming systems is needed to stop the spread of infectious pathogens. Disease monitoring in terms of surveillance and rapid diagnosis is an integral part of the aquaculture process. It allows for appropriate action to be readily taken when pathogens are detected and diseases are diagnosed before they become a significant problem for the farmer.
The design and development of methods for identifying pathogens in farmed aquatic organisms has progressed. Molecular detection techniques have been developed over the last decade, and identification of microorganisms is chiefly based on the analysis of nucleic acids. The advantages of these techniques include specificity, sensitivity, speed, and automation (Palomino-Camargo et al. 2014). Loop-mediated isothermal amplification (LAMP) is a molecular technique that amplifies DNA with high specificity, efficiency, and rapidity under isothermal conditions without requiring expensive specialized equipment (Notomi et al. 2000). The amplification requires 2 outer primers (F3 and B3), 2 inner primers (forward inner primer, FIP, and backward inner primer, BIP), and 2 loop primers (forward loop, LF, and backward loop, LB) (Notomi et al. 2000). These primers are complementary to 6 regions of the DNA sequence, ensuring very high specificity (Notomi et al. 2000, Nagamine et al. 2002). The primers bind laterally to different sites using strand-displacing Bst DNA polymerase, providing high amplification efficiency, with DNA being amplified to 109-1010 copies in 15-60 min (Notomi et al. 2000, Nagamine et al. 2002, Tomita et al. 2008, Mori and Notomi 2009, Li and Macdonald 2015). This method does not require a denatured DNA template (Nagamine et al. 2001), which allows users to avoid complicated DNA refinement protocols, and has shown high tolerance to inhibitory substances present in biological samples (Boehme et al. 2007, Kaneko et al. 2007, Mori and Notomi 2009, Francois et al. 2011). The amplified products can be detected by gel electrophoresis, a turbidimeter, lateral flow dipstick, or color or fluorescent indicators (Mori et al. 2001, Iwamoto et al. 2003, Mori et al. 2004, Tomita et al. 2008, Goto et al. 2009).
LAMP has been used to detect shellfish pathogens in many countries (Kono et al. 2004, Mekata et al. 2006, Yamazaki et al. 2008, Jaroenram et al. 2009, Mekata et al. 2009, Chen and Ge 2010, Arunrut et al. 2011, Maralit et al. 2012, Xu et al. 2012, Feng et al. 2013, Kongrueng et al. 2015). Many reports show inconsistencies with LAMP protocols for detecting pathogens from different geographic locations. Gene sequences of infectious agents may vary depending on the geographic region as a result of a set of variables that favor dispersion and potential modifications in their genotype. It is necessary to choose specific primers and probes for genomes from each location to avoid incorrect diagnoses. Here, we propose identifying potentially dangerous pathogens specific to Mexico in L. vannamei and Crassostrea spp. using LAMP. LAMP technology has great potential as a technique for the rapid, sensitive, and specific detection of pathogens that can be used regularly in farms or laboratories because it does not need expensive machines or highly trained personnel.
Materials and methods
Sample collection and DNA extraction
Tissue of L. vannamei infected with WSSV and Vibrio parahaemolyticus, and tissues of the Cortez oyster (Crassostrea corteziensis) and the Pacific oyster (Crassostrea gigas) infected with Perkinsus spp. were obtained from farms in northwest Mexico. We selected tissues following the manual of the World Organization for Animal Health (OIE 2019). We also used uninfected organisms as negative controls. DNA was extracted from tissues using the organic extraction method, as described in Sambrook et al. (1989), and it was resuspended by adding TE Buffer (10 mM, pH 8.0). DNA purity and concentration were estimated using the Nanodrop 1000 spectrophotometer (Thermo Fisher Scientific; MA, USA). DNA integrity was visualized by agarose gel electrophoresis (1.5%) using the UVP Biodoc-It 2 imaging system (Analytik Jena; Jena, Germany) and the fluorescent dye GelRed (Biotium; Fremont, CA).
Positive samples
DNA from infected and uninfected specimens was extracted and used as a template for end-point PCR amplification to obtain positive and negative control tissues using the LAMP outer primers (F3 and B3). PCR amplicon sizes were 208 bp for the internal transcribed spacer 2 (ITS-2) (Perkinsus spp.), 213 bp for orf191 (WSSV), 231 bp for orf36 (WSSV), 229 bp for thermolabile hemolysin (tlh) (V. parahaemolyticus), and 183 for tox-R (V. parahaemolyticus). We cleaned up the PCR products for sequencing with Genewiz (South Plainfield, NJ). The obtained sequence fragments were analyzed using the NCBI BLAST Sequence Analysis Tool (https://blast.ncbi.nlm.nih.gov/Blast.cgi, 20 November 2021) to corroborate the identity of the pathogen.
LAMP reaction conditions
For LAMP, we determined the candidate target genes in the first-round analysis of many sets of primers. We then chose those that successfully amplified with Mexican samples (Table 1) with high repeatability, efficiency, and specificity. Primers were synthesized by T4 Oligo (Irapuato, Mexico). The LAMP assay was carried out in a total of 25-µL volume containing 1 µL (40 µM) of each of the inner primers (FIP and BIP), 1 µL (5 µM) of each of the outer primers (F3 and B3), 1 µL (20 µM) of each of the loop primers (LF and LB), 4 µL (0.6 mM of each nucleotide) dNTP mix (Thermo Fisher Scientific), 1 µL (5 M) Betaine (Sigma-Aldrich), 1.5 µL (100 mM) MgSO4 (New England Biolabs), 1 µL (8 U·µL-1) of Bst DNA polymerase large fragment (New England Biolabs), 2.5 µL of ThermoPol buffer 10× (New England Biolabs), 1.0 µL of target DNA, and 8 µL of Milli-Q water. The tox-R primers (Chen and Ge 2010) have only one loop primer, and they were added as described for the concentration above, adding 1 µL more of Milli-Q water. Incubation was then carried out at 61, 62, 63, 64, and 65 ºC for 60 min, and the reaction was terminated by heating at 90 ºC for 2 min. LAMP reactions were performed using the BioRad T100 Thermal Cycler (CA, USA). The products (10 µL) were electrophoresed on 2% agarose gels to determine the optimal conditions. When the LAMP products are visualized using agarose gel electrophoresis, many bands of different sizes, up to the loading well, are documented. The resulting amplicon mixtures were confirmed with 10 µL of the products by fluorescence, using 1 µL of SYBR safe (Thermo Fisher Scientific; 1:500 dilution in Milli-Q water), and they were added to the solution after the reaction was completed. Gel and fluorescent tubes were documented using the UVP Biodoc-It 2 imaging system (Analytik Jena). DNA extracts from the uninfected shrimp and oyster samples were included as negative controls.
Table 1 Sequences of LAMP primers.
| Pathogen | Genes | Primer | Sequence (5′3′) | Source |
| Perkinsus spp. | ITS2 (FJ841985) | F3 | TGGATTTTGGTATTTCAAAACGA | Feng et al. (2013) |
| B3 | ATGTTTGTATCGGGAAGAAGAG | |||
| FIP | AGTGCTTATCGCACTTCGCTCAAACTCTCAACGATGGATGC | |||
| BIP | GATTTGCAGAATTCCGTGAACCAGCACTGATATGTATGTACAAAGAGG | |||
| LF | TCCTTCATCGATTCTCGAGC | |||
| LB | TCAACGCATACTGCACAAAG | |||
| WSSV | orf191 (AF332093) | F3 | AAAGTCTCATTTTAACAAGAGGA | Jaroenram et al. (2009) |
| B3 | ATTGGTCCAGTCTCAGCC | |||
| FIP | CCATCTCTCTGAAGCGAGGAAAATTTTAATTTCTTTGCTCGAGGCC | |||
| BIP | TGCTCTAGAGAATGCAGTACCTCTTTTGCGTAGTTCTTGCACGATT | |||
| LF | TCTCCAGCCGGAGCCAAGT | |||
| LB | ACAGAACCATAACAATGGTGAATGG | |||
| WSSV | orf36 (AF369029) | F3 | AAACACCGGATGGGCTAA | Mekata et al. (2009) |
| B3 | CAAGGCAATACAGAATGCG | |||
| FIP | TCCTGTCTTCAGGGAATACATATGCTCAGGGAAGAAATAGACCATG | |||
| BIP | GGACCCAAATCGAAATATAAGGCCTATGTTGCCCAAGATCCAC | |||
| LF | GTTAAGAATGATGCATCTAGTGCGA | |||
| LB | TGGAACAAAAGATGCTGCTCA | |||
| Vibrio parahaemolyticus | tlh (M36437) | F3 | AGCTACTCGAAAGATGATCC | Yamazaki et al. (2008) |
| B3 | GGTTGTATGAGAAGCGATTG | |||
| FIP | ATGTTTTTAAATGAAACGGAGCTCCGGCAAAAAACGAAGATGGT | |||
| BIP | ACGTCGCAAAACGTTATCCGGCGAAGAACGTAATGTCTG | |||
| LF | ACCAGTAGCCGTCAATG | |||
| LB | TTAGATTTGGCGAACGAGA | |||
| Vibrio parahaemolyticus | tox-R (L11929) | F3 | TTGGATTCCACGCGTTAT | Chen and Ge. (2010) |
| B3 | CGTTCAATGCACTGCTCA | |||
| FIP | TGAGATTCCGCAGGGTTTGTAATTATTTTTGGCACTATTACTACCG | |||
| BIP | GTTCCGTCAGATTGGTGAGTATCTAGAAGGCAACCAGTTGTT | |||
| Loop | AGAACGTACCAGTGATGACACC |
WSSV, white spot syndrome virus; FIP, forward inner primer; BIP, backward inner primer; LF, forward loop primer; LB, backward loop primer.
The LAMP temperature, sensitivity, reaction time, and specificity were optimized for each pathogen sample. To evaluate sensitivity, 10-fold serial dilutions of the total genomic DNA, which was extracted from infected tissues and isolates, were used as templates to select the best dilution to work with, and LAMP was carried out for a predetermined time (63 min). To determine optimal amplification time, LAMP reactions were carried out at the temperature and DNA concentration previously chosen for each pathogen for 10, 20, 30, 40, 50, and 60 min; the time in the negative control was performed at 60 min. LAMP specificity was examined using the template DNA for other pathogens and uninfected samples. For identifying cross-reactivity with the ITS2 gene for Perkinsus spp., we used the toxic dinoflagellates Gymnodinium catenatum, Alexandrium tamiyavanichii, and Alexandrium catenella. For diagnosis of specificity for tlh and tox-R for V. parahaemolyticus, we used Vibrio campbelli, Photobacterium damselae, Pseudomonas aeruginosa, Providencia rettgeri, and Acinetobacter baumannii. For the diagnosis of specificity for orf36 and orf191 to evaluate WSSV, we used the shrimp hypodermal and hematopoietic necrosis virus, oyster herpes virus, and squash leaf curl virus. DNA extraction was done as previously mentioned, and LAMP was carried out with the optimal conditions obtained above.
Results
Representative PCR products for the positive samples of tissues infected with the pathogens under study can be observed in Figure 1. These results were obtained with primers F3 and B3. Figures 2-6 show the gel electrophoresis and fluorescent tubes from the LAMP assays for the tested pathogens. The LAMP assay for detecting Perkinsus spp. in Crassostrea spp. tissues using the primers from the ITS2 gene region showed optimal amplification conditions at every probed temperature (61 to 65 ºC). Results were confirmed by the tubes with fluorescence (Fig. 2a). The assay for examining LAMP sensitivity showed that optimal DNA concentrations were 3.6 to 36.0 ng (Fig. 2b). The LAMP assay for determining optimal time started at 30 min and was kept on for 60 min (Fig. 2c). To detect V. parahaemolyticus in L. vannamei, we used the tlh (Fig. 3) and tox-R (Fig. 4) genes. With the tlh primers, the temperature optimization probe showed that amplification was performed at every temperature tested from 61 to 65 ºC (Fig. 3a). Figure 3b shows results from the LAMP sensitivity assay, and amplification was performed in all dilutions tested from 2.5 pg to 250.0 ng of total DNA. Optimization of the LAMP reaction time showed that amplification started at 40 min and continued for up to 60 min (Fig. 3c). The temperature optimization of the LAMP reaction with the tox-R primers showed that amplification was performed successfully from 61 to 64 ºC (Fig. 4a). LAMP sensitivity showed amplification at 25.0 to 250.0 ng of DNA (Fig. 4b), and time optimization started at 40 min and continued for up to 60 min (Fig. 4c). To detect WSSV in L. vannamei, we chose 2 sets of primers, orf36 (Fig. 5) and orf191 (Fig. 6). With the orf36 primer, optimization of LAMP assay temperature showed that amplification was performed at 61 to 65 ºC (Fig. 5a), sensitivity showed that the DNA amplified only at a concentration of 42.0 ng (Fig. 5b), and optimal amplification time was 30 to 60 min (Fig. 5c). With orf191, the optimal temperature showed that amplification occurred at all temperatures probed from 61 to 65 ºC (Fig. 6a), sensitivity showed that LAMP amplification was performed from 42.0 pg to 42.0 ng of DNA (Fig. 6b), and optimization of the LAMP reaction time showed that amplification started at 30 min and it kept on for up to 60 min (Fig. 6c).
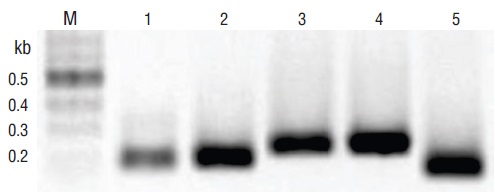
Figure 1 Pathogens identified in tissues of infected Crassostrea spp. and Litopenaeus vannamei using end-point PCR amplification of DNA with the particular external primers for loop-mediated isothermal amplification (F3 and B3). Lane M, marker (2 log DNA ladder; kb, kilobases); lane 1, Perkinsus spp. (ITS2); lane 2, white spot syndrome virus (WSSV) (orf191); lane 3, WSSV (orf36); lane 4, Vibrio parahaemolyticus (thermolabile hemolysin [tlh]); lane 5, V. parahaemolyticus (tox-R). PCR products were visualized on 2% agarose gel electrophoresis.
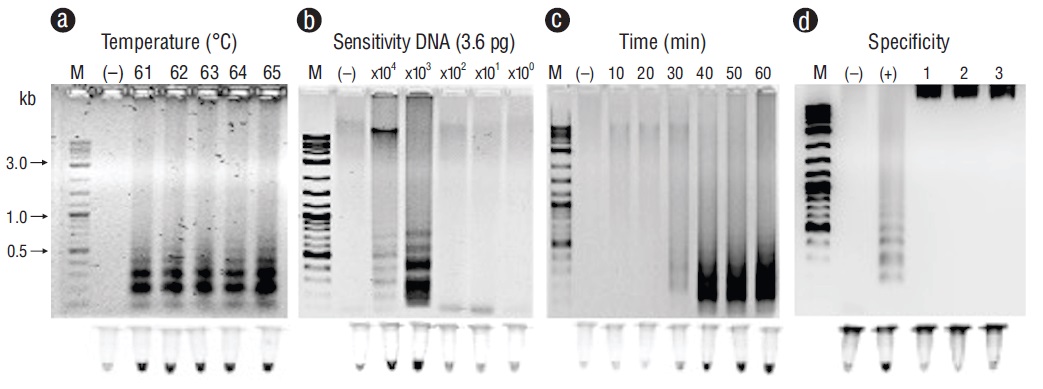
Figure 2 Optimization for the ITS2 gene with loop-mediated isothermal amplification optimization for detecting Perkinsus spp. in tissues of Crassostrea spp. (a) Temperature (ºC). (b) Sensitivity (pg). (c) Time of reaction (min). (d) Specificity: lane (+), Perkinsus marinus; lane 1, Gymnodinium catenatum; lane 2, Alexandrium tamiyavanichii; lane 3, Alexandrium catenella. Lane M, marker (2 log DNA ladder; kb, kilobases); lane (-), negative control (DNA from uninfected tissue). Products were visualized on 2% agarose gel electrophoresis and fluorescent tubes.
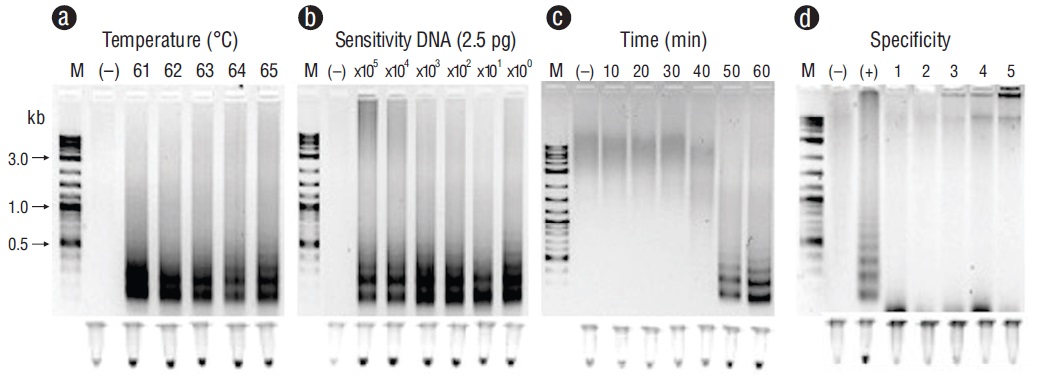
Figure 3 Optimization for the thermolabile hemolysin (tlh) gene with loop-mediated isothermal amplification for detecting Vibrio parahaemolyticus in tissues of Litopenaeus vannamei. (a) Temperature (ºC). (b) Sensitivity (pg). (c) Time of reaction (min). (d) Specificity: lane (+), V. parahaemolyticus; lane 1, Vibrio campbelli; lane 2, Photobacterium damselae; lane 3, Pseudomonas aeruginosa; lane 4, Providencia rettgeri; lane 5, Acinetobacter baumannii. Lane M, marker (2 log DNA ladder; kb, kilobases); lane (-), negative control (DNA from uninfected tissue). Products were visualized on 2% agarose gel electrophoresis and fluorescent tubes.
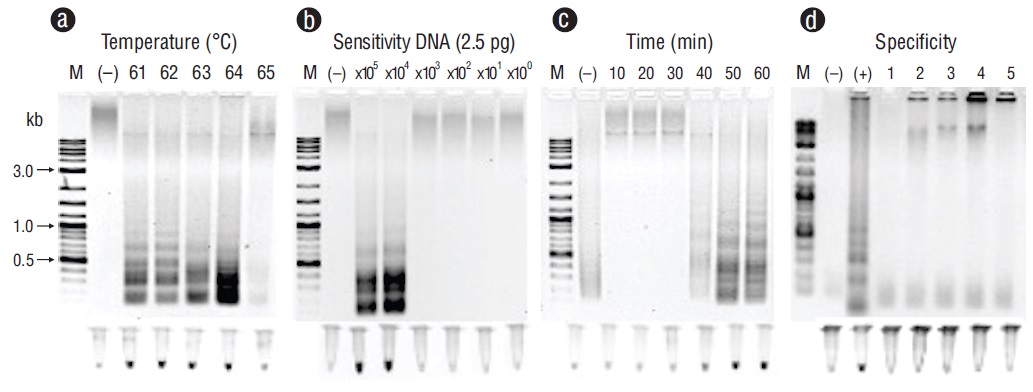
Figure 4 Optimization for the tox-R gene with loop-mediated isothermal amplification for detecting Vibrio parahaemolyticus in tissues of Litopenaeus vannamei. (a) Temperature (ºC). (b) Sensitivity (pg). (c) Time of reaction (min). (d) Specificity: lane (+), V. parahaemolyticus; lane 1,Vibrio campbelli; lane 2, Photobacterium damselae; lane 3, Pseudomonas aeruginosa; lane 4, Providencia rettgeri; lane 5, Acinetobacter baumannii. Lane M, marker (2 log DNA ladder; kb, kilobases); lane (-), negative control (DNA from uninfected tissue). Products were visualized on 2% agarose gel electrophoresis and fluorescent tubes.
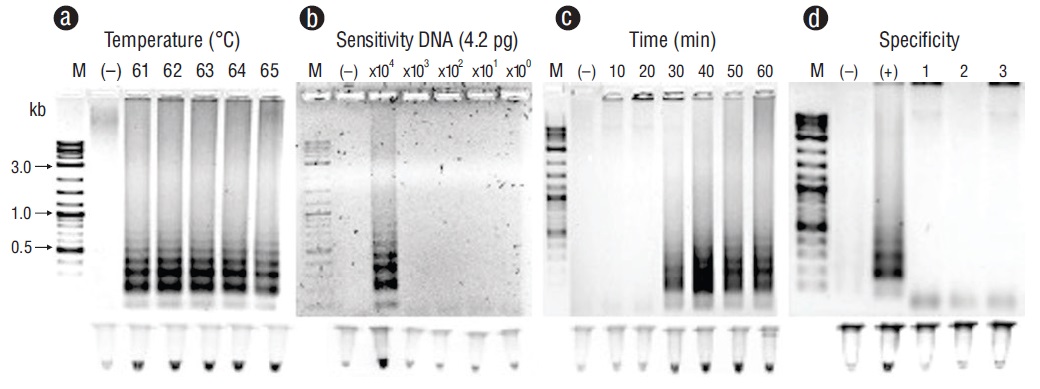
Figure 5 Optimization for the orf36 gene with loop-mediated isothermal amplification for detecting white spot syndrome virus (WSSV) in tissues of Litopenaeus vannamei. (a) Temperature (ºC). (b) Sensitivity (pg). (c) Time of reaction (min). (d) Specificity: lane (+), WSSV; lane 1, shrimp hypodermal and hematopoietic necrosis virus; lane 2, oyster herpes virus; lane 3, squash leaf curl virus. Lane M, marker (2 log DNA ladder; kb, kilobases); lane (-), negative control (DNA from uninfected tissue). Products were visualized on 2% agarose gel electrophoresis and fluorescent tubes.
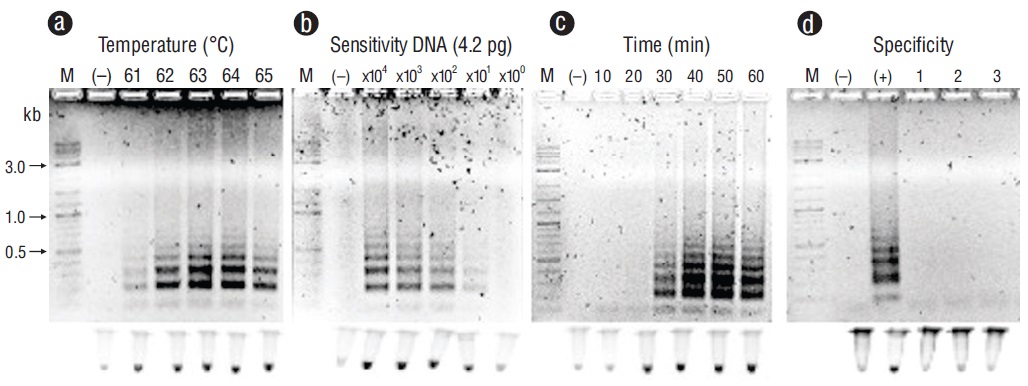
Figure 6 Optimization for the orf191 gene with loop-mediated isothermal amplification for detecting white spot syndrome virus (WSSV) in tissues of Litopenaeus vannamei. (a) Temperature (ºC). (b) Sensitivity (pg). (c) Time of reaction (min). (d) Specificity: lane (+), WSSV; lane 1, shrimp hypodermal and hematopoietic necrosis virus; lane 2, oyster herpes virus; lane 3, squash leaf curl virus. Lane M, marker (2 log DNA ladder; kb, kilobases); lane (-), negative control (DNA from uninfected tissue). Products were visualized on 2% agarose gel electrophoresis and fluorescent tubes.
We tested the cross-reaction with other viruses and bacterial templates to determine LAMP method specificity (Figs. 2d, 3d, 4d, 5d, 6d). No positive results for LAMP were found for any of the samples, which indicates that the LAMP method showed high specificity for the probed shellfish pathogens.
Discussion
In this study, we evaluated the use of LAMP to detect pathogens in tissues of shrimp and oysters cultured in Mexico. We described a LAMP diagnostic protocol for Perkinsus spp., WSSV, and V. parahaemolyticus. These pathogens cause serious global oyster and shrimp production losses, which result in severe economic injuries. The World Organization for Animal Health considers WSSV to be the most common virus in shrimp cultures; furthermore, another type of pathogen that gravely affects shrimp cultures is the bacterium V. parahaemolyticus, which acts as an opportunistic agent, causing massive losses (OIE 2019). The protozoa Perkinsus marinus and Perkinsus olsenii are organisms that affect oyster cultures, primarily Crassostrea spp. (OIE 2019). Diseases caused by these pathogens must be declared to the World Organization for Animal Health as soon as they are detected.
Accurate detection of shellfish pathogens is a crucial step in the process of disease management. Numerous detection methods have been developed to monitor and control these pathogens in cultures. Techniques based on DNA amplification have sensitivity and specificity advantages that make them valuable pathogen detection tools. LAMP is a sensitive method that can amplify a few DNA copies in less than an hour under isothermal conditions (Notomi et al. 2000, Mori et al. 2001, Nagamine et al. 2002, Mori and Notomi 2009). It has been used to detect shellfish pathogens in many countries; however, many reports show inconsistencies in LAMP protocols for detecting pathogens from different geographic locations, as there is a set of variables that favor dispersion and potential modifications in pathogen genotypes. We tested many different sets of primers for each pathogen, and we developed the protocol using the primers and assay conditions for LAMP reactions that were most suitable for pathogens in Mexican aquaculture systems. Our assay could also be used in wild-type organisms, different development stages, and other shellfish species when the pathogen is present.
The LAMP protocol we describe here is a sensitive, specific, and rapid method for detecting shellfish pathogens. Many reports have evaluated LAMP and argue that it has many advantages over other molecular techniques. LAMP is a single-step procedure that requires 4 to 6 primers that bind laterally to different sites using strand-displacement Bst DNA polymerase, permitting extremely specific amplification under isothermal conditions (Notomi et al. 2000). It does not require a denatured DNA template, avoiding time-consuming DNA extraction methods, and has low susceptibility to inhibitory compounds; the results can be visualized by the naked eye with color or fluorescent DNA dyes. As a result, it allows the use of simple and cost-effective reaction equipment and simplified protocols (Notomi et al. 2000, Nagamine et al. 2001, Kaneko et al. 2007, Tomita et al. 2008, Goto et al. 2009, Mori and Notomi 2009, Francois et al. 2011, Li and Macdonald 2015, Tanner et al. 2015).
The protocol that we show in this work is a basic LAMP method, designed to be carried out mainly in the laboratory. LAMP has proven to be broadly functional for on-site detection of pathogens due to its adaptability to varied detection approaches and settings, including many commercial portable kits, for which the workflow does not require highly qualified personnel (Kiatpathomchai et al. 2008, Jaroenram et al. 2009, Khunthong et al. 2013, Mori et al. 2013, Tomlinson 2013, Dhama et al. 2014, Notomi et al. 2015, Besuschio et al. 2017, Khan et al. 2017, Wong et al. 2018, Nzelu et al. 2019, Becherer 2020). Rapid and accurate detection of latent infections can provide farmers with reliable information on the infection status for early prevention and control of disease outbreaks. As efforts to raise production levels continue and research interest in aquaculture grows, the use of biosurveillance methods is also expanding. Research facilities can then use the technology, universities, government agencies, and shellfish farms to become a staple for detecting pathogens in aquaculture in Mexico and other countries.











 texto en
texto en 


