Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Salud mental
Print version ISSN 0185-3325
Salud Ment vol.33 n.3 México May./Jun. 2010
Actualización por temas
Endomorphin peptides: pharmacological and functional implications of these opioid peptides in the brain of mammals. Part two
Las endorfinas: implicaciones farmacológicas y funcionales de estos péptidos opioides en el cerebro de los mamíferos. Segunda parte
Philippe Leff Gelman,1 Norma Estela González Herrera,2 Maura Epifanía Matus Ortega,1 Enrique Beceril Villanueva,3 Carlos Téllez Santillán,3 Alberto Salazar Juárez,1 Benito Antón Palma1
1 Laboratorio de Neurobiología Molecular y Neuroquímica de Adicciones. Subdirección de Investigaciones Clínicas, Instituto Nacional de Psiquiatría, Ramón de la Fuente Muñiz.
2 Laboratorio de Oncología Molecular. Sección de Posgrado e Investigación. Escuela Superior de Medicina, Instituto Politécnico Nacional.
3 Laboratorio de Psicoinmunología. Dirección de Neurociencias Instituto Nacional de Psiquiatría Ramón de la Fuente Muñiz.
*Corresponding author.
Philippe Leff Gelman.
Laboratorio de Neurobiología Molecular y Neuroquímica de Adicciones.
Subdirección de Investigaciones Clínicas,
Instituto Nacional de Psiquiatría Ramón de la Fuente Muñiz.
Calzada México–Xochimilco 101,
San Lorenzo Huipulco,
Tlalpan, 14370,
México D F,
email: pleff@imp.edu.mx
Abstract
Endomorphin–1 (EM1) and Endomorphin–2 (EM2) represent the two endogenous C–terminal amide tetrapeptides shown to display a high binding affinity and selectivity for the µ–opioid receptor as reported previously (see previous paper, Part I). Endomorphins injected into the VTA were shown to enhance the development of behavioral sensitization responses to amphetamine (AMPH), besides of inducing an increase of locomotion (horizontal) activity in animals. These studies showed that EM2 was significantly more potent than EM1 in modulating the increased opioid–mediated ambulatory responses by altering the dopamine (DA) projecting system in the globus pallidus in tested animals. Several transmission systems (e.g., GABA) have been shown to participate in the endormorphin–induced locomotor responses. EM1 injected into the VTA produced potent rewarding effects in rodents, similar to the rewarding responses produced by distinct opiate compounds. The opioid rewarding responses induced by EM1–2 were shown to be mediated via the activation of both GABAergic and the dopamine (VTA–NAc–PFCx) transmission systems in the brain. Moreover, EM1–2 peptides injected into the VTA, but not in the NAc, produced similar related–rewarding responses induced by low doses of morphine. However, ICV administration of EM1 was shown to enhance a significant conditioned–place preference (CPP); whereas EM2 displayed a place aversion in tested animals.
With regard to stress–related behaviors and physiological responses in mammals, endomorphin peptides have been proposed to modulate the HPA axis function via activation of the NTS–projecting neural system impinging on hypothalamic neurons, and/or via activation of the PAG (ventrolateral area) mediating analgesic responses–induced by stress. EM1–2 peptides have been shown to induce mood–related behaviors. For instance, administration of EM1 induced an increased anxiolytic response in mice when tested in elevated plus maze paradigms, results that showed that the µ–opioid receptor modulates mood–related responses in animals and humans, as well. Interesting enough is the recent observation that EM1–2 peptides may induce antidepressant–like behaviors in animals models of stress and depression, whereby EM1–2 peptides have been shown to up–regulate in a dose–dependent manner the neuronal expression of the BDNF mRNA in rat limbic areas involved in stress and depressive–like behaviors. Thus, these studies led to the proposition that endomorphin peptides may play crucial roles in psychiatric disorders (e.g., depression, schizophrenia). Furthermore, over the past years, it has been shown that µ–opioid receptor agonists (e.g., morphine, DAMGO; morphine–6β–glucuronide) displayed potent orexigenic activities in the CNS of mammals, similar to that displayed by EM1–2 peptides, whose dose–dependent orexigenic activity appears to be mediated by the endogenous opioid peptide, Dynorphin A, acting on its cognate κ–opioid receptor at the hypothalamus.
Extensive studies revealed the activity of the EOS (e.g., β–endorphin) on the regulation of gonadal hormones and sexually–induced behaviors (e.g., lordosis) in female rats. β–endorphin or morphiceptin have been shown to facilitate lordosis behaviors in estrogen– and/or estrogen/progesterone primed rats, whereas EM1–2 peptides injected into third ventricle or into the diagonal band (DB) produced dose– and time–dependent, naloxone–reversible lordosis responses in female rats. These results posit that EM1–2 peptides produce their sexual behaviors and mating responses via modulating the cell release of LHRH and modulating GABA transmission system in the brain. Endomorphins have been shown to impair short– and long–term memory processing in mice when exposed to different learning paradigms. These opioid mediated effects appear to be regulated through the interaction of both cholinergic and dopaminergic transmissions in the brain. In addition, endomorphins have been shown to modulate cardiovascular and respiratory bioactivities, acting on several rostrocaudal areas of the CNS of mammals. Administration of EM1–2 peptides induced a significant reduction of heart rate and blood pressure in normotensive and hypertensive rats, via regulation of GABA and glutamate transmission systems. Although the exact endogenous mechanisms by which EM1–2 peptides produce their vasoactive responses are still unclear, several studies suggested that the peptide activity depends on the synthesis and release of nitric oxide (NO) from endothelial cells enhanced by activation of µ–opioid receptors. Studies on respiratory function showed that EM1–2 peptides attenuate and produce significant respiratory depression in tested animals. Finally, EM1–2 peptides have been shown to induce important inhibitory gastrointestinal effects via the activation of µ–opioid receptors localized in myenteric–plexus neurons that innervate smooth–muscle cells producing a dose–dependent– and CTOP–reversible inhibition of electrically–induced twitch ileum contractions, probably mediated through a reduced release response of several peptide and non–peptide transmitters.
Key words: Endomorphins, physiology, locomotor sensitization responses, opioid reward, stress, HPA axis, sex, feeding, cardiovascular, respiratory, anxiolytic, social defeat.
Resumen
La endomorfina–1 (EM1) y la endomorfina–2 (EM2) son dos péptidos bioactivos que poseen la más alta afinidad de unión selectiva por el receptor opioide µ en comparación con la unión de distintos ligandos agonistas a este subtipo de receptor opioide (véase resumen y texto del capítulo anterior, parte I). Estudios farmacológicos y conductuales han demostrado que la inyección de las EM1–2 en el área ventrotegmental (AVT) genera respuestas conductuales de sensibilización locomotora a la anfetamina (AMPH), además de incrementar la actividad locomotora de tipo horizontal en los roedores tratados. Estos estudios mostraron que la EM2 fue significativamente más potente que la EM1 en inducir las respuestas locomotoras detectadas, mediadas a través de la alteración de la actividad sináptica de dopamina (DA) y en el globus pallidus de los animales tratados. Asimismo, estudios fármaco–conductuales similares demostraron que otros sistemas de transmisión participan conjuntamente con el sistema dopaminérgico en la generación de los efectos locomotores inducidos por las EM1–2, como es el caso del sistema gabaérgico (GABA). Más aún, la inyección de EM1 en la región AVT del cerebro de roedores mostró generar respuestas potentes de recompensa placentera, similares a las reportadas por distintos alcaloides opiáceos de alto potencial adictivo, posterior a su administración sistémica. Más aún, la inyección de endomorfinas en la región AVT del cerebro del roedor, mas no en el núcleo accumbens (NAc), mostró generar respuestas de recompensa paralela a la generada posteriormente a la administración de dosis bajas de morfina.
En línea con los efectos farmacológicos inducidos por las EM1–2, estudios fármaco–conductuales demostraron que la administración ICV de la EM1 fue capaz de generar respuestas de preferencia de lugar en roedores tratados CPP, por sus siglas en inglés, conditioned place preference, en tanto que la administración de EM2 generó respuestas opuestas, esto es, respuestas de aversión al lugar. Estudios conductuales relacionados con el fenómeno de estrés mostraron que las EM1–2 son capaces de modular la actividad funcional del eje HHA (eje hipotálamo/hipófisis/glándula adrenal) a través de la activación del sistema de proyección neuronal del tracto solitario (NTS, por sus siglas en inglés), al hipotálamo y/o a través de la activación del área ventrolateral de la sustancia gris periacueductal (PAG, por sus siglas en inglés); componente importante del sistema opioide endógeno, que median respuestas analgésicas (antinociceptivas) inducidas por estímulos estresantes. Asimismo, la administración de endomorfinas (v.g., EM1) mostró generar incrementos de conductas de naturaleza ansiolítica en ratones expuestos a paradigmas experimentales de generación de conductas estresantes (v.g., laberinto elevado). Estos estudios sugieren que la generación de conductas de estrés–emocional inducidas por las endomorfinas es mediada a través de la activación del receptor opioide µ en neuronas del hipotálamo responsables de regular la secreción de factores liberadores de distintas hormonas hipofisiarias (v.g., CRH, LHRH). Más aún, resulta interesante que las endomorfinas sean capaces de inducir conductas antidepresivas o de tipo antidepresivos como se ha reportado recientemente en modelos animales de estrés y depresión. Estos estudios mostraron que las respuestas conductuales de reacción al estrés y las conductas antidepresivas mediadas por las EM1–2 están ligadas con la expresión neuronal del mensajero de RNA que codifica para el factor trófico (BDNF, por sus siglas en inglés, brain derived neurotrophic factor), en áreas del sistema limbico, y que es inducida en forma dosis–dependiente por las endomorfinas, posterior a su administración ICV. Por lo tanto, estos estudios han permitido proponer que las endomorfinas cumplen un papel relevante durante el curso o desarrollo de las enfermedades mentales (v.g., esquizofrenia y depresión). En extensión a estos estudios conductuales, estudios recientes han demostrado la actividad orexigénica de las endomorfinas en forma similar a lo previamente detectado con distintos ligandos agonistas del receptor opioide µ (v.g., morfina, DAMGO; morfina–6β–glucurónido). Si bien estos estudios mostraron que tanto las EM1–2 como diversos agonistas del receptor opioide µ exhiben potentes actividades orexigénicas en el SNC de roedores, la actividad de las EM1–2 parece depender de la actividad de la dinorfina A y su unión sobre su receptor opioide K en neuronas hipotalámicas. Más aún, diversos estudios han mostrado que el sistema opioide endógeno (a través de la β–endorfina) regula conductas de naturaleza sexual y apareamiento (v.g., lordosis), además de modular la secreción y/o actividad de hormonas de origen gonadal (estrógenos, progesterona).
Estudios similares en roedores hembras mostraron que la microinyección de EM1–2 en áreas específicas del sistema límbico y/ o la administración IT de ambos péptidos era capaz de generar respuestas sexuales de apareamiento, similares a las detectadas por la p–endorfina y morficeptina en la misma especie de animal, siendo bloqueados los efectos por la administración de naloxona. Estas respuestas conductuales inducidas por las EM1–2 mostraron estar ligadas a la liberación neuronal de LHRH, como de la activación y modulación del sistema de transmisión gabaérgico. En cuanto a las funciones de memoria y aprendizaje, diferentes estudios han demostrado que la administración ICV de EM1–2 en ratones expuestos a diferentes paradigmas de aprendizaje experimental, los péptidos opioides alteran significativamente los mecanismos de procesamiento y consolidación de memoria a corto y largo plazo en los animales tratados. Estos efectos parecen depender de la modulación del sistema opioide (v.g., el receptor opioide µ) sobre los sistemas de transmisión colinérgica y dopaminérgica en el cerebro de los mamíferos. Asímismo, diversos estudios han demostrado que tanto las EM1–2 como los alcaloides opiáceos y opioides endógenos modulan funciones cardiovasculares y respiratorias. En este contexto, diversos estudios mostraron que la administración de EM1–2 en ratas normotensas e hipertensas produce cambios fisiológicos significativos en la presión sanguínea y la frecuencia cardiaca. Si bien no están del todo esclarecidos los mecanismos por los cuales las endomorfinas producen sus respuestas cardiovasculares, diversos estudios sugieren que la actividad de estos péptidos está en función de la actividad e interacción de los sistemas de transmisión gabaérgico y glutamatérgico, respectivamente. Más aún, otros estudios sugieren que las respuestas fisiológicas de estos péptidos dependen de la actividad del óxido nitroso (NO, por sus siglas en inglés) liberado de los vasos sanguíneos, en respuesta de la activación del receptor opioide µ. Finalmente, diversos estudios han mostrado que las EM1–2 y la activación del receptor opioide µ producen efectos inhibitorios sobre la contracción del músculo liso del tracto gastrointestinal, generados a través de una reducción sostenida en la liberación de neurotransmisores de terminales sinápticas del plexo mientérico, mismas que inervan el tejido muscular liso del tracto gastrointestinal.
Palabras clave: Endomorfinas, fisiología, sensibilización locomotora, estrés, recompensa a opiacios, eje HPA, sexo, alimentación, cardiovascular, respiratorio, ansiolítico, conducta social.
VII. OPIOID RECEPTOR AGONISTS AND BEHAVIORAL SENSITIZATION
Repeated injections of psychoactive drugs into animals or humans usually lead either to a decrement of behavioral responses (tolerance) or an increase (sensitization) of psychomotor effects.1 Behavioral sensitization is a term often used to describe the neurochemical responsiveness and behavioral effects detected from repeated and intermittent administration of lower doses of a psychoactive drug.2 This phenomenon creates a drug <<preference>> state, which allows a drug to be used frequently once the substance of abuse has <<sensitized>> the active sites in the brain. Thus, drug–sensitization plays a crucial role in the development and maintenance of drug addiction,3 which may persist for long–term periods after drug withdrawal,4 enhancing an overwhelming urge (craving) for increased drug–seeking and drug–taking behaviors associated with a loss of behavioral control during long–term periods of abstinence.3,5 Interesting enough is that all drugs abused by humans have been shown to generate drug–rewarding effects and behavioral sensitization responses in animal models of drug addiction.6 The neuroanatomical and neurochemical bases of drug sensitization led researchers to focus on the mesocorticolimbic projecting dopamine (DA) pathway (neuron and axon fibers that emerge from the VTA and project to both NAC and mPFCx).7 Several interacting transmission systems besides of the dopamine (DA) system8,9 impinging directly or indirectly on VTA neurons; have been shown to mediate several of the evoked–behavioral sensitization responses to drug of abuse in animals, which includes the inhibitory GABAergic system,10 the glutamate (GLU)/aspartate (ASP) excitatory neurotransmission system,11 and the endogenous µ and δ–dependent opioid receptor systems12 (figure 1). Microdialysis experiments demonstrated that rats exposed to either systemic administration or direct injection of µ–opioid receptor agonists (e.g., DAMGO and morphine) into the VTA (but not in the NAc)12,13 enhanced a significant increase of DA release in the NAc,14–17 and conversely, administration of µ–opioid receptor antagonists (e.g., CTOP) attenuated the release of DA in the NAc.12 Likewise, chronic administration of endomorphins in the VTA produced a significant effect on the development of locomotor sensitization responses to amphetamine (AMPH).18 Endomorphin treatment significantly increased the tissue concentration of GLU and its metabolites in several limbic structures (e.g., NAc, mPFCx, CPu) in either EM1–2 plus AMPH–treated rats or AMPH–treated animals, used as controls. These results demonstrated that µ–opioid receptor agonists, including both EM1–2 peptides, induce behavioral sensitization responses in animals mediated via the activation of both GABA and GLU transmission systems in the VTA.10,19,20

VIII. ENDOMORPHINS MODULATING LOCOMOTOR BEHAVIORAL RESPONSES
Specific subcortical structures of the brain have been shown to play an important role in the control of movement.21,22
Within the basal ganglia, the globus pallidus contains a subpopulation of neurons that expresses high levels of µ–opioid receptor mRNA.21,22 µ–opioid receptor agonists have been shown to increase locomotor sensitization responses that are influenced by a number of variables (e.g., ligand concentration, experimental paradigm, and timing of experiment performed). Most studies using psychomotor paradigms have shown that µ– and δ–opioid receptor agonists enhanced an increment of ambulatory responses, whereas κ–opioid receptor agonists produced opposite responses.23,24 In this context, locomotor responses (e.g., horizontal and vertical locomotor) mediated by activation of the µ–opioid receptor by morphine–binding its cognate µ–opioid receptor25–27 and grooming behavior mediated by activation of δ–opioid binding sites (localized in the VTA, NAc, and PAG)26, 28, 29 have been shown to depend on the synthesis and release of DA from both nigrostriatal and mesolimbic dopaminergic neurons.30 Interactions between DA and opioid systems in the brain have been extensively reported. These studies showed that opiates (e.g., morphine, heroin) increase behavioral sensitivity responses to DA agonists, enhancing an increase supersensitivity of DA receptors and the expression of stereotypic behaviors mediated by the activation of D1R/D2R–receptor by ligand agonists.31 For instance, morphine potentiated an apomorphine–dependent climbing behavior in wild–type mice, as opposed to the mutant–µ–opioid receptor knockout mice.32 Thus, these set of results demonstrated that µ–receptor ligands alter the DA projecting system by potentiating the climbing behavior responses in mice induced after administration of D1R/D2R ligand agonists.33
Endomorphin peptides, like morphine, were found to increase locomotion (horizontal) activity34,35 without affecting the vertical locomotor activity19 in mice. These studies showed that low concentrations of EM2 (0.3 and 1.0 µg/animal, ICV administration) induce similar behavioral responses displayed by higher concentrations of EM1 peptide (10–30 µg/mouse, ICV administration). These results led researchers to posit that EM1–2 peptides not only activate different µ–opioid receptor subtypes in the basal ganglia,36,37 but could modulate different opioid systems (i.e. enkephalinergic, dynorphinergic) that appear to be implicated in the expression of the sensitization responses mediated by endomorphins.38,39 Similar studies demonstrated that morphine injected into the globus pallidus produced a robust increase in locomotor activity in mice,40 whereas EM1 induced localized stereotyped behavioral responses (e.g., orofacial dyskinesia).41 These opioid–mediated behavioral responses led authors to propose that the locomotor activity induced by morphine could be mediated via the activation of δ– and κ–opioid receptors, whereas EM1–inducing inhibitory activities would depend mostly on the activation of µ–opioid binding sites.41 As shown for the interaction of GABA and several endogenous opioid peptides (e.g., enkephalins),42 EM1 and GABA could mediate opposite behavioral responses in the control of movement at the globus pallidus. The resulting chemical unbalance induced in both neurotransmission systems could lead to the development of motor dysfunctions and the manifestation of localized dyskinesias.19
IX. ENDOMORPHINS REGULATING OPIATE REWARDING RESPONSES
Extensive studies have shown that the chronic administration of µ–opioid–receptor agonists (e.g., DAMGO, morphine, codeine, and sufentanyl)4345 produce potent drug– and/or stress–rewarding effects, associated to the development of drug withdrawal symptoms and physical dependence in animals.46,47 Quite interesting to note is that opposite (non–rewarding) aversion responses in rodents appeared after administration of selective κ–opioid receptor agonists.48
Drug–rewarding effects have been shown to be mediated via the interaction between GABAergic neurons and the mesolimbic/VTA–dopaminergic transmission system (see extensive reviews in49–51) (figure 1). As described above, opioid receptor agonists inhibit GABAergic inputs to VTA/ dopaminergic principal cells that project the NAc, inducing a disinhibitory effect, which in turn enhances a potent release of DA into this limbic structure.10,52 Endomorphin peptides injected into the posterior area of the VTA induced a conditioned–place preference (CPP), displaying similar behavioral responses to those exhibited by morphine or DAMGO.53 Moreover, EM1 injected into the VTA produced a potent rewarding effect in rodents exposed to the drug self–administration paradigm. However, injection of same peptide or DAMGO into the NAc produced poor and delayed rewarding effects compared to the VTA–detected responses.53 These data suggested the absence or poor expression of µ–opioid–receptor sites in this mesolimbic area.54
ICV administration of endomorphin peptides shed inconsistent results on the induced rewarding responses. For instance, some authors reported that EM1 mediated a significant CPP, whereas EM2 displayed significant place aversion effects in mice.55 Conversely, other authors reported that ICV administration of EM1–2 peptides (at low doses, 15 µg ) induced significant antinociceptive responses in mice56 producing no–effects on the CPP paradigm.57 However, higher doses of EM1 (30 µg) produced barrel rotation of the body trunk, whereas EM2 evoked a significant place preference condition in tested mice.57 Such discrepancies led authors to posit that the differential behavioral responses mediated after EM1–2 administration could be due to the activation of distinct opioid receptors (e.g., µ– and δ– opioid receptors); to the expression of a different pharmacogenetic background in animals; to the asymmetric expression of opioid receptor sites in targeted brain areas and cells; and/or to the activation of different molecular mechanism that drive the rewarding effects and behavioral responses to opioid substances.19
Moreover, differences in the behavioral effects induced by endomorphin peptides could be due to the activation of µ–opioid receptors expressed in neurons localized at the brainstem PAG region, involved in the generation of aversive behaviors.58 These effects have been associated to the disruption of the HPA axis, in addition to the deregulation of different mesolimbic transmission systems involved in rewarding functions, as shown in addicted humans exhibiting a history of long–term opiate abuse59 (figures 1 and 2).
X. ENDOMORPHINS IMPLICATED IN STRESS–INDUCING ALTERED BEHAVIORS
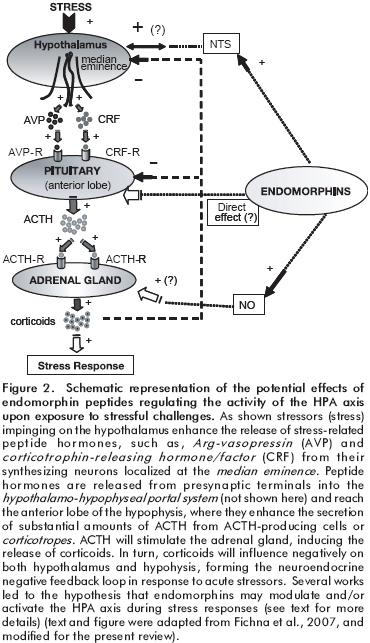
Stressors have been implicated in the development of several psychiatric illnesses, where the HPA axis and endogenous opioid system have been shown to play a crucial role in stress responses. Although the precise role of endogenous opioid peptides and receptors to stress stimuli has not been fully elucidated, over the past years several works showed the existing interactions between stressors, the HPA axis and the endogenous opioid system, (see reference,19 and references therein). These works showed a close relationship between the levels of the opioid ligands, corticosteroids, pituitary hormone levels, and immune–borne hormones (e.g., cytokines).19 Activation of the HPA axis by external/internal stressful stimuli (e.g., stress, immune challenge) leads to the increase secretion of corticotrophin–releasing factor (CRF) and Arg–vasopressin (AVP) from the median eminence of the hypothalamus, enhancing the cell–release of ACTH from the anterior lobe of the pituitary.60 Increased serum levels of ACTH enhance the release of glucocorticoids from the adrenal gland, which exert a negative feedback on pituitary adrenocorticotrophs and limbic regions of the mammalian CNS (e.g., amygdale), enhancing the homeostatic and neuroendocrine balance along the HPA axis.61 Some authors have postulated that the endogenous opioid system, driven through β–endorphin (binding µ–opioid receptors) at the hypothalamus exerts a potent inhibitory activity on the HPA axis.62,63 In this context, pharmacological studies showed that acute morphine administration, acting on µ–opioid receptors expressed along the HPA axis,64 produced an important increase of ACTH and adrenal secretion of corticosterone65 (figure 2).
Based on the aforementioned results, ICV administration of EM1 or EM2 peptides (10 µg) produced no stimulatory activity on the HPA axis and displayed no neuroendocrine effect on the ACTH and corticosterone secretion.60 Moreover, EM1 failed to block the stimulatory effect of morphine on the ACTH–induced increased levels of corticosterone in plasma. Furthermore, chronic activation of the HPA axis by exposure of animals to chronic stress paradigms (e.g., chronic inflammatory stress of adjuvant–induced arthritis, the restraint stress model, and the immune–based lipopolysaccharide stress model) showed that plasma corticosterone, ACTH and β–endorphin were dramatically increased, whereas levels of endomorphin peptides showed no detectable changes compared to controls. These data suggested that EM1–2 peptides appear to display no significant roles on the neuroendocrine modulation of the HPA axis, mediating stress responses to challenging stimuli.60
Based on that µ–opioid receptors are expressed on pituitary cells and neuronal cells within the hypothalamus, and receptor ligand agonists (e.g., morphine) properly induce a potent activation of the HPA axis, it has been proposed that the lack of endomorphin–induced neuroendocrine effects on the HPA axis could be due merely to the central metabolism or degradation of these peptides (see degradation of EM1–2 peptides in previous paper, part I) (figure 2).
Other plausible explanation offered relies on that µ–opioid receptor agonists display different stimulating properties of Gi/Go protein, as demonstrated in animal models of pain.38,66 Thus, intracellular molecules may provide important insights into the differential cell–responses of endomorphin peptides modulating several neural systems involved in distinct physiological effects and responses.
For instance, different works showed recently that nitric oxide (a chemical messenger molecule involved in different physiological and pathological processes in mammals) released from cells mediates several physiological responses induced by opioid peptides (e.g., β–endorphin) or morphine67–69 besides of the aforementioned endomorphin bioactivities (e.g., vasodilatory responses, modulation of HPA axis activity).35,67 This situation led researchers to propose the term <<endomorphin–NO–HPA axis>>,35,70 where endomorphin peptides acting either on hypothalamic neurons, may stimulate the HPA axis; or through the proposed NTS–endomorphin projecting neural system impinging on hypothalamic neuron cells (this neural component has been shown previously to activate directly the HPA axis in animals) (see previous studies in references71,72) (figure 2).
Other neural pathways emerging from the PAG have been shown to mediate stress–induced immobility and analgesia in adult rats and rat pups.73 Pharmacological experiments showed that µ–opioid receptor antagonists (e.g., naltrexone, CTOP) acting on the ventrolateral PAG blocked several analgesic responses mediated by endogenous opioids, providing evidences that endomorphins could mediate PAG–inducing stress–related analgesic effects in rats, as shown for different µ–opioid receptor ligand agonists.73 Thus, these neural and neuroendocrine driving mechanisms provide important insights that endomorphin peptides might be directly or indirectly involved in the activation of the HPA axis, enhancing the release of CRH from PVN/hypothalamic neurons.74,75
XI. ENDOMORPHINS INVOLVED IN MOOD–RELATED AND PSYCHIATRIC DISORDERS
Over the past decades extensive studies demonstrated that ICV (central)76,77 or IP (peripheral)78,79 administration of µ–opioid receptor agonists (e.g., morphine) produce anxiolytic responses, whereas µ–opioid receptor antagonists promote anxiogenic effects.80 These responses were shown to be mediated through the interaction of the EOS and the GABAergic system (e.g., GABA and BZD);81,82 the monoaminergic (e.g., 5–HT, BZD) and peptidergic systems.19
Moreover, these pharmacological and behavioral studies demonstrated that the anxiolytic and anxiogenic activities of opiates substances are dose– and site–dependent after their local administration into the rat neural tissue. For instance, low doses of morphine injected into rat midbrain tectum induced anxiolytic–like responses, whereas the injection of higher doses displayed anxiogenic–like effects.83 Converserly, morphine injected into the dorsal PAG84 or lateral septum of the rat brain85 has been shown to produce aversive responses. Previous reports describing the neuroanatomical co–localization between endomorphins and µ–opioid receptors in both limbic and brainstem regions and nuclei in the CNS of rodents,86,87 led to postulate the hypothesis that EM1–2 could modulate mood–related behaviors (e.g., anxiety and stress–related behaviors) in animals and humans.19 For instance, ICV administration of EM1 into mice induced an increased anxiolytic behavior responses in the elevated plus maze88 supporting previous observations that administrations of µ–opioid receptor agonists in humans produce anxiolytic symptomology (e.g., drowsiness, warmth feelings, and sensation of well–being).80
Earlier findings showed the expression of high density µ–opioid receptors and high concentration of endogenous opioid ligands (e.g., β–endorphin) in limbic areas of animals exposed to stressful challenges.89–92 These data led authors to postulate that the EOS and endomorphins play a crucial role in modulation in psychiatric disorders,93 such as depression and schizophrenia,94–98 in spite of the absence of a clear therapeutic benefit of opioid ligands to treat mental illnesses.19 Molecular and behavioral studies showed that knockout mice lacking the µ–opioid receptor display altered emotional states consistent of depressive–like behaviors, similar to those studies that have extensively demonstrated the use of a wide variety of µ–opioid receptor agonists, as antidepressant agents (e.g., oxycodone and oxymorphone)99 for treating depressive symptoms among many other mental illnesess.100–102 For instance, morphine has been used as an antidepressant–like agent to relieve stress behaviors in experimental animals.103 These data demonstrate the importance of the EOS in the etiology of mental disorders, besides of the controversial issue on the clinical use of µ–opioid receptor agonists as therapeutical agents to relieve psychiatric disorders.19
Based on the immunoreactive co–localization of EM1–2 peptides and µ–opioid receptors in both forebrain (e.g., septum, NAc, amygdala, thalamic nuclei) and brainstem regions (e.g., LC) in the CNS of mamamals, and shown previously with regard to their functional implication in the pathophysiology of depression;86,104,105 several authors have postulated the importance of these amide tetra–peptides in the etiology of depressive disorders, based on their potential antidepressant–like effects observed in animal models of depression and stress.19 The antidepressant–like effects detected for both EM1–2 peptides (0.3–30 µg/animal, ICV) in mice were dose–dependent and short–lasting (enduring only 10–15 min after their brain administration).19 The magnitude of the antidepressant responses displayed by both peptides was comparable to the several compounds shown to display potent antidepressant activities.106,107 These studies provided strong evidences that these opioid peptides, like conventional antidepressants, may generate antidepressant–like responses (reducing behavioral–immobility in paradigms of stress and depression [e.g., forced swimming test, FST])108 and which may be blocked by µ–opioid receptor antagonists (e.g., naloxone, (β–funal–trexamine) but not with selective δ– or κ–opioid receptor antagonists (e.g., naltrindole, nor–binaltorphimine, respectively).19 Additional studies regarding the implication of EM1–2 in the pathophysiology of depression showed that ICV administration of endomorphins (e.g.,20–50µg/animal), into rats induces a dose–dependent up–regulation of BDNF mRNA expression in limbic areas of the rat brain (mPFCx, hippocampus, amygdala) that was blocked by specific µ–opioid receptor antagonists (e.g., naltrexone), but not with specific δ–opioid receptor antagonists (e.g., naltrindole).109
This neurotrophic factor (BDNF) has been shown to modulate primordial functions, such as neuronal survival, differentiation, and plasticity,110 and shown recently to play an important role in the therapeutic actions of several antidepressants acting on different neurotransmission systems.111–115
XII. ENDOMORPHINS MEDIATING FOOD–INTAKE BEHAVIOR
The neural pathways and transmission systems that regulate food–intake behavior in mammals are complex.19 Several regulatory peptides (i.e., NPY, GHRH, 26RFa peptide) have been shown to display orexigenic activities in the brain.116 119 In a similar context, µ–opioid receptor ligand agonists (e.g., morphine, DAMGO), including the active morphine metabolite (e.g., morphine–6β glucuronide), have been shown to display an orexigenic activity120–123 regulating gustatory neural pathways arising from the NTS neurons124,125 (a neural pathway projecting to hypothalamic areas and other limbic structures).126 In a similar context, ICV administration of EM1 or EM2 (0.03–30 nmol) produced a dose–dependent food–intake behavior in non food–deprived mice for up to 4 h after peptide injection.88 These EM1–2 mediated effects were attenuated by the specific µ–opioid receptor antagonist, β–funaltrexamine.127,128 However, the endogenous κ–opioid receptor ligand peptide, Dynorphin A (DYN A), was shown to display a potent stimulatory food–intake bevavior compared to EM1–2 peptides. These results posit that the orexigenic activity induced by EM1–2 peptides appears to be mediated, via activation of κ–opioid receptors, where µ–opioid receptors appear to play a minor role in this opioid and non–opioid dependent physiological activity.19
XIII. ENDOMORPHINS MODULATING SEXUAL BEHAVIOR RESPONSES
Extensive studies demonstrated the direct and indirect effects of endogenous opioid peptides (e.g., β–endorphin) on gonadal hormones, regulating both sexually–induced behaviors (e.g., lordosis) and reproductive functions in female rats.129–131 These effects have been shown to be mediated through the activation of µ–opioid receptors expressed along the limbo–hypothalamic neural circuits that mediate the release of gonadal hormones (LH, FSH) from the pituitary and the release of its releasing peptide hormone (LHRH) from the hypothalamus.132 LH/FSH have been extensively shown to influence female rat sexual behavior,133 besides of modulating the release and expression of sexual steroids during mating behavior.134 Based on the neuroanatomical distribution and cell expression of µ–opioid receptors within specific hypothalamic and mesencephalic regions that coordinate and regulate female reproductive behavior (e.g., VMH, mPOA, MCG),104,135 several authors showed that µ–opioid receptor ligand agonists produce dual effects on lordosis in hormonally–primed female rodents during mating.135,136 In a similar context, ICV or local administration of low doses of β–endorphin into the MCG or mPOA produces a potent inhibition of lordosis in gonadectomized, steroid–primed female rats.137–140 Converserly, similar route of administration of high doses of β–endorphin or morphiceptin facilitated lordosis in estrogen– or estrogen/ progesterone primed rats.137,138,141–143 Moreover, ICV administration of EM1–2 peptides into the third ventricle or bilateral infusion into the diagonal band (DB) (septum–horizontal limb of the diagonal band, MS–HDB, an area shown to project axon fibers to the mPOA of the hypothalamus)144 produced dose– and time–dependent lordosis responses in female rats135 which was attenuated with naloxone.134 However, similar responses were not detected when peptides were injected into VMH, mPOA or MCG.134 These results led authors to propose that EM1–2 peptides modulate104,145 the release of active neuropeptides (e.g., LHRH) and non–peptide (Ach, GABA) transmitters within the MS–HDB, inducing their opioid–dependent behavioral effects.19
XIV. ENDOMORPHINS INVOLVED IN LEARNING AND MEMORY PROCESSING
The implication of the EOS among several other brain transmitters in learning and memory has been extensively studied and reported elsewhere.19 For instance, the EOS has been shown to play important roles in operant and classic conditioning and different cognitive tasks, including memory processing.19 These studies showed that either µ–opioid receptor (e.g., DAMGO and Tyr–D–Arg–Phe–β–Ala) and δ–selective opioid receptor (e.g.,D–Pen2,L–Pen5–enkephalin and D–Ala2–deltorphin II) agonists, respectively, induced an impairment of both short–term and long–term memory processing in mice exposed either to passive avoidance paradigms146–148 or spatial memory tasks.149 Conversely, µ–opioid receptor antagonists enhanced memory retention in animals exposed to different learning tasks.150 Similarly, κ–opioid receptor agonists (e.g., dynorphin A1–13) have been shown to attenuate aberrant learning and memory processing in rodents exposed to aversive and non–aversive memory tasks.151
With regard to endomorphin molecules, a single report from Ukai et al.152 showed that endomorphins impaired short–term memory processing in mice exposed to spontaneous alternation performance task. Whereas both tetrapeptides induced an important inhibitory activity on long–term memory processing when tested in passive avoidance learning task in mice,153,154 EM2 peptide was shown to mediate its memory attenuating effect155 by inducing an opioid receptor dependent cytosolic and mitochondrial protein synthesis mechanism in the lobus paraolfactorius in chicks (a brain area structurally related to the caudate putamen in mammals).156,157 These studies showed that EM2 reverted the amnesic effects induced by anisomycin administration into chicks, and blocked the inhibitory effect on protein synthesis induced by this drug in this related striatal–brain structure.155 However, other authors proposed that this EM2–dependent inhibition of passive avoidance learning task resulted from a functional disconnection of the hippocampus, a brain area known to be crucially important for processing and conversion of short–term memories into long–term memories (see reference158). Besides of the aforementioned studies, several authors proposed that both cholinergic and dopaminergic transmitter systems could mediate or participate in the opioid peptide–induced long–term memory impairment, although their exact roles have not been clearly elucidated.19 In this context, behavioral and pharmacological studies showed that spatial working memory requires at least the interaction between µ–opioid receptor ligand agonists (including both EM1/EM2 peptides) and the ACh transmission system.159 Cellular studies demonstrated that endomorphin peptides decreased significantly the release of ACh from neuronal cells in brain areas associated with memory processing and storage153,154,160 and that physostigmine (a cholinesterase inhibitor) reverted the endomorphin induced passive avoidance learning impaired response.146,154
Besides of the interaction between ACh and opioid system, several authors showed that D2–receptor antagonists were capable of attenuating the EM2–induced passive avoidance learning impairment in rodents.153 These results suggested that the inhibitory effect of EM2 would be mediated from stimulation of heterosynaptic D2 receptors expressed in dopaminergic neurons innervating both the striatum and the NAc, during acquisition and consolidation of memory.153 In addition, ICV administration of EM1–2 peptides was shown to increase BDNF mRNA expressions in the hippocampus and amygdala.109 This trophic factor, acting via its neuronal NT–3 receptor subtype, has been shown to mediate several plastic events in the brain, such as development and establishment of long–term potentiation (LTP) in hippocampal neurons; morphologic changes in active synapses and neurons in brain regions involved in learning and memory processing (e.g., hippocampus and cortex).161,162 Thus, based on the information described above, endomorphins acting via µ–opioid receptors could be implicated in learning and memory processing in several areas of the brain regulating BDNF activity on neuronal cells.19
XV. ENDOMORPHINS REGULATING CARDIOVASCULAR AND RESPIRATORY BIOACTIVITIES
Over the past years it has been shown that several regions along the rostracaudal axis (e.g., VLM, NTS, LH, PVN) of the rat brain, including the dorsal hippocampus and limbic system, regulate cardiovascular and respiratory bioactivities, areas shown also to express a high density of µ–opioid receptors in neurons within each functional brain region.19
1. Cardiovascular effects
Although several works have acknowledged the important role of the EOS mediating cardiovascular responses, the effects of distinct opioid receptor ligand agonists on blood pressure and heart rate have been unclear and confusing. For instance, local injections of µ, δand κ–opioid receptor agonists into the specific areas of the rat brain (e.g., PVN, DH, RVLM) in normotensive and/or hypertensive animals, were shown to reduce heart rate and blood pressure,163 whereas ICV administration of µ–opioid receptor agonists (e.g., morphine, (β–endorphin, DAMGO) produced hypotension in different species.93,164–166
In a similar context, ICV or IV administration of EM1–2 produced a significant reduction of heart rate and blood pressure in normotensive and hypertensive rats167–170 showing a reversible and dose–dependent biphasic change in systemic arterial pressure165 upon administration of µ–opioid receptor antagonists (e.g., naloxone or β–funaltrexamine). These results suggest that endomorphin peptides produce their cardioavascular responses via µ–opioid binding sites.164,168–170 However, differences in the administration route of EM2 into rats (IV versus ICV) produced different graded responses,171 which led authors to suggest that although peripheral µ–opioid receptors might play an important role in the opioid peptide–inducing hypotensive effects in animals,172 the mechanisms by which endomorphin peptides mediate both peripheral and central cardiovascular activities are not completely elucidated.
This assumption is based on previous results that demonstrated, on the one hand, that bradycardia results from activation of the vagus nerve, and bilateral vagotomy (or atropine) abolished EM1 effect on heart rate in rats, suggesting thus, that EM1–2 mediate their vascular effects via activation of vagal afferents.171 On the other hand, EM1–2 peptides which exert potent inhibitory activities on neurons,173–175 their cellular activities are expected to increase blood pressure and heart rate due an increase response on neuronal firing on cardiovascular regulatory areas at both medulla and cervical spinal cord.176–178 Recent studies showed that infusion of EM2 into the rat NTS (mNTS) attenuated reflex responses upon stimulation of the carotid sinus and aortic baroreceptors,176–178 showing that depressor and bradycardic responses resulted from a peripheral inhibitory effect on the baroreflex response and from the excitatory activity of mNTS neurons. These authors proposed that EM2 mediated its cardiovascular responses after being injected into the mNTS (a brainstem area control under local inhibitory–GABAergic neurons179,180 and glutamate [GLU] projecting neurons descending from insular cortex)181,182 through direct activation of µ–opioid receptors expressed on mNTS–GABA neurons, producing an hyperpolarization response, decreased of GABA release and increase of GLU release from presynaptic terminals, enhancing an overall reduction of GABAergic activity on mNTS postsynaptic neurons (disinhibitory effect) leading thus, to a final increase of neuronal excitability of mNTS neurons.19,176–178
Moreover, EM2 acting on µ–opioid receptors expressed on glutamate baroreceptor afferent terminals in response to baroreceptor stimulation were shown to decrease GLU release, resulting in attenuation of the baroreflex activity.19 Despite that the depressor and bradycardic responses induced by EM2 could be explained through the aforementioned cellular mechanisms,19,183 the endogenous mechanisms by which EM1–2 induced vasoactive responses are still unclear and further research may clarify this issue. However, several authors proposed different mechanisms by which endomorphins may mediate their hypotensive responses. One proposed mechanism is through the intracellular stimulation of NO synthesis164,184 and NO release from the vessels185 after binding their cognate opioid receptor on endothelial cells. Other mechanisms proposed is through the hyperpolarization of vascular smooth muscle cells and/or the cell–release of vasodilator prostanglandins or via an endomorphin inhibition of presynaptic release of NA from nerve endings distributed along vessel walls.172 Whichever the mechanisms involved, none of these have been completely confirmed, which requires further research to clarify the exact endogenous opioid–dependent mechanism.
2. Respiratory effects
Several studies demonstrated that µ–opioid receptor agonists induce a potent respiratory depression.186 Opioid–receptor agonists, such as morphine (see reference19 and references therein), heroin,187–189 fentanyl,190,191 buprenorphine,192–194 and DAMGO171,195 have been shown to induce a respiratory depression by decreasing the sensitivity of brainstem nuclei regulating–respiratory activity to carbon dioxide, followed by a decrement of respiratory rate (see reviews in46,196,197).
Although immunochemical studies showed that both EM1–2–LI198 and µ–opioid receptors199,200 appear to be colocalized at specific neuroanatomical areas and nuclei of the brainstem of mammals (e.g., NTS and PBN)104 shown to play a crucial role in the respiratory control,19 very few studies have focused on the effects of endomorphin peptides on respiratory activity. For instance, IV administration of supraanalgesic doses of EM1 and EM2 peptides in rats produced biphasic responses, characterized by rapid initial ventilatory depression (inhibitory effect that lasts for 4–6 s) followed by an increase ventilation activity (excitatory effect that lasts for 10–12 min).201 In contrast to the monotonically–induced decreased ventilation activity by morphine, EM1–2 appear to mediate their depressant respiratory activity via activation of central µ–opioid receptors, based on that methylnaloxone (a peripheral restricted µ–opioid receptor antagonist) was unable to block peripherically the opioid effects, whereas naloxone prevented the respiratory–depressed activity induced by endomorphin peptides.201 Interestingly, EM1–2 peptides administered (I.V.) at doses higher above their corresponding analgesic threshold or higher above their respiratory depression–threshold dose (as shown for DAMGO or morphine) showed to attenuate hypercapnic ventilatory responses in rats, producing a respiratory depression in tested animals.201 Although EM1–2 peptides induced a weaker depression activity on ventilatory responses compared to the hypercapnia effects induced by either DAMGO or morphine,201 the reduced effects mediated by EM1–2 peptides have been extensively discussed based on several pharmacokinetic and pharmacodynamic parameters and factors discussed elsewhere.19,201 In this context, the increased ventilation activity induced by EM1–2 peptides appeared to be mediated through a central non–opioid mechanism, based on that conventional µ–opioid receptor antagonists were unable to block the increased respiratory response.201
Interaction of endomorphins and other neurotransmission systems regulating respiration activity202,203 showed that both EM1 and EM2 produced dose–dependent inhibition of tachykinin–mediated contractions of the guinea pig bronchus, with the exception of EM1 activity which was blocked by naloxone, whereas that of EM2 was not antagonized by any µ–opioid receptor antagonist.202
In the same context, Patel et al.203 showed that EM1–2 peptides produced a dose–dependent inhibition and naloxone–reversible antagonism activity on cholinergic induced–contractile responses of the guinea pig trachea. These studies showed that EM1–2 peptides, acting on µ–opioid receptors distributed in rodent airways,204 interact with both cholinergic and tachykinergic fiber types.202,203 Thus, EM1–2 peptides were shown to induce a potent inhibition on the electrically–evoked release of ACh from cholinergic nerves innervating guinea pig trachea,203 including the release of NA from nonadrenergic postganglionic nerve fibers innervating the airway–smooth muscle cells.203 Recent studies showed that ICV administration of EM1 in mice induced an increased oxygen consumption that was blocked by naloxone, suggesting a µ–opioid receptor peptide dependent effect.205
XVI. ENDOMORPHINS INVOLVED IN GASTROINTESTINAL ACTIVITY
Opioid agonists regulating gastrointestinal activity have been extensively reported. IHC techniques and binding studies, using neuromuscular preparations, showed that µ–opioid receptors may be localized in smooth muscle cells and neurons, as well206 where µ–opioid receptor agonists (e.g.,morphine) have been shown to exert, in a naloxone–reversible fashion, their inhibitory effects on GI activity and/or motility,207–209 besides of modulating the evoked release of neurotransmitter release from nerve terminals (e.g., ACh, NA)209–211 and influencing peristaltic reflex.207
In a similar context, EM1–2 peptides were shown to modulate different GI activities. For instance, application of a concentration range of EM1–2 peptides (10–12 M to 10–6 M) on a specific guinea pig ileum preparation (longitudinal muscle–myenteric plexus preparations from ileum) produced a dose–dependent– and CTOP–reversible inhibition of the amplitude of electrically–induced twitch ileum contractions.212 However, EM1–2 peptides failed to inhibit muscle contractions induced by ACh stimulation. Moreover, both peptides displayed a potent inhibitory effect on the ascending excitatory reflex and increased stimulation of the descending inhibitory reflex, inducing an increase latency on the onset of ileum muscle–contractile responses.213 Similar inhibitory responses were detected at the smooth and striated muscles of the rat esophagus.214
Overall, these results led authors to suggest that EM1–2 induce their inhibitory GI effects, via activation of µ–opioid receptors localized either in presynaptic terminals of non–adrenergic/non–cholinergic inhibitory neurons and/or in myenteric–plexus neurons that innervate smooth–muscle cells.212,214 Activation of µ–opioid receptors may lead to a reduced release response of different neurotransmitters (e.g., NO, VIP), including ACh and NA from local and myenteric neurons, respectively.210–213
XVII. ENDOGENOUS OPIOIDS AND SOCIAL BEHAVIORS
A clear example of stress–inducing increased HPA activity occurs in foraging and defensive behaviors in animals.215 For example, after social defeat, subordinate animals display physiological, neuroendocrine, neurochemical and behavioral changes induced by the endogenous stress–driving mechanisms in socially–interacting species.216 These behavioral and functional changes have been suggested to be highly connected to the development of fear, anxiety, depression, and panic disorders,217,218 including the development of drug–seeking and drug–taking behaviors in both animals and humans.219 Moreover, few studies have shown that µ–opioid receptor results to be up–regulated in restricted regions of the rat brain (e.g., VTA) after social defeat.220 ICV administration of EM1 into Syrian hamsters failed to inhibit the consolidation of conditioned defeat (without stimulating locomotor activity or inducing sedation),221 whereas morphine impaired the consolidation of newly acquired memories in rats and mice.222–225 These authors suggested that such reduced or failed behavioral responses mediated by EM1 could be due to cellular responses and pharmacological activities mediated through the binding of the peptide to its cognate receptor (see detailed explanations in19 and references therein). For instance, morphine, DAMGO, besides of other potent endomorphin peptide analogs (e.g., Tyr–D–Arg–Phe–β–Ala),88,226,227 have been shown to inhibit memory retrieval, increase anxiolytic responses and produce fear–conditioning responses in animals.77,78,228,229 Such responses appear to depend on the activation of NA neurons and NA neural system.230
ACKNOWLEDGEMENTS
This review is dedicated to the memory of Professor Ramón de la Fuente Muñiz, whose vision and knowledge in distinct areas on the neuroscience research led to the support and development of several projects in our lab. Moreover, we gratefully thank the following institutions for their funding to the present manuscript: National Institute of Psychiatry (Instituto Nacional de Psiquiatria) Mexico–Project INPRF–NC092318.1; INP–2040, ICYTDF–2007; CONACYT–SALUD–2003–C01–14; CONACyT/SEP–COI–47804; CONACyT–FOSSIS/SALUD–2007–COI–69373; Megaproyecto UNAM MP6–16; Fundación Gonzalo Río Arronte A.C. CONADIC and BIRMEX are also acknowledged for supporting the preparation of this manuscript.
REFERENCES
1. Segal DS, Kuczenski R. Repeated binge exposures to amphetamine and methamphetamine: behavioral and neurochemical characterization. J Pharmacol Exp Ther 1997; 282:561–573. [ Links ]
2. Spyraki C, Fibiger HC, Phillips AG. Attenuation of heroin reward in rats by disruption of the mesolimbic dopamine system. Psychopharmacology (Berl) 1983; 79:278–283. [ Links ]
3. Robinson TE, Berridge KC. The neural basis of drug craving: an incentive–sensitization theory of addiction. Brain Res Brain Res Rev 1993; 18:247–291. [ Links ]
4. Robinson TE, Becker JB. Enduring changes in brain and behavior produced by chronic amphetamine administration: a review and evaluation of animal models of amphetamine psychosis. Brain Res 1986; 396:157–198. [ Links ]
5. Wise RA, Bozarth MA. A psychomotor stimulant theory of addiction. Psychol Rev 1987; 94:469–492. [ Links ]
6. Stewart J, Badiani A. Tolerance and sensitization to the behavioral effects of drugs. Behav Pharmacol 1993; 4:289–312. [ Links ]
7. Spanagel R. Modulation of drug–induced sensitization processes by endogenous opioid systems. Behav Brain Res 1995; 70:37–49. [ Links ]
8. Hooks MS, Kalivas PW. Involvement of dopamine and excitatory amino acid transmission in novelty–induced motor activity. J Pharmacol Exp Ther 1994; 269:976–988. [ Links ]
9. Vezina P. D1 dopamine receptor activation is necessary for the induction of sensitization by amphetamine in the ventral tegmental area. J Neurosci 1996; 16:2411–2420. [ Links ]
10. Johnson SW, North RA. Opioids excite dopamine neurons by hyperpolarization of local interneurons. J Neurosci 1992; 12:483–488. [ Links ]
11. Karler R, Chaudhry IA, Calder LD et al. Amphetamine behavioral sensitization and the excitatory amino acids. Brain Res 1990; 537:76–82. [ Links ]
12. Spanagel R, Herz A, Shippenberg TS. Opposing tonically active endogenous opioid systems modulate the mesolimbic dopaminergic pathway. Proc Natl Acad Sci U S A 1992; 89:2046–2050. [ Links ]
13. Devine DP, Leone P, Wise RA. Mesolimbic dopamine neurotransmission is increased by administration of mu–opioid receptor antagonists. Eur J Pharmacol 1993; 243:55–64. [ Links ]
14. Di CG, Imperato A. Opposite effects of mu and kappa opiate agonists on dopamine release in the nucleus accumbens and in the dorsal caudate of freely moving rats. J Pharmacol Exp Ther 1988; 244:1067–1080. [ Links ]
15. Di CG, Imperato A. Drugs abused by humans preferentially increase synaptic dopamine concentrations in the mesolimbic system of freely moving rats. Proc Natl Acad Sci U S A 1988; 85:5274–5278. [ Links ]
16. Pentney RJ, Gratton A. Effects of local delta and mu opioid receptor activation on basal and stimulated dopamine release in striatum and nucleus accumbens of rat: an in vivo electrochemical study. Neuroscience 1991; 45:95–102. [ Links ]
17. Spanagel R, Herz A, Shippenberg TS. The effects of opioid peptides on dopamine release in the nucleus accumbens: an in vivo microdialysis study. J Neurochem 1990; 55:1734–1740. [ Links ]
18. Chen JC, Liang KW, Huang EY. Differential effects of endomorphin–1 and –2 on amphetamine sensitization: neurochemical and behavioral aspects. Synapse 2001; 39:239–248. [ Links ]
19. Fichna J, Janecka A, Costentin J et al. The endomorphin system and its evolving neurophysiological role. Pharmacol Rev 2007; 59:88–123. [ Links ]
20. Wolf ME, Xue CJ. Amphetamine and D1 dopamine receptor agonists produce biphasic effects on glutamate efflux in rat ventral tegmental area: modification by repeated amphetamine administration. J Neuro–chem 1998; 70:198–209. [ Links ]
21. Delfs JM, Kong H, Mestek A et al. Expression of mu opioid receptor mRNA in rat brain: an in situ hybridization study at the single cell level. J Comp Neurol 1994; 345:46–68. [ Links ]
22. Peckys D, Landwehrmeyer GB. Expression of mu, kappa, and delta opioid receptor messenger RNA in the human CNS: a 33P in situ hybridization study. Neuroscience 1999; 88:1093–1135. [ Links ]
23. Obeso JA, Rodriguez–Oroz MC, Rodriguez M et al. Pathophysiology of levodopa–induced dyskinesias in Parkinson's disease: problems with the current model. Ann Neurol 2000; 47:S22–S32. [ Links ]
24. Kuzmin A, Sandin J, Terenius L et al. Dose– and time–dependent bimodal effects of kappa–opioid agonists on locomotor activity in mice. J Pharmacol Exp Ther 2000; 295:1031–1042. [ Links ]
25. Austin MC, Kalivas PW. Enkephalinergic and GABAergic modulation of motor activity in the ventral pallidum. J Pharmacol Exp Ther 1990; 252:1370–1377. [ Links ]
26. Schildein S, Agmo A, Huston JP et al. Intraaccumbens injections of substance P, morphine and amphetamine: effects on conditioned place preference and behavioral activity. Brain Res 1998; 790:185–194. [ Links ]
27. Stinus L, Robert C, Karasinski P et al. Continuous quantitative monitoring of spontaneous opiate withdrawal: locomotor activity and sleep disorders. Pharmacol Biochem Behav 1998; 59:83–89. [ Links ]
28. Joyce EM, Iversen SD. The effect of morphine applied locally to mesencephalic dopamine cell bodies on spontaneous motor activity in the rat. Neurosci Lett 1979; 14:207–212. [ Links ]
29. Morgan MM, Whitney PK, Gold MS. Immobility and flight associated with antinociception produced by activation of the ventral and lateral/dorsal regions of the rat periaqueductal gray. Brain Res 1998; 804:159–166. [ Links ]
30. Cunningham ST, Kelley AE. Opiate infusion into nucleus accumbens: contrasting effects on motor activity and responding for conditioned reward. Brain Res 1992; 588:104–114. [ Links ]
31. de la BS, Patey G, Marcais H et al. Changes in dopamine receptors in mouse striatum following morphine treatments. Life Sci 1979; 24:2333–2342. [ Links ]
32. Ritzmann RF, Walter R, Bhargava HN et al. Blockage of narcotic–induced dopamine receptor supersensitivity by cyclo(Leu–Gly). Proc Natl Acad Sci U S A 1979; 76:5997–5998. [ Links ]
33. Jang C, Park Y, Tanaka S et al. Involvement of mu–opioid receptors in potentiation of apomorphine–induced climbing behavior by morphine: studies using mu–opioid receptor gene knockout mice. Brain Res Mol Brain Res 2000; 78:204–206. [ Links ]
34. Bujdoso E, Jaszberenyi M, Tomboly C et al. Behavioral and neuroendocrine actions of endomorphin–2. Peptides 2001; 22:1459–1463. [ Links ]
35. Bujdoso E, Jaszberenyi M, Gardi J et al. The involvement of dopamine and nitric oxide in the endocrine and behavioural action of endomorphin–1. Neuroscience 2003; 120:261–268. [ Links ]
36. Sakurada S, Zadina JE, Kastin AJ et al. Differential involvement of mu–opioid receptor subtypes in endomorphin–1– and –2–induced antinociception. Eur J Pharmacol 1999; 372:25–30. [ Links ]
37. Sakurada S, Hayashi T, Yuhki M et al. Differential antagonism of endo–morphin–1 and endomorphin–2 spinal antinociception by naloxonazine and 3–methoxynaltrexone. Brain Res 2000; 881:1–8. [ Links ]
38. Sanchez–Blazquez P, Rodriguez–Diaz M, DeAntonio I et al. Endomorphin–1 and endomorphin–2 show differences in their activation of mu opioid receptor–regulated G proteins in supraspinal antinociception in mice. J Pharmacol Exp Ther 1999; 291:12–18. [ Links ]
39. Tseng LF, Narita M, Suganuma C et al. Differential antinociceptive effects of endomorphin–1 and endomorphin–2 in the mouse. J Pharmacol Exp Ther 2000; 292:576–583. [ Links ]
40. Anagnostakis Y, Krikos Y, Spyraki C. Pallidal substrate of morphine–induced locomotion. Eur Neuropsychopharmacol 1992; 2:65–72. [ Links ]
41. Mehta A, Bot G, Reisine T et al. Endomorphin–1: induction of motor behavior and lack of receptor desensitization. J Neurosci 2001; 21:4436–4442. [ Links ]
42. Bayon A, Anton B, Leff P et al. Release of proteins, enzymes, and the neuroactive peptides, enkephalins, from the striatum of the freely moving rat. Ann N Y Acad Sci 1986; 473:401–417. [ Links ]
43. Devine DP, Wise RA. Self–administration of morphine, DAMGO, and DPDPE into the ventral tegmental area of rats. J Neurosci 1994; 14:1978–1984. [ Links ]
44. Negus SS, Henriksen SJ, Mattox A et al. Effect of antagonists selective for mu, delta and kappa opioid receptors on the reinforcing effects of heroin in rats. J Pharmacol Exp Ther 1993; 265:1245–1252. [ Links ]
45. Piepponen TP, Kivastik T, Katajamaki J et al. Involvement of opioid mu 1 receptors in morphine–induced conditioned place preference in rats. Pharmacol Biochem Behav 1997; 58:275–279. [ Links ]
46. Reisine T, Pasternak G. Opioid analgesics and antagonists. In: Hardman J, Gilman A, Limbird L eds. Goodman and Gilman's The Pharmacological Basis of Therapeutics. New York.: McGraw–Hill; 1996:521–555. [ Links ]
47. Rockhold RW, Liu N, Coleman D et al. The nucleus paragigantocellularis and opioid withdrawal–like behavior. J Biomed Sci 2000; 7:270–276. [ Links ]
48. Becker A, Grecksch G, Brodemann R et al. Morphine self–administration in mu–opioid receptor–deficient mice. Naunyn Schmiedebergs Arch Pharmacol 2000; 361:584–589. [ Links ]
49. Kalivas PW, Volkow ND. The neural basis of addiction: a pathology of motivation and choice. Am J Psychiatry 2005; 162:1403–1413. [ Links ]
50. Tomkins DM, Sellers EM. Addiction and the brain: the role of neurotransmitters in the cause and treatment of drug dependence. CMAJ 2001; 164:817–821. [ Links ]
51. Wolf ME. Addiction: making the connection between behavioral changes and neuronal plasticity in specific pathways. Mol Interv 2002; 2:146–157. [ Links ]
52. Margolis EB, Hjelmstad GO, Bonci A et al. Kappa–opioid agonists directly inhibit midbrain dopaminergic neurons. J Neurosci 2003; 23:9981–9986. [ Links ]
53. Zangen A, Ikemoto S, Zadina JE et al. Rewarding and psychomotor stimulant effects of endomorphin–1: anteroposterior differences within the ventral tegmental area and lack of effect in nucleus accumbens. J Neurosci 2002; 22:7225–7233. [ Links ]
54. Churchill L, Klitenick MA, Kalivas PW. Dopamine depletion reorganizes projections from the nucleus accumbens and ventral pallidum that mediate opioid–induced motor activity. J Neurosci 1998; 18:8074–8085. [ Links ]
55. Narita M, Ozaki S, Ioka M et al. Different motivational effects induced by the endogenous mu–opioid receptor ligands endomorphin–1 and –2 in the mouse. Neuroscience 2001; 105:213–218. [ Links ]
56. Wilson AM, Soignier RD, Zadina JE et al. Dissociation of analgesic and rewarding effects of endomorphin–1 in rats. Peptides 2000; 21:1871–1874. [ Links ]
57. Huang EY, Chen CM, Tao PL. Supraspinal anti–allodynic and rewarding effects of endomorphins in rats. Peptides 2004; 25:577–583. [ Links ]
58. Sante AB, Nobre MJ, Brandao ML. Place aversion induced by blockade of mu or activation of kappa opioid receptors in the dorsal periaqueductal gray matter. Behav Pharmacol 2000; 11:583–589. [ Links ]
59. Kreek MJ, Koob GF. Drug dependence: stress and dysregulation of brain reward pathways. Drug Alcohol Depe nd 1998; 51:23–47. [ Links ]
60. Coventry TL, Jessop DS, Finn DP et al. Endomorphins and activation of the hypothalamo–pituitary–adrenal axis. J Endocrinol 2001; 169:185–193. [ Links ]
61. Harbuz MS, Lightman SL. Responses of hypothalamic and pituitary mRNA to physical and psychological stress in the rat. J Endocrinol 1989; 122:705–711. [ Links ]
62. Kreek MJ, Borg L, Zhou Y et al. Relationships between endocrine functions and substance abuse syndromes: heroin and related short–acting opiates in addiction contrasted with methadone and other long–acting opioid agonists used in pharmacotherapy of addiction. In: Pfaff D ed. Hormones, Brain and Behavior. San Diego: Academic Press; 2002. [ Links ]
63. Nikolarakis KE, Almeida OF, Herz A. Feedback inhibition of opioid peptide release in the hypothalamus of the rat. Neuroscience 1987; 23:143–148. [ Links ]
64. Mellon RD, Bayer BM. Role of central opioid receptor subtypes in morphine–induced alterations in peripheral lymphocyte activity. Brain Res 1998; 789:56–67. [ Links ]
65. Ignar DM, Kuhn CM. Effects of specific mu and kappa opiate tolerance and abstinence on hypothalamo–pituitary–adrenal axis secretion in the rat. J Pharmacol Exp Ther 1990; 255:1287–1295. [ Links ]
66. Sanchez–Blazquez P, Garzon J. Pertussis toxin differentially reduces the efficacy of opioids to produce supraspinal analgesia in the mouse. Eur J Pharmacol 1988; 152:357–361. [ Links ]
67. Champion HC, Bivalacqua TJ, Zadina JE et al. Vasodilator responses to the endomorphin peptides, but not nociceptin/OFQ, are mediated by nitric oxide release. Ann N Y Acad Sci 1999; 897:165–172. [ Links ]
68. Granados–Soto V, Rufino MO, Gomes Lopes LD et al. Evidence for the involvement of the nitric oxide–cGMP pathway in the antinociception of morphine in the formalin test. Eur J Pharmacol 1997; 340:177–180. [ Links ]
69. Gholami A, Haeri–Rohani A, Sahraie H et al. Nitric oxide mediation of morphine–induced place preference in the nucleus accumbens of rat. Eur J Pharmacol 2002; 449:269–277. [ Links ]
70. Calignano A, Persico P, Mancuso F et al. L–arginine modulates morphine–induced changes in locomotion in mice. Ann Ist Super Sanita 1993; 29:409–412. [ Links ]
71. Ter Horst GJ, de BP, Luiten PG et al. Ascending projections from the solitary tract nucleus to the hypothalamus. A Phaseolus vulgaris lectin tracing study in the rat. Neuroscience 1989; 31:785–797. [ Links ]
72. Boscan P, Pickering AE, Paton JF. The nucleus of the solitary tract: an integrating station for nociceptive and cardiorespiratory afferents. Exp Physiol 2002; 87:259–266. [ Links ]
73. Wiedenmayer CP, Barr GA. Mu opioid receptors in the ventrolateral periaqueductal gray mediate stress–induced analgesia but not immobility in rat pups. Behav Neurosci 2000; 114:125–136. [ Links ]
74. Saper B. Central autonomic system. In: Paxinos G ed. The Rat Nervous System. New York.: Academic Press, 1995:107–135. [ Links ]
75. Swanson LW. The hypothalamus. In: Bjorklund A, Hokfelt T, Swanson LW eds. Handbook of Chemical Neuroanatomy. New York: Elsevier; 1987. [ Links ]
76. Costall B, Jones BJ, Kelly ME et al. Exploration of mice in a black and white test box: validation as a model of anxiety. Pharmacol Biochem Behav 1989; 32:777–785. [ Links ]
77. Motta V, Penha K, Brandao ML. Effects of microinjections of mu and kappa receptor agonists into the dorsal periaqueductal gray of rats submitted to the plus maze test. Psychopharmacology (Berl) 1995; 120:470–474. [ Links ]
78. Koks S, Soosaar A, Voikar V et al. BOC–CCK–4, CCK(B)receptor agonist, antagonizes anxiolytic–like action of morphine in elevated plus–maze. Neuropeptides 1999; 33:63–69. [ Links ]
79. Zarrindast MR, Rostami P, Zarei M et al. Intracerebroventricular effects of histaminergic agents on morphine–induced anxiolysis in the elevated plus–maze in rats. Basic Clin Pharmacol Toxicol 2005; 97:276–281. [ Links ]
80. Tsuda M, Suzuki T, Misawa M et al. Involvement of the opioid system in the anxiolytic effect of diazepam in mice. Eur J Pharmacol 1996; 307:7–14. [ Links ]
81. Kang YS, Park JH. Brain uptake and the analgesic effect of oxytocin—its usefulness as an analgesic agent. Arch Pharm Res 2000; 23:391–395. [ Links ]
82. Sasaki K, Fan LW, Tien LT et al. The interaction of morphine and gamma–aminobutyric acid (GABA)ergic systems in anxiolytic behavior: using mu–opioid receptor knockout mice. Brain Res Bull 2002; 57:689–694. [ Links ]
83. Brandao ML, Anseloni VZ, Pandossio JE et al. Neurochemical mechanisms of the defensive behavior in the dorsal midbrain. Neurosci Biobehav Rev 1999; 23:863–875. [ Links ]
84. Nobre MJ, Ribeiro dos SN, Aguiar MS et al. Blockade of mu– and activation of kappa–opioid receptors in the dorsal periaqueductal gray matter produce defensive behavior in rats tested in the elevated plus–maze. Eur J Pharmacol 2000; 404:145–151. [ Links ]
85. Le MJ, Cagniard B, Cazala P. Modulation of anxiety by mu–opioid receptors of the lateral septal region in mice. Pharmacol Biochem Behav 2006; 83:465–479. [ Links ]
86. Zadina JE. Isolation and distribution of endomorphins in the central nervous system. Jpn J Pharmacol 2002; 89:203–208. [ Links ]
87. Kieffer BL. Opioids: first lessons from knockout mice. Trends Pharmacol Sci 1999; 20:19–26. [ Links ]
88. Asakawa A, Inui A, Momose K et al. Endomorphins have orexigenic and anxiolytic activities in mice. Neuroreport 1998; 9:2265–2267. [ Links ]
89. Belenky GL, Holaday JW. The opiate antagonist naloxone modifies the effects of electroconvulsive shock (ECS) on respiration, blood pressure and heart rate. Brain Res 1979; 177:414–417. [ Links ]
90. Bodnar RJ, Klein GE. Endogenous opiates and behavior: 2003. Peptides 2004; 25:2205–2256. [ Links ]
91. Mansour A, Khachaturian H, Lewis ME et al. Anatomy of CNS opioid receptors. Trends Neurosci 1988; 11:308–314. [ Links ]
92. Waksman G, Hamel E, Fournie–Zaluski MC et al. Autoradiographic comparison of the distribution of the neutral endopeptidase <<enkephalinase>> and of mu and delta opioid receptors in rat brain. Proc Natl Acad Sci USA 1986; 83:1523–1527. [ Links ]
93. Vaccarino AL, Olson GA, Olson RD et al. Endogenous opiates: 1998. Peptides 1999; 20:1527–1574. [ Links ]
94. Darko DF, Risch SC, Gillin JC et al. Association of beta–endorphin with specific clinical symptoms of depression. Am J Psychiatry 1992; 149:1162–1167. [ Links ]
95. Gabilondo AM, Meana JJ, Garcia–Sevilla JA. Increased density of mu–opioid receptors in the postmortem brain of suicide victims. Brain Res 1995; 682:245–250. [ Links ]
96. Gross–Isseroff R, Dillon KA, Israeli M et al. Regionally selective increases in mu opioid receptor density in the brains of suicide victims. Brain Res 1990; 530:312–316. [ Links ]
97. Lindstrom LH, Widerlov E, Gunne LM et al. Endorphins in human cerebrospinal fluid: clinical correlations to some psychotic states. Acta Psychiatr Scand 1978; 57:153–164. [ Links ]
98. Scarone S, Gambini O, Calabrese G et al. Asymmetrical distribution of beta–endorphin in cerebral hemispheres of suicides: preliminary data. Psychiatry Res 1990; 32:159–166. [ Links ]
99. Stoll AL, Rueter S. Treatment augmentation with opiates in severe and refractory major depression. Am J Psychiatry 1999; 156:2017. [ Links ]
100. Makino M, Kitano Y, Komiyama C et al. Involvement of central opioid systems in human interferon–alpha induced immobility in the mouse forced swimming test. Br J Pharmacol 2000; 130:1269–1274. [ Links ]
101. Makino M, Kitano Y, Komiyama C et al. Human interferon–alpha increases immobility in the forced swimming test in rats. Psychopharmacology (Berl) 2000; 148:106–110. [ Links ]
102. Vilpoux C, Carpentier C, Leroux–Nicollet I et al. Differential effects of chronic antidepressant treatments on micro– and delta–opioid receptors in rat brain. Eur J Pharmacol 2002; 443:85–93. [ Links ]
103. Eschalier A, Fialip J, Varoquaux O et al. Study of the clomipramine–morphine interaction in the forced swimming test in mice. Psychopharmacology (Berl) 1987; 93:515–519. [ Links ]
104. Martin–Schild S, Gerall AA, Kastin AJ et al. Differential distribution of endomorphin 1– and endomorphin 2–like immunoreactivities in the CNS of the rodent. J Comp Neurol 1999; 405:450–471. [ Links ]
105. Schreff M, Schulz S, Wiborny D et al. Immunofluorescent identification of endomorphin–2–containing nerve fibers and terminals in the rat brain and spinal cord. Neuroreport 1998; 9:1031–1034. [ Links ]
106. Cryan JF, Mombereau C, Vassout A. The tail suspension test as a model for assessing antidepressant activity: review of pharmacological and genetic studies in mice. Neurosci Biobehav Rev 2005; 29:571–625. [ Links ]
107. Petit–Demouliere B, Chenu F, Bourin M. Forced swimming test in mice: a review of antidepressant activity. Psychopharmacology (Berl) 2005; 177:245–255. [ Links ]
108. Porsolt RD, Bertin A, Jalfre M. Behavioral despair in mice: a primary screening test for antidepressants. Arch Int Pharmacodyn Ther 1977; 229:327–336. [ Links ]
109. Zhang H, Torregrossa MM, Jutkiewicz EM et al. Endogenous opioids upregulate brain–derived neurotrophic factor mRNA through delta– and micro–opioid receptors independent of antidepressant–like effects. Eur J Neurosci 2006; 23:984–994. [ Links ]
110. Binder DK, Scharfman HE. Brain–derived neurotrophic factor. Growth Factors 2004; 22:123–131. [ Links ]
111. Castren E. Neurotrophins as mediators of drug effects on mood, addiction, and neuroprotection. Mol Neurobiol 2004; 29:289–302. [ Links ]
112. D'Sa C, Duman RS. Antidepressants and neuroplasticity. Bipolar Disord 2002; 4:183–194. [ Links ]
113. Hashimoto K, Shimizu E, Iyo M. Critical role of brain–derived neurotrophic factor in mood disorders. Brain Res Brain Res Rev 2004; 45:104–114. [ Links ]
114. Shimizu E, Hashimoto K, Okamura N et al. Alterations of serum levels of brain–derived neurotrophic factor (BDNF) in depressed patients with or without antidepressants. Biol Psychiatry 2003; 54:70–75. [ Links ]
115. Shirayama Y, Chen AC, Nakagawa S et al. Brain–derived neurotrophic factor produces antidepressant effects in behavioral models of depression. J Neurosci 2002; 22:3251–3261. [ Links ]
116. Chartrel N, Dujardin C, Anouar Y et al. Identification of 26RFa, a hypothalamic neuropeptide of the RFamide peptide family with orexigenic activity. Proc Natl Acad Sci U S A 2003; 100:15247–15252. [ Links ]
117. Inui A, Okita M, Nakajima M et al. Neuropeptide regulation of feeding in dogs. Am J Physiol 1991; 261:R588–R594. [ Links ]
118. Sakurai T, Amemiya A, Ishii M et al. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein–coupled receptors that regulate feeding behavior. Cell 1998; 92:573–585. [ Links ]
119. Vaccarino FJ, Bloom FE, Rivier J et al. Stimulation of food intake in rats by centrally administered hypothalamic growth hormone–releasing factor. Nature 1985; 314:167–168. [ Links ]
120. Glass MJ, Billington CJ, Levine AS. Opioids and food intake: distributed functional neural pathways? Neuropeptides 1999; 33:360–368. [ Links ]
121. Gosnell BA, Levine AS, Morley JE. The stimulation of food intake by selective agonists of mu, kappa and delta opioid receptors. Life Sci 1986; 38:1081–1088. [ Links ]
122. Morley JE, Levine AS, Grace M et al. An investigation of the role of kappa opiate receptor agonists in the initiation of feeding. Life Sci 1982; 31:2617–2626. [ Links ]
123. Woods JS, Leibowitz SF. Hypothalamic sites sensitive to morphine and naloxone: effects on feeding behavior. Pharmacol Biochem Behav 1985; 23:431–438. [ Links ]
124. Matsuo R, Shimizu N, Kusano K. Lateral hypothalamic modulation of oral sensory afferent activity in nucleus tractus solitarius neurons of rats. J Neurosci 1984; 4:1201–1207. [ Links ]
125. Moufid–Bellancourt S, Velley L. Effects of morphine injection into the parabrachial area on saccharin preference: modulation by lateral hypothalamic neurons. Pharmacol Biochem Behav 1994; 48:127–133. [ Links ]
126. Kuhn CM, Windh RT. Endocrine actions of opiates. In: Watson RR ed. Biochemistry and Physiology of Substance Abuse. Boca Raton, FL.: CRC Press; 1989:247–277. [ Links ]
127. Leventhal L, Mathis JP, Rossi GC et al. Orphan opioid receptor antisense probes block orphanin FQ–induced hyperphagia. Eur J Pharmacol 1998; 349:R1–R3. [ Links ]
128. Leventhal L, Silva RM, Rossi GC et al. Morphine–6beta–glucuronide–induced hyperphagia: characterization of opioid action by selective antagonists and antisense mapping in rats. J Pharmacol Exp Ther 1998; 287:538–544. [ Links ]
129. Argiolas A. Neuropeptides and sexual behaviour. Neurosci Biobehav Rev 1999; 23:1127–1142. [ Links ]
130. Gilbert CL, Boulton MI, Goode JA et al. The timing of parturition in the pig is altered by intravenous naloxone. Theriogenology 2000; 53:905–923. [ Links ]
131. Pfaus JG, Gorzalka BB. Opioids and sexual behavior. Neurosci Biobehav Rev 1987; 11:1–34. [ Links ]
132. Bakker J, Baum MJ. Neuroendocrine regulation of GnRH release in induced ovulators. Front Neuroendocrinol 2000; 21:220–262. [ Links ]
133. Vathy I, van der PJ, Vincent PA et al. Intracranial dialysis and microinfusion studies suggest that morphine may act in the ventromedial hypothalamus to inhibit female rat sexual behavior. Horm Behav 1991; 25:354–366. [ Links ]
134. Sinchak K, Micevych PE. Progesterone blockade of estrogen activation of mu–opioid receptors regulates reproductive behavior. J Neurosci 2001; 21:5723–5729. [ Links ]
135. Pfaff DW, Schwartz–Gliblin S, McCarthy MM et al. Cellular and molecular mechanisms of female reproductive behaviors. In: Knobil E, Neill JD eds. The Physiology of Reproduction. New York: Raven Press; 1994. [ Links ]
136. van Furth WR, van Emst MG, Van Ree JM. Opioids and sexual behavior of male rats: involvement of the medial preoptic area. Behav Neurosci 1995; 109:123–134. [ Links ]
137. Pfaus JG, Pendleton N, Gorzalka BB. Dual effect of morphiceptin on lordosis behavior: possible mediation by different opioid receptor subtypes. Pharmacol Biochem Behav 1986; 24:1461–1464. [ Links ]
138. Pfaus JG, Gorzalka BB. Selective activation of opioid receptors differentially affects lordosis behavior in female rats. Peptides 1987; 8:309–317. [ Links ]
139. Sirinathsinghji DJ. Modulation of lordosis behaviour in the female rat by corticotropin releasing factor, beta–endorphin and gonadotropin releasing hormone in the mesencephalic central gray. Brain Res 1985; 336:45–55. [ Links ]
140. Sirinathsinghji DJ. Regulation of lordosis behaviour in the female rat by corticotropin–releasing factor, beta–endorphin/corticotropin and luteinizing hormone–releasing hormone neuronal systems in the medial preoptic area. Brain Res 1986; 375:49–56. [ Links ]
141. Torii M, Kubo K. The effects of intraventricular injection of beta–endorphin on initial estrogen action to induce lordosis behavior. Physiol Behav 1994; 55:157–162. [ Links ]
142. Torii M, Kubo K, Sasaki T. Differential effects of beta–endorphin and Met– and Leu–enkephalin on steroid hormone–induced lordosis in ovariectomized female rats. Pharmacol Biochem Behav 1997; 58:837–842. [ Links ]
143. Torii M, Kubo K, Sasaki T. Facilitatory and inhibitory effects of beta–endorphin on lordosis in female rats: relation to time of administration. Horm Behav 1999; 35:271–278. [ Links ]
144. Jakab RL, Leranth C. Septum. In: Paxinos G ed. The Rat Nervous System. San Diego: Academic Press; 1995. [ Links ]
145. Loughlin SE, Leslie FM, Fallon JH. Endogenous opioid systems. In: Paxinos G ed. The Rat Nervous System. San Diego: Academic Press; 1995. [ Links ]
146. Itoh J, Ukai M, Kameyama T. Dynorphin A–(1–13) potently improves the impairment of spontaneous alternation performance induced by the mu–selective opioid receptor agonist DAMGO in mice. J Pharmacol Exp Ther 1994; 269:15–21. [ Links ]
147. Ukai M, Mori K, Hashimoto S et al. Tyr–D–Arg–Phe–beta–Ala–NH2, a novel dermorphin analog, impairs memory consolidation in mice. Eur J Pharmacol 1993; 239:237–240. [ Links ]
148. Ukai M, Takada A, Sasaki Y et al. Stimulation of delta1– and delta2–opioid receptors produces amnesia in mice. Eur J Pharmacol 1997; 338:1–6. [ Links ]
149. Bostock E, Gallagher M, King RA. Effects of opioid microinjections into the medial septal area on spatial memory in rats. Behav Neurosci 1988; 102:643–652. [ Links ]
150. Ferry B, McGaugh JL. Role of amygdala norepinephrine in mediating stress hormone regulation of memory storage. Acta Pharmacol Sin 2000; 21:481–493. [ Links ]
151. Ukai M, Kobayashi T, Kameyama T. Dynorphin A–(1–13) attenuates basal forebrain–lesion–induced amnesia in rats. Brain Res 1993; 625:355–356. [ Links ]
152. Ukai M, Watanabe Y, Kameyama T. Effects of endomorphins–1 and –2, endogenous mu–opioid receptor agonists, on spontaneous alternation performance in mice. Eur J Pharmacol 2000; 395:211–215. [ Links ]
153. Ukai M, Lin HP. Endomorphins 1 and 2 induce amnesia via selective modulation of dopamine receptors in mice. Eur J Pharmacol 2002; 446:97–101. [ Links ]
154. Ukai M, Lin HP. Involvement of mu(1)–opioid receptors and cholinergic neurotransmission in the endomorphins–induced impairment of passive avoidance learning in mice. Behav Brain Res 2002; 129:197–201. [ Links ]
155. Freeman FM, Young IG. Identification of the opioid receptors involved in passive–avoidance learning in the day–old chick during the second wave of neuronal activity. Brain Res 2000; 864:230–239. [ Links ]
156. Durstewitz D, Kroner S, Gunturkun O. The dopaminergic innervation of the avian telencephalon. Prog Neurobiol 1999; 59:161–195. [ Links ]
157. Metzger M, Jiang S, Wang J et al. Organization of the dopaminergic in–nervation of forebrain areas relevant to learning: a combined immuno–histochemical/retrograde tracing study in the domestic chick. J Comp Neurol 1996; 376:1–27. [ Links ]
158. Ukai M, Watanabe Y, Kameyama T. Endomorphins 1 and 2, endogenous mu–opioid receptor agonists, impair passive avoidance learning in mice. Eur J Pharmacol 2001; 421:115–119. [ Links ]
159. Kameyama T, Ukai M, Shinkai N. Ameliorative effects of tachykinins on scopolamine–induced impairment of spontaneous alternation performance in mice. Methods Find Exp Clin Pharmacol 1998; 20:555–560. [ Links ]
160. Ragozzino ME, Wenk GL, Gold PE. Glucose attenuates a morphine–induced decrease in hippocampal acetylcholine output: an in vivo microdialysis study in rats. Brain Res 1994; 655:77–82. [ Links ]
161. Huang EJ, Reichardt LF. Neurotrophins: roles in neuronal development and function. Annu Rev Neurosci 2001; 24:677–736. [ Links ]
162. Malcangio M, Lessmann V. A common thread for pain and memory synapses? Brain–derived neurotrophic factor and trkB receptors. Trends Pharmacol Sci 2003; 24:116–121. [ Links ]
163. Sun SY, Liu Z, Li P et al. Central effects of opioid agonists and naloxone on blood pressure and heart rate in normotensive and hypertensive rats. Gen Pharmacol 1996; 27:1187–1194. [ Links ]
164. Champion HC, Bivalacqua TJ, Friedman DE et al. Nitric oxide release mediates vasodilator responses to endomorphin 1 but not nociceptin/OFQ in the hindquarters vascular bed of the rat. Peptides 1998; 19:1595–1602. [ Links ]
165. Champion HC, Bivalacqua TJ, Lambert DG et al. Endomorphin 1 and 2, the endogenous mu–opioid agonists, produce biphasic changes in systemic arterial pressure in the cat. Life Sci 1998; 63:L131–L136. [ Links ]
166. Olson GA, Olson RD, Vaccarino AL et al. Endogenous opiates: 1997. Peptides 1998; 19:1791–1843. [ Links ]
167. Champion HC, Zadina JE, Kastin AJ et al. The endogenous mu–opioid agonists, endomorphin 1 and 2, have vasodilator activity in the hindquarters vascular bed of the rat. Life Sci 1997; 61:L–15. [ Links ]
168. Czapla MA, Champion HC, Zadina JE et al. Endomorphin 1 and 2, endogenous mu–opioid agonists, decrease systemic arterial pressure in the rat. Life Sci 1998; 62:L175–L179. [ Links ]
169. Kwok EH, Dun NJ. Endomorphins decrease heart rate and blood pressure possibly by activating vagal afferents in anesthetized rats. Brain Res 1998; 803:204–207. [ Links ]
170. Makulska–Nowak HE, Gumulka SW, Lipkowski AW et al. Effects of endomorphin–2 on arterial blood pressure and pain threshold in spontaneously hypertensive rats and modification of these effects by betafunaltrexamine and nor–binaltorphimine. Life Sci 2001; 69:581–589. [ Links ]
171. Czapla MA, Gozal D, Alea OA et al. Differential cardiorespiratory effects of endomorphin 1, endomorphin 2, DAMGO, and morphine. Am J Respir Crit Care Med 2000; 162:994–999. [ Links ]
172. Rialas CM, Fimiani C, Bilfinger TV et al. Endomorphin–1 and –2 inhibit human vascular sympathetic norepinephrine release: lack of interaction with mu 3 opiate receptor subtype. Zhongguo Yao Li Xue Bao 1998; 19:403–407. [ Links ]
173. Dun NJ, Dun SL, Wu SY et al. Endomorphins: localization, release and action on rat dorsal horn neurons. J Biomed Sci 2000; 7:213–220. [ Links ]
174. Guyenet PG, Stornetta RL, Schreihofer AM et al. Opioid signalling in the rat rostral ventrolateral medulla. Clin Exp Pharmacol Physiol 2002; 29:238–242. [ Links ]
175. Wu SY, Dun SL, Wright MT et al. Endomorphin–like immunoreactivity in the rat dorsal horn and inhibition of substantia gelatinosa neurons in vitro. Neuroscience 1999; 89:317–321. [ Links ]
176. Dampney RA. Functional organization of central pathways regulating the cardiovascular system. Physiol Rev 1994; 74:323–364. [ Links ]
177. Sapru HN. Glutamate circuits in selected medullo–spinal areas regulating cardiovascular function. Clin Exp Pharmacol Physiol 2002; 29:491–496. [ Links ]
178. Viard E, Sapru HN. Endomorphin–2 in the medial NTS attenuates the responses to baroreflex activation. Brain Res 2006; 1073–1074:365–373. [ Links ]
179. Izzo PN, Sykes RM, Spyer KM. gamma–Aminobutyric acid immuno–reactive structures in the nucleus tractus solitarius: a light and electron microscopic study. Brain Res 1992; 591:69–78. [ Links ]
180. Maqbool A, Batten TF, McWilliam PN. Ultrastructural Relationships Between GABAergic Terminals and Cardiac Vagal Preganglionic Motoneurons and Vagal Afferents in the Cat: A Combined HRP Tracing and Immunogold Labelling Study. Eur J Neurosci 1991; 3:501–513. [ Links ]
181. Owens NC, Sartor DM, Verberne AJ. Medial prefrontal cortex depressor response: role of the solitary tract nucleus in the rat. Neuroscience 1999; 89:1331–1346. [ Links ]
182. Torrealba F, Muller C. Glutamate immunoreactivity of insular cortex afferents to the nucleus tractus solitarius in the rat: a quantitative electron microscopic study. Neuroscience 1996; 71:77–87. [ Links ]
183. Kasamatsu K, Chitravanshi VC, Sapru HN. Depressor and bradycardic responses to microinjections of endomorphin–2 into the NTS are mediated via ionotropic glutamate receptors. Am J Physiol Regul Integr Comp Physiol 2004; 287:R715–R728. [ Links ]
184. Champion HC, Kadowitz PJ. D–[Ala2]endomorphin 2 and endomorphin 2 have nitric oxide–dependent vasodilator activity in rats. Am J Physiol 1998; 274:H1690–H1697. [ Links ]
185. Champion HC, Bivalacqua TJ, Zadina JE et al. Role of nitric oxide in mediating vasodilator responses to opioid peptides in the rat. Clin Exp Pharmacol Physiol 2002; 29:229–232. [ Links ]
186. Haji A, Takeda R, Okazaki M. Neuropharmacology of control of respiratory rhythm and pattern in mature mammals. Pharmacol Ther 2000; 86:277–304. [ Links ]
187. Comer SD, Collins ED, MacArthur RB et al. Comparison of intravenous and intranasal heroin self–administration by morphine–maintained humans. Psychopharmacology (Berl) 1999; 143:327–338. [ Links ]
188. Kishioka S, Ko MC, Woods JH. Diltiazem enhances the analgesic but not the respiratory depressant effects of morphine in rhesus monkeys. Eur J Pharmacol 2000; 397:85–92. [ Links ]
189. Vivian JA, Kishioka S, Butelman ER et al. Analgesic, respiratory and heart rate effects of cannabinoid and opioid agonists in rhesus monkeys: antagonist effects of SR 141716A. J Pharmacol Exp Ther 1998; 286:697–703. [ Links ]
190. Gerak LR, Brandt MR, France CP. Studies on benzodiazepines and opioids administered alone and in combination in rhesus monkeys: ventilation and drug discrimination. Psychopharmacology (Berl) 1998; 137:164–174. [ Links ]
191. Ma D, Sapsed–Byrne SM, Chakrabarti MK et al. Synergistic interaction between the effects of propofol and midazolam with fentanyl on phrenic nerve activity in rabbits. Acta Anaesthesiol Scand 1998; 42:670–677. [ Links ]
192. Benedetti F, Amanzio M, Baldi S et al. Inducing placebo respiratory depressant responses in humans via opioid receptors. Eur J Neurosci 1999; 11:625–631. [ Links ]
193. Kishioka S, Paronis CA, Lewis JW et al. Buprenorphine and methoclocinnamox: agonist and antagonist effects on respiratory function in rhesus monkeys. Eur J Pharmacol 2000; 391:289–297. [ Links ]
194. Schuh KJ, Walsh SL, Stitzer ML. Onset, magnitude and duration of opioid blockade produced by buprenorphine and naltrexone in humans. Psychopharmacology (Berl) 1999; 145:162–174. [ Links ]
195. Takita K, Herlenius E, Lindahl SG et al. Age– and temperature–dependent effects of opioids on medulla oblongata respiratory activity: an in vitro study in newborn rat. Brain Res 1998; 800:308–311. [ Links ]
196. Martin WR. Pharmacology of opioids. Pharmacol Rev 1983; 35:283–323. [ Links ]
197. Shook JE, Watkins WD, Camporesi EM. Differential roles of opioid receptors in respiration, respiratory disease, and opiate–induced respiratory depression. Am Rev Respir Dis 1990; 142:895–909. [ Links ]
198. Pierce TL, Wessendorf MW. Immunocytochemical mapping of endomorphin–2–immunoreactivity in rat brain. J Chem Neuroanat 2000; 18:181–207. [ Links ]
199. Moss IR. Respiratory responses to single and episodic hypoxia during development: mechanisms of adaptation. Respir Physiol 2000; 121:185–197. [ Links ]
200. Moss IR, Laferriere A. Prenatal cocaine raises mu–opioid receptor density in piglet cardiorespiratory medulla. Neurotoxicol Teratol 2000; 22:3–10. [ Links ]
201. Czapla MA, Zadina JE. Reduced suppression of CO2–induced ventilatory stimulation by endomorphins relative to morphine. Brain Res 2005; 1059:159–166. [ Links ]
202. Fischer A, Undem BJ. Naloxone blocks endomorphin–1 but not endomorphin–2 induced inhibition of tachykinergic contractions of guinea–pig isolated bronchus. Br J Pharmacol 1999; 127:605–608. [ Links ]
203. Patel HJ, Venkatesan P, Halfpenny J et al. Modulation of acetylcholine release from parasympathetic nerves innervating guinea–pig and human trachea by endomorphin–1 and –2. Eur J Pharmacol 1999; 374:21–24. [ Links ]
204. Belvisi MG, Stretton CD, Verleden GM et al. Inhibition of cholinergic neurotransmission in human airways by opioids. J Appl Physiol 1992; 72:1096–1100. [ Links ]
205. Asakawa A, Inui A, Ueno N et al. Endomorphin–1, an endogenous mu–opioid receptor–selective agonist, stimulates oxygen consumption in mice. Horm Metab Res 2000; 32:51–52. [ Links ]
206. Storr M, Hahn A, Gaffal E et al. Effects of endomorphin–1 and –2 on mu–opioid receptors in myenteric neurons and in the peristaltic reflex in rat small intestine. Clin Exp Pharmacol Physiol 2002; 29:428–434. [ Links ]
207. Allescher HD, Storr M, Brechmann C et al. Modulatory effect of endogenous and exogenous opioids on the excitatory reflex pathway of the rat ileum. Neuropeptides 2000; 34:62–68. [ Links ]
208. Asai T, Mapleson WW, Power I. Effects of nalbuphine, pentazocine and U50488H on gastric emptying and gastrointestinal transit in the rat. Br J Anaesth 1998; 80:814–819. [ Links ]
209. Nishiwaki H, Saitoh N, Nishio H et al. Relationship between inhibitory effect of endogenous opioid via mu–receptors and muscarinic autoinhibition in acetylcholine release from myenteric plexus of guinea pig ileum. Jpn J Pharmacol 1998; 77:279–286. [ Links ]
210. Cosentino M, Marino F, Bombelli R et al. Modulation of neurotransmitter release by opioid mu– and kappa–receptors from adrenergic terminals in the myenteric plexus of the guinea–pig colon: effect of alpha 2–autoreceptor blockade. Neurosci Lett 1997; 222:75–78. [ Links ]
211. Yokotani K, Osumi Y. Involvement of mu–receptor in endogenous opioid peptide–mediated inhibition of acetylcholine release from the rat stomach. Jpn J Pharmacol 1998; 78:93–95. [ Links ]
212. Tonini M, Fiori E, Balestra B et al. Endomorphin–1 and endomorphin–2 activate mu–opioid receptors in myenteric neurons of the guinea–pig small intestine. Naunyn Schmiedebergs Arch Pharmacol 1998; 358:686–689. [ Links ]
213. Storr M, Gaffal E, Schusdziarra V et al. Endomorphins 1 and 2 reduce relaxant non–adrenergic, non–cholinergic neurotransmission in rat gastric fundus. Life Sci 2002; 71:383–389. [ Links ]
214. Storr M, Geisler F, Neuhuber WL et al. Endomorphin–1 and –2, endogenous ligands for the mu–opioid receptor, inhibit striated and smooth muscle contraction in the rat oesophagus. Neurogastroenterol Motil 2000; 12:441–448. [ Links ]
215. Miczek KA, Thompson ML, Tornatzky W. Subordinate animals: behavioral and physiological adaptations and opioid tolerance. In: Brown MR, Koob GF, Rivier C eds. Neurobiology and Neuroendocrinology of Stress. Ney York: Marcel Dekker; 1991. [ Links ]
216. Martinez M, Calvo–Torrent A, Pico–Alfonso MA. Social defeat and subordination as models of social stress in laboratory rodents: a review. Aggress Behav 1998; 24:241–256. [ Links ]
217. Blanchard RJ, Nikulina JN, Sakai RR et al. Behavioral and endocrine change following chronic predatory stress. Physiol Behav 1998; 63:561–569. [ Links ]
218. Blanchard RJ, Herbert M, Sakai RR et al. Chronic social stress: changes in behavioral and physiologicalindices of emotion. Aggress Behav 1998; 24:307–321. [ Links ]
219. Haney M, Maccari S, Le MM et al. Social stress increases the acquisition of cocaine self–administration in male and female rats. Brain Res 1995; 698:46–52. [ Links ]
220. Nikulina EM, Hammer RP, Jr., Miczek KA et al. Social defeat stress increases expression of mu–opioid receptor mRNA in rat ventral tegmental area. Neuroreport 1999; 10:3015–3019. [ Links ]
221. Whitten RD, Jasnow AM, Albers HE et al. The effects of endomorphin–1 on conditioned defeat in Syrian hamsters (Mesocricetus auratus). Brain Res 2001; 914:74–80. [ Links ]
222. Izquierdo I. Effect of naloxone and morphine on various forms of memory in the rat: possible role of engogenous opiate mechanisms in memory consolidation. Psychopharmacology (Berl) 1979; 66:199–203. [ Links ]
223. Introini IB, McGaugh JL, Baratti CM. Pharmacological evidence of a central effect of naltrexone, morphine, and beta–endorphin and a peripheral effect of met– and leu–enkephalin on retention of an inhibitory response in mice. Behav Neural Biol 1985; 44:434–446. [ Links ]
224. Castellano C, Cestari V, Cabib S et al. The effects of morphine on memory consolidation in mice involve both D1 and D2 dopamine receptors. Behav Neural Biol 1994; 61:156–161. [ Links ]
225. Rudy JW, Kuwagama K, Pugh CR. Isolation reduces contextual but not auditory–cue fear conditioning: a role for endogenous opioids. Behav Neurosci 1999; 113:316–323. [ Links ]
226. Saha N, Datta H, Sharma PL. Effects of morphine on memory: interactions with naloxone, propranolol and haloperidol. Pharmacology 1991; 42:10–14. [ Links ]
227. Ukai M, Kobayashi T, Mori K et al. Attenuation of memory with Tyr–D–Arg–Phe–beta–Ala–NH2, a novel dermorphin analog with high affinity for mu–opioid receptors. Eur J Pharmacol 1995; 287:245–249. [ Links ]
228. File SE, Rodgers RJ. Partial anxiolytic action of morphine sulphate following microinjection into the central nucleus of the amygdala in rats. Pharmacol Biochem Behav 1979; 11:313–318. [ Links ]
229. Motta V, Brandao ML. Aversive and antiaversive effects of morphine in the dorsal periaqueductal gray of rats submitted to the elevated plus–maze test. Pharmacol Biochem Behav 1993; 44:119–125. [ Links ]
230. Introini IB, Baratti CM. The impairment of retention induced by beta–endorphin in mice may be mediated by a reduction of central cholinergic activity. Behav Neural Biol 1984; 41:152–163. [ Links ]














