Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista mexicana de fitopatología
versión On-line ISSN 2007-8080versión impresa ISSN 0185-3309
Rev. mex. fitopatol vol.35 no.3 Texcoco sep. 2017
https://doi.org/10.18781/r.mex.fit.1702-1
Scientific articles
Dieback disease of Prunus sp. associated an Armillaria spp. in commercial orchards in Michoacan, Mexico
1Campo Experimental Valle de México-INIFAP. Km. 13.5 Carretera Los Reyes-Texcoco. C.P. 56250. Coatlinchán, Estado de México.
2Centro Nacional de Referencia Fitosanitaria, SENASICA-SAGARPA. Km. 37.5 Carretera Federal México-Pachuca, C.P. 55740, Tecámac, Estado de México.
3Estación Nacional de Epidemiología, Cuarentena y Saneamiento Vegetal. Centro Nacional de Referencia Fitosanitaria Dirección General de Sanidad Vegetal, SENASICA-SAGARPA. Km. 21 Carretera Amazcala-Chichimequillas, Municipio El Marqués C.P. 76247, Santiago de Querétaro, Querétaro.
4Sitio Experimental Querétaro-INIFAP. Luis Pasteur 414, Colonia Valle Alameda C.P. 76040 Santiago de Querétaro, Querétaro.
The producing area of peach (Prunus persica) and plum (Prunus domestica) in Michoacan is located at Zinapecuaro county. Recently there have been symptoms of a progressive deterioration of trees that causes their death, increasing the risk of fruit production failure. The aim of this study was to identify and characterize molecularly the responsible pathogen. Five sites with previous experiences of having the problem were sampled: root, trunk and soil samples were collected. Signs of Armillaria spp. were found, which was confirmed by molecular sequencing of the Tef-1α gene (translation elongation factor 1α). The phylogenetic analysis performed, grouped the sequences as news species, phylogenetically related in the same clade as A. mellea, which is a very aggressive pathogen. At present in Mexico, tolerant rootstocks have not been identified and it is therefore recommended to continue searching for genetic resistance and improve orchard disease management.
Key words: Molecular characterization; phylogenetic analysis; Tef-1α; molecular sequence
El área productora de durazno (Prunus persica) y ciruelo (Prunus domestica) en Michoacán, se localiza en el municipio de Zinapécuaro. Recientemente se han presentado síntomas de un deterioro progresivo de los árboles que les ocasiona la muerte, poniendo en riesgo la producción de fruta. El objetivo de este estudio fue identificar y caracterizar molecularmente el patógeno responsable. En las localidades de Ucareo, Jeráhuaro, Huajúmbaro, San Joaquín y La Galera con antecedentes del problema, se colectaron muestras de raíz, tronco y suelo. Se identificaron signos característicos del hongo Armillaria spp., confirmado por secuenciación molecular del gen Tef-1α (translation elongation factor 1α). El análisis filogenético agrupó las secuencias como dos especies nuevas, filogenéticamente emparentada en el mismo clado que A. mellea, que es una especie patógena agresiva. En México no se cuenta actualmente con portainjertos tolerantes, y por tanto, se recomienda continuar con estudios de búsqueda de resistencia genética y manejo de la enfermedad en las huertas.
Palabras clave: Caracterización molecular; análisis filogenético; Tef-1α; secuenciación molecular
The largest peach (Prunus persica) and plum (Prunus domestica) producing area in Michoacan is located at Zinapecuaro county (19° 52’ northern latitude, 100° 50’ western longitude and 2400 to 2700 masl) in the northwestern area of the state. The topography includes the Trans-Mexican Volcanic Belt, with temperate climate and summer rains. The average annual rainfall is 622 mm and temperatures range between 3 and 34 °C (Villaseñor, 2005). The peach and plum producing area has expanded to areas of mixed pine-oak and fir-oak forests; the peach production area planted on 1256 ha represents 30 % of the state’s total area, and is concentrated in five locations: Ucareo, Jerahuaro, Huajumbaro, San Joaquin and La Galera, where 6,950 tons valued at $ 41.7 million pesos were harvested in 2015 (SAGARPA, 2015). The area planted to plum was 2827 ha in 2014, which produced 13,316 tons valued at $ 47.1 million pesos (SAGARPA, 2014).
In recent years, peach and plum trees in some commercial orchards have been affected by progressive damage and gradual death that may occur within a few months. The first symptoms appear during the flowering stage (leaf wilting, yellowing at the top of the trees, stunting, base canker, tree neck exudate, lack of vigor and death).
The Armillaria genus includes over 40 species. Identifying this genus based on morphological traits is complicated because, in most cases, only asexual or vegetative structures are found. One of the issues extensively discussed in the literature is the lack of sexual structures that would allow identifying the genus based on classical taxonomy (Kim et al., 2006; Brazee et al., 2011; Ross-Davis et al., 2012). Over the past few years, molecular data, specifically DNA sequences, have been used to identify species of fungi and conduct phylogenetic studies (Cai et al., 2011; Guo et al., 2016; Koch et al., 2017). With the contemporary taxonomy of this fungus, comparing DNA sequences has led to the discovery of new species that are not related to morphological species (Maphosa et al., 2006), a fact that has had implications for identifying the fungus.
In natural forest areas and fruit orchards, species of Armillaria genus have been detected (Cibrián et al., 2007) that behave as saprophytes or pathogens (Ross-Davis et al., 2012). Even after infected trees are cut, the mycelium survives as a saprophyte, and residual roots can remain buried in the soil and serve as inoculum (Redfern and Filip, 1991). The inoculum can survive from a few years to decades (Baumgartner and Rizzo, 2001), and combined with the lack of efficient disease control methods (Adaskaveg et al., 1999, Aguin-Casal et al., 2006), contributes to significantly reduce fruit yields during the whole life of both infected orchards (Baumgartner, 2004) and subsequent orchards.
In Mexico, the fungus is distributed in the states of Baja California, Chiapas, Chihuahua, State of Mexico, Durango, Guanajuato, Guerrero, Jalisco, Michoacan, Morelos, Nuevo Leon, Oaxaca, Puebla, Queretaro, Veracruz and in forest areas of Mexico City (Valdés et al., 2004; Cibrián et al., 2007; Elías-Román et al., 2013; Klopfenstein et al., 2014; Fernández-Pavía, et al., 2015). The objective of this research was to identify and molecularly characterize the pathogen associated with dieback of peach and plum trees. The hypothesis of this research was that Armillaria spp. is responsible for causing peach and plum tree dieback in the fruit production area of Zinapecuaro county, Michoacan.
MATERIALS AND METHODS
Sampling
In 2015 and 2016, five sites were sampled in peach and plum commercial orchards located at the upper, intermediate and lower part of the Zinapecuaro production area (Table 1). Trees were identified with different damage symptoms (leaf wilting, tree top yellowing, stunting, basal canker, neck exudate, lack of vigor and death) at each site. A destructive sampling was undertaken to collect samples of primary and secondary roots and trunks at a height of 15-30 cm. It was verified that pathogenic signs and symptoms were present in the root system of the tree, where in some cases rhizomorphs, typical vegetative structures of Armillaria, and whitish-brown mycelial fans between bark and wood were found. Soil samples were taken at a depth of 30-60 cm.
Processing plant and soil samples
Samples of plant material were thoroughly washed to remove soil; they were then cut into pieces 1 cm long, placed in 1.1 % sodium hypochlorite and gently shaken for 10 min. The pieces were then immersed in 70 % alcohol and washed with sterile distilled water between changes. The rhizomorphs were washed with 3 % hydrogen peroxide for 5 min, rinsed twice with sterile distilled water and dried with sterile paper towels, following the method of Harrigton et al. (1992). Around 15-20 woodchips with mycelium and brown-to-black rhizomorphs were sown in Petri dishes containing BDS (Benomyl-Dicloran-Streptomycin) selective culture medium (Worrall, 1991; Aguín et al., 1998). The dishes were covered with aluminum foil, placed in polypaper bags and incubated at 20 °C in darkness during 4-5 weeks.
To explore the mycoflora population present in soil at the collection sites, the soil dilution PDA-TS (potato-dextrose-agar-tergitol) technique was used (Steiner and Watson 1965). The dishes were kept at a temperature of 22-24 °C for 7 to 10 days, period during which the fungi developed. Isolates were transferred to water-agar medium to be purified using the hyphae-point technique and sown individually in dishes containing water agar and PDA.
Molecular identification. DNA extraction, specific amplification and sequencing
After obtaining pure isolates of the fungi, they were molecularly characterized. DNA extraction was performed using the AxyPrep™ Multisource Genomic DNA Miniprep (Axygen) kit, according to the manufacturer’s instructions. PCR was performed using the ARMEF-3-A (CGT GAY TTY ATC AAG AAC ATG) and ARMEF-R (TAC CCG TTC GGC GAT CAA TCT) primers designed in the amplified region for the Tef-1α gen (translation elongation factor 1α) (Ross-Davis et al., 2012). Amplified reactions were performed in a standard TProfessional thermocycler (Biometra, Germany) using a final volume of 25 µL with the following reagents: Taq DNA Buffer PCR 1X, MgCl2 1 mM, dNTP’s 0.2mM, Platinum Taq DNA polymerase 1U (Invitrogen, USA), 10 µM of each primer and 4 µL of total DNA. The amplification program was as follows: 96 °C for 3 min, 35 cycles at 95 °C for 30 s, 58 °C for 30 s, 72 °C for 45 s, and a final extension at 72 °C for 4 min. To verify the presence of the fragment electrophoresis in 2 % agarose gels stained with ethidium bromide (5 mg L-1) was utilized; the gels were documented using the Gel Doc (Bio-Rad, USA) photo documentation system. The PCR products were purified and sequenced in both directions by the Macrogen Inc. (Seoul, South Korea).
Multiple alignment and phylogenetic analysis
Sequences of the TEF-1a gene in the isolates were edited and manually aligned using BioEdit 7.2.5 software (Hall, 1999). The sequences were compared with those available in the National Center of Biotechnology Information (NCBI) database using the BLAST® algorithm. For the phylogenetic analysis, all the sequences of Armillaria spp. isolates from Michoacan were grouped in a single consensus sequence, and sequences of isolates of 34 Armillaria species characterized worldwide were used, according to the method of Maphosa et al. (2006), Hasegawa et al. (2010), Ross-Davis et al. (2012), Elías-Román et al. (2013) and Guo et al. (2016). Pleurotus ostreatus (NCBI accession number AY883432) was used as a root node outside the group. All the sequences were aligned with the CLUSTAL-W algorithm and analyzed by the Neighbor-joining (NJ) method (Saitou and Nei 1987) using Molecular Evolutionary Genetics Analysis software (MEGA 7). One thousand support value replications (Bootstrap) were used to obtain a consensus tree with a 50 % majority rule (Tamura et al., 2004). For this task, the Kimura 2 Replacement Parameter-2 Model (K2P) was used, according to the method of Hasegawa et al. (2010).
RESULTS AND DISCUSSION
Morphological identification
The strains collected in orchards 1, 2 and 5 (Table 1) from the bark of trees infected with fungal mycelium had a fan-like growth pattern and the rhizomorphs from roots showed a cottony creamy yellow growth in the culture medium. Rhizomorphs with dichotomic growth formed after 20 days and hyphae characteristic of the Armillaria genus formed at 28 days. No basidiocarps were found at the study sites before, during or after the rainy season.
Samples of plant material collected from orchards 3 and 4 did not include Armillaria spp. isolates. The genera Trichoderma sp., Pilatoporus sp., Mucor sp., Penicillium sp., Aspergillus sp. and Fomitopsis sp. were found in the soil isolates from all the sampling sites. Fomitopsis sp. is an associated pathogen that can cause trees to decay and die (Cibrián et al., 2007). In orchard 5, the presence of Fusarium oxysporum was detected, a root pathogen that damages the vascular system.
Molecular identification. Multiple alignment and phylogenetic analysis
The DNA sequences obtained in this study do not have reliable coverage and have not been identified (≥ 99 %) as being one of the known species of Armillaria reported in NCBI; thus it appears to be a new species of the genus. However, the sequences were 99 % similar to those reported as being Armillaria spp. by Elías-Román et al. (2013), based on isolates from the State of Mexico, concluded that root rot in peach trees is caused by a species of the Armillaria genus that has not yet been described. The sequences obtained in this study are kept in the GeneBank with accession numbers KY611443 to KY611450 and are available. These sequences and those reported by Elías-Román et al. (2013) are from the same organism, which belongs to a species not yet described in the literature.
Based on the alignment of 48 sequences of the EF-1α gene from at least 34 species of the Armillaria genus that have been characterized worldwide. Following the results reported by Maphosa et al. (2006), Hasegawa et al. (2010), Ross-Davis et al. (2012), Elías-Román et al. (2013) and Guo et al. (2016), a phylogenetic analysis was performed using the Neighbor-joining algorithm. A 50 % or higher probability consensus tree was obtained in which the Armillaria species were grouped into four main clades (Figure 1). The EF-1a gene was useful for separating all the species. The studied sequences were grouped in the same clade as the species Armillaria mellea, but in different branches (Figure 1). This confirmed that they are new species that appear to be phylogenetically related to A. mellea. However, the phylogenetic analysis, BLAST and the sequence analysis (single-nucleotide polymorphisms, insertions and deletions) all clearly distinguish both organisms as being different species, which agrees with the results reported by Elías-Román et al. (2013) using the isolates obtained in the State of Mexico.
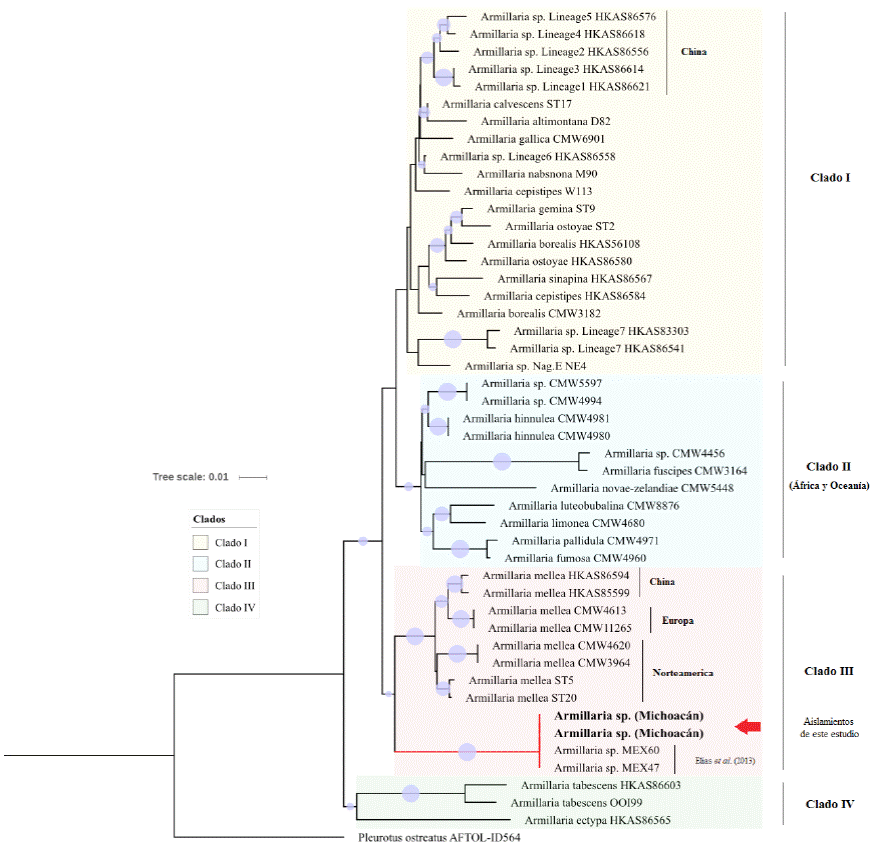
Figure 1 Phylogenetics of Armillaria genus developed using the Neighbor-joining (NJ) algorithm inferred from sequences of the EF-1α gen, including isolates collected in Michoacan, Mexico. A similar topology was obtained using ML and MP. Pleurotus ostreatus was used as a root node outside the group. Bootstrap support values (1000 replications) for NJ (≥ 50%) are shown in the internodes with a blue circle.
In the Armillaria genus, species grouped in the same clade as A. mellea and A. ostoyae are associated with or considered aggressive pathogens that usually cause diseases in various fruit and forest trees, compared with species that are saprophytes or that behave as weak pathogens and that are grouped in the other clades (Ross-Davis et al., 2012; Guo et al., 2016). In this study, the phylogenetic analysis grouped the sequences in the same clade as A. mellea sequences that have been reported in North America, Asia and Europe.
In a parallel study, the cumulative incidence of dieback over three years in orchard 1 was calculated at 21.3 % (145/679 trees) (Rivas-Valencia and Fernández-Montes et al., 2017). This could be interpreted as being an epidemic of great importance, caused by a species of Armillaria closely related to A. mellea.
The information about new Armillaria species in Mexico is based on reports from Alvarado and Blanchette (1994) on native species from forest regions of central Mexico showed that some of the isolates obtained in the state of Puebla were not compatible with reference isolates of species known in North America, which suggests that they belong to a different species. Due to the lack of sequences, it is not known if those isolates belong to the same biological species studied in this research. However, using biotechnological tools and, specifically, DNA sequence data, it is possible to identify the Armillaria species, which is essential to evaluate the threat of disease and its required management.
CONCLUSIONS
Dieback of peach and plum trees at Zinapecuaro county, Michoacan, is associated with the presence of the Armillaria genus. Based on the analysis of DNA sequences, two isolates were identified in Michoacan that differ from those obtained in peach orchards in the State of Mexico. Isolates of Armillaria from Michoacan and the State of Mexico are genetically related to A. mellea, considered to be an aggressive pathogen. No tolerant rootstocks to Armillaria have been identified so far; therefore, it is recommended to continue further studies to find genetic disease resistance as well as improve disease management methods in the orchards.
REFERENCES
Adaskaveg JE, Forster H, Wade L, Thompson DF and Connell JH. 1999. Efficacy of sodium tetrathiocarbonate and propiconazole in managing Armillaria root rot of almond on peach rootstock. Plant Disease 83:240-246. Disponible en línea: http://apsjournals.apsnet.org/doi/pdf/10.1094/PDIS.1999.83.3.240 [ Links ]
Aguín O, Mansilla P, Pintos C and Salinero MC. 1998. La podredumbre blanca de la raíz de la vid. Vida Rural España 47: 1-2. Disponible en línea: http://www.mapama.gob.es/ministerio/pags/biblioteca/revistas/pdf_vrural/Vrural_1998_72_46_47.pdf [ Links ]
Aguín-Casal O, Mansilla-Vazquez JP and Sainz-Oses MJ. 2006. In vitro selection of an effective fungicide against Armillaria mellea and control of white root rot of grapevine in the field. Pest Management. Science. 62, 223-228. http://dx.doi.org/10.1002/ps.1149 [ Links ]
Alvarado-Rosales D and Blanchette RA. 1994. Armilllaria species from forests of central Mexico. Phytopathology 84:1106. [ Links ]
Barnett LH and Hunter BB. 1998. Illustrated genera of imperfect fungi. The American Phytopathologycal Society. St. Paul, Minnesota, USA. 218 pp. [ Links ]
Baumgartner K and Rizzo DM. 2001. Ecology of Armillaria species in mixed-hardwood forests of California. Plant Disease 85: 947-951. https://doi.org/10.1094/PDIS.2001.85.9.947 [ Links ]
Baumgartner K. 2004. Root collar excavation for postinfection control of Armillaria root disease of grapevine. Plant Disease 88:1235-1240. https://apsjournals.apsnet/pdf/10.1094/PDIS.2004.88.11.1235 [ Links ]
Brazee NJ, Hulvey JP and Wick RL. 2011. Evaluation of partial tef1, rpb2, and nLSU sequences for identification Armillaria calvescens and Armillaria gallica from northeastern North America. Fungal Biology 115, 741-749. https://doi.org/10.1016/j.funbio.2011.05.008 [ Links ]
Cai L, Giraud T, Zhang N, Begerow D, Cai G and Shivas RG. 2011. The evolution of species concepts and species recognition criteria in plant pathogenic fungi. Fungal Diversity 50:121-133. Disponible en línea: http://max2.ese.u-psud.fr/publications/Cai_2011.pdf [ Links ]
Cibrián TDD, Alvarado R y García SE (eds). 2007. Enfermedades Forestales de México/Forest Diseases in Mexico. Universidad Autónoma de Chapingo; CONAFOR-SEMARNART, México; Forest Service USDA, EUA; NRCAN Forest Service, Canadá y Comisión Forestal de América del Norte, COFAN, FAO, Chapingo, México. 587 pp. [ Links ]
Domsch KH, Gams W and Anderson TH. 1980. Compendium of Soil Fungi. Academic Press. Great Briain. 859 pp. [ Links ]
Elías-Román RD, Guzmán-Plazola RA, Klopfenstein NB, Alvarado-Rosales D, Calderon-Zavala, G, Mora-Aguilera JA, Kim MS and García-Espinosa R. 2013. Incidence and phylogenetic analyses of Armillaria spp. associated with root disease in peach orchards in the State of Mexico, Mexico. Forest Pathology 43: 390-401. http://dx.doi.org/10.1111/efp.12043 [ Links ]
Fernández-Pavía SP, Gregorio-Cipriano R, Rodríguez-Alvarado G, Fernández-Pavía YL, Mondragón-Flores A, Gómez-Dorantes N, Lozoya-Saldaña H, Rodríguez-Fernández R y Herrera-Camacho J. 2015. Enfermedades de Especies Vegetales en México. Universidad Michoacana de San Nicolás de Hidalgo. Morelia, Mich., México. 425 pp. [ Links ]
Gubler WD. 1992. Armillaria root rot. In: Grape Pest Management Edition 2 Publication #3343 (Flaherty, D.L., Christensen, L., Lanini, W.T., Marois, J.J., Phillips, P.A. and Wilson, L.T., eds), Oakland, CA: University of California, Division of Agriculture and Natural Resources. 92-93 pp. [ Links ]
Guo T, Wang HC, Xue WQ, Zhao J and Yang ZL. 2016. Phylogenetic analyses of Armillaria reveal at least 15 phylogenetic lineages in China, seven of which are associated with cultivated Gastrodia elata. PLoS ONE 11(5): e0154794. Disponible en línea: http://journals.plos.org/plosone/article/file?id=10.1371/journal.pone.0154794&type=printable [ Links ]
Hall TA. 1999. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symposium Series 41:95-98. Disponible en línea: http://brownlab.mbio.ncsu.edu/JWB/papers/1999Hall1.pdf [ Links ]
Harrigton TC, Worall JJ and Baker FA. 1992. Armillaria. In: Methods for Research on Soilborne Phytopathogenic Fungi. APS Press M.N. St. Paul. USA. 81 pp. [ Links ]
Hasegawa E, Yuko O, Tsutomu H and Kikuchi T. 2010. Sequence-based identification of Japanese Armillaria species using the elongation factor-1 alpha gene. Mycologia 102: 898-910. http://dx.doi.org/10.3852/09-238 [ Links ]
Kim MS, Klopfenstein NB, Hanna JW and McDonald GI. 2006. Characterization of North American Armillaria species: genetic relationships determined by ribosomal DNA sequences and AFLP markers Forest Pathology 36:145-164. http://dx.doi.org/10.1111/j.1439-0329.2006.00441.x [ Links ]
Klopfenstein NB, Hanna JW, Cannon PG, Medel-Ortiz R, Alvarado-Rosales D, Lorea-Hernández F, Elías-Román RD and Kim M.S. 2014. First report of the Armillaria root-disease pathogen, Armillaria gallica, associated with several woody hosts in three states of Mexico. Plant Disease 98:1280. http://dx.doi.org/10.1094/PDIS-04-14-0349-PDN [ Links ]
Maphosa L, Wingfield BD, Coetzee MPA, Mwenje E and Wingfield MJ. 2006. Phylogenetic relationships among Armillaria species inferred from partial elongation factor 1-alpha DNA sequence data. Australasian Plant Pathology 35: 513-520. https://doi.org/10.1071/AP06056 [ Links ]
Rachel A. Koch RA, Wilson AW, Séné O, Henkel TW, and Aime MC. 2017. Resolved phylogeny and biogeography of the root pathogen Armillaria and its gasteroid relative, Guyanagaster. BMC Evolutionary Biology 17:33. https://doi.org/10.1186/s12862-017-0877-3 [ Links ]
Redfern DB and Filip GM. 1991. Inoculum and infection. In: Armillaria Root Disease Agriculture Handbook No. 691 (Shaw, C.G., III and Kile, G.A., eds), Washington, DC: United States Department of Agriculture Forest Service. 48-61 pp. [ Links ]
Rivas-Valencia P y Fernández-Montes MR. 2017. Patrón espacial de muerte descendente de durazno ocasionada por Armillaria sp. en Michoacán. Suplemento Revista Mexicana de Fitopatología Volumen 35: S128. Disponible en línea: http://rmf.smf.org.mx/suplemento/Suplemento352017.html [ Links ]
Ross-Davis LA, Hanna WJ, Kim MS and Klopfenstein BN. 2012. Advances toward DNA-based identification and phylogeny of North American Armillaria species using elongation factor-1 alpha gene. Mycoscience 53:161-165. https://doi.org/10.1007/s10267-011-0148-x [ Links ]
Saitou N and Nei M. 1987. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Molecular Biology and Evolution 4:406-25. Disponible en línea: http://www.montefiore.ulg.ac.be/~lwh/IBIOINFO/Saitou-Nei-NJM-87.pdf [ Links ]
Secretaría de Agricultura, Ganadería, Desarrollo Rural, Pesca y Alimentación (SAGARPA). 2015. Anuarios estadísticos de la producción agrícola. Servicio de Información Agroalimentaria y Pesquera (SIAP). México, D. F. http://www.siap.sagarpa.gob.mx (consulta: Enero, 2017). [ Links ]
Secretaría de Agricultura, Ganadería, Desarrollo Rural, Pesca y Alimentación (SAGARPA). Sistema de Información Agroalimentaria de Consulta, 1980-2014 (SIACON). México. 2014. http://www.gob.mx/acciones-y-programas/produccion-agricola-33119 (consulta: Febrero, 2017). [ Links ]
Steiner GW and Watson RD. 1965. Use for Surfactants in the soil dilution and plate count method. Phytopathology 55: 728-730. [ Links ]
Tamura K, Nei M and Kumar S. 2004. Prospects for inferring very large phylogenies by using the neighbor-joining method. Proceedings of the National Academy of Sciences 101: 11030-5 DOI:10.1073/pnas.0404206101 [ Links ]
Valdés M, Córdova J, Valenzuela R y Fierros AM. 2004. Incremento del fitopatógeno Armillaria mellea (Vahl.:Fr.) Karsten en bosque de pino-encino, en relación al grado de disturbio por tratamiento silvícola. Revista Chapingo Serie Ciencias Forestales y del Ambiente 10: 99-103. Disponible en línea: http://www.redalyc.org/articulo.oa?id=62910205 [ Links ]
Villaseñor GLE (editora). 2005. La biodiversidad en Michoacán: Estudio de Estado. 2005. Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, Secretaría de Urbanismo y Medio Ambiente, Universidad Michoacana de San Nicolás de Hidalgo. México. Disponible en línea: http://www.biodiversidad.gob.mx/region/EEB/pdf/EE_MICHOACAN_2005.pdf [ Links ]
Worrall JJ. 1991. Media for selective isolation of hymenomycetes. Mycologia 83: 296-302. DOI: 10.2307/3759989 [ Links ]
Acknowledgments
The authors wish to thank Ing. Fortino Mendoza, Antonio Durán, Rafael Mendoza, Gerardo Mendoza and Jesús Guerrero for their help in conducting this study, and are grateful for the partial funding provided by Fondos Fiscales INIFAP Convocatoria 2015-2016, and SAGARPA/COFUPRO funds 2016 for the project “Generación de nuevas variedades de durazno en México” DF1600000799.
Received: February 17, 2017; Accepted: June 30, 2017











 texto en
texto en 



