Ovarian cancer includes the epithelial cancer which develops on the ovary surface. It is the most common type of cancer and represents 85% of the cases, especially in women between 45 to 59 years old. The germ cell type which starts in the egg (immature germination cell) represents 10% and it is more frequent in less than 30 year old young women (figure 1).
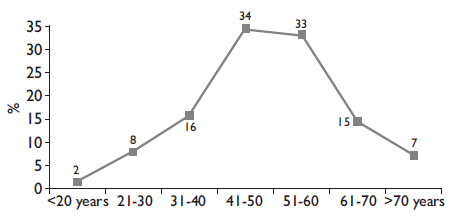
Source: cases registry, "Programa Cáncer de Ovario 2011-2015", INCan * total of cases: 868 newly diagnosed
Figure I Distribution of patients with epithelial ovarian cancer by age*
The stromal tumors are very scarce (barely 2-3%) and affect the ovarian tissue, but they can be functional and produce hormones. There can also be ovary sarcomas and neuroendocrine tumors, which are very rare malignancies.1,2
Epithelial ovarian cancer (EOC) develops from a complex cystic formation (solid and liquid) of the epithelial cells which cover the ovaries or from a cancer in situ of the Fallopian tube fimbria. The epithelial histology can be high-grade papillary serous, low-grade papillary serous, mucinous, endometrioid, and clear-cell. The most frequent subtype which causes the highest rate of mortality is the high-grade papillary serous type; followed by the endometrioid type.3 The risk of epithelial ovarian cancer is high, mostly during menopause.
EOC is the most common gynecologic neoplasia and the first gynecological cancer cause of death.4 Worldwide, 238 719 new cases are recorded every year; in Mexico around 4 000 new cases are estimated each year.5 This neoplasia is underestimated in comparison to other widely-known cancer, such as breast and cervical cancer.
EOC has non-specific symptoms which might be confused with irritable bowel syndrome or non-specific pelvic discomforts; many female patients go first to a gastroenterologist or a gynecologist not specialized in oncology,6,7 this means 6 to 12 months can pass before establishing an accurate diagnosis.8 Many of these patients may have incomplete and inappropriate surgeries; therefore, they must have a new one.9,10
If a patient undergoes a surgery performed by a doctor who is not an oncologist, she has an adverse prognosis factor of survival because her risk of having an incomplete surgery increases four times.10 Every patient with a complex cystic image (mixed with solid and liquid component) in one or both ovaries documented by ultrasonography (US), computed tomography (CT), or with an adnexal lump should be admitted in an institution with oncological attention suspecting cancer. Unfortunately, cancer centers in Mexico demand a pathology report which confirms diagnosis of cancer in order the patient to be admitted. This is not possible in the case of EOC because the diagnosis is surgical.
It is also essential the implementation of educational programs taken physically and on line, as well as radiology programs for evaluation of ovarian cystic images that examine the cyst size, the thickness of the capsule, presence or absence of papillary projections and presence of septa inside the cyst.11
The primary care physicians, general physicians, family physicians, general surgeons, and specially gynecologists must be supported by a radiologist with experience in transvaginal ultrasound (TVUS) in order to have a better evaluation of ovarian cysts.
Risk to develop EOC
The average risk to develop EOC in the general population is 1.8%. When there is an isolated family background of ovarian or breast cancer, this risk rises up to 5%, which is considered an intermediate risk.
When the hereditary pattern consists of two direct line family members with ovarian or breast cancer and it presents the BRCA gene 1 or 2 mutation, the risk in BRCA 1 case increases from 35% to 45% more than in general population; in BRCA 2, the risk of developing ovarian cancer increases from 15 to 25%. This is classified as high risk.12
If the patient has two direct line relatives with ovarian or breast cancer and the BRCA gene 1 and 2 mutations have not been determined or, if the test results have been negative for these mutations, the patient is considered high risk because there can be deletions and genes associated to BRCA 1 and 2 mutated that may not be shown.13
Other risk factors are infertility, which increases the relative risk 2.6 times more than general population;14 policystic ovary syndrome increases 2.56 times;15 endometriosis 2.04-3.05 times;16 hormone replacement therapy (for three years average or more) increases the relative risk (RR) in 1.41 times;17 smoking, 2.1 times more than the general population;18 and intrauterine device with a RR of 1.76.19
Protective factors
The use of oral contraceptives for three years decreases the risk 0.73 times more than in general population.20 Breastfeeding over twelve months has a protective factor of 0.72;21 pregnancy has 0.6; and bilateral tubal occlusion has a protective factor of 0.69.22
Prevention of EOC
The prevention applies to cases of family cancer, women who have BRCA 1 and 2 gene mutation, and who are considered in high risk to develop EOC. Nowadays, it is calculated that this mutation represents 10% of all EOC cases, although such figure may be higher; however a bilateral salpingo-oophorectomy (BSO) is recommended. Chemoprevention could be given in contraceptives, but it is not a universal recommendation.23
EOC early detection
Screening studies performed in ovarian cancer so far have been very controversial because they were designed twenty years ago. Then, there was scarce knowledge about the histological, molecular subtypes and their biological behavior. These studies include five histological types with different molecular behavior and cellular growth.3
Likewise, the pelvic and transvaginal ultrasound (PUS and TVUS, respectively) are also used, but the latter is more useful. There are numerous screening studies, but eight of them are the most important. PLCO (Program of Lung, Colon and Ovarian Cancer Screening) analyzed 7 237 women between 55 to 74 years old; 34 253 of them had CA 125 test for six years plus TVUS for four years; 34 304 were in observation with a 13-year follow up. This study revealed that the screening with CA 125 and TVUS does not achieve a mortality reduction, compared to the observation group. The negative result was obvious because its design was created over 20 years ago and the most common of these five types is the high-grade serous papillary. This type is known for presenting early clinical manifestations as well as cellomic dissemination to the peritoneum which covers the bowels. It can also appear within six months, so the performance of the TVUS study every year is useless.24
In spite of this, a Japanese trial followed the same methodology performing US every year (in this case was pelvic), but there was a bigger proportion of stage I with no statistical significance.25
The results of the University of Kentucky were positive for the screening group with TVUS every six months to find stages I, II with better survival (SV) for the screening group: 84.6 vs. 53.7%. Nevertheless, this trial comes from only one center which is very specialized in evaluation of ovary cysts and, therefore, is hard to reproduce.26
The NOCEDEP trial -focused on high-risk women to establish an early stage diagnosis- was negative.27 The UKFOKCS also analyzed high-risk women and suggests as performing a BSO, the BRCA 1 and 2 gene mutation test, and just a screening test on women who do not want surgery.28
ROC, UKCTOCS (not conclusive yet), and ROCA use algorithms to diagnose early ovarian cancer. They are based on the monitoring of CA 125 when its baseline value is 30 u/ml or higher. They check the values every 3 months, in addition to the performance of TVUS. However, ROC and ROCA are very promising.29-31
Keeping a record with a symptom index (non-specific abdominal pain, sensation of abdominal distention, constipation alternating with diarrhea, rectal tenesmus, pelvic sore, and feeling of early satiety) did not reach a predictive value of 10. Even so, it is suggested that the patient should have a TVUS every 6 months and the measuring of CA 125 in case of having these symptoms more than 12 times a month.32
Dr. Kurman stated that in order to identify ovarian cancer in early stages it is central to focus on the tumor size rather than on the clinical stage. Even though 75% of the patients are diagnosed in advanced stages (III and IV) around the world, that such figure may be higher in less developed countries. A patient in IIIa and IIIb is different from a patient in IIIC because the first two stages imply less tumor volume and higher possibilities of performing an exploratory laparotomy. That will also have a higher probability of achieving an optimal cytoreduction. This is strictly defined as the absence of visible macroscopic disease or, at least, disease with implants <.5 cm or <1cm, with a better prognosis, which may be performed in patients who, at the end, achieve implants > 1cm. Patients in IIIc have a shorter survival than patients in IIIa and IIIB; thus, the aim of establishing a diagnosis with advanced disease is preferable to try to establish an early diagnose, which is rather difficult to achieve, especially the papillary serous, high grade ovarian cancer, as well as the endometrioid with very early dissemination.33
Although, current evidence level to perform screening in ovarian cancer is weak; the National Comprehensive Cancer Network (NCCN) guidelines (2009) recommends applying to:
Women with high risk of developing ovarian cancer; that is to say, women with breast or ovarian cancer background or who have BRCA gene mutation, TVUS and CA 125 tests every six months;
Disease which began at 35 years or 5 to 10 years before the age the close relative was diagnosed with ovarian or breast cancer;
Women with BRCA mutation to whom are strongly recommended prophylactic BSO after meeting the desire of having a baby.34
Surgical staging of ovarian cancer, treatment with surgery, and chemotherapy
EOC is classified by stages:
Stages I and II, confined to the pelvis,
Stage III is sub-classified in IIIA (retroperitoneal lymph nodes and/or microscopic extra-pelvic peritoneal disease) and IIIB (microscopic extra-pelvic peritoneal disease with metastases < 2 cm; it is also possible to find retroperitoneal lymph nodes and disease disseminating to the liver capsule or spleen),
IIIC, macroscopic disease extra-pelvic peritoneal disease > 2cm, like in IIIB where it is possible to find retroperitoneal lymph nodes and extension to the liver capsule and spleen,
IVA, with pleural effusion and positive cytology,
IVB, metastases in hepatic, splenic parenchyma, in extra-abdominal organs, affection of inguinal nodes or affected nodes in another site35
Patients should be attended by a multidisciplinary team (gynecologist, oncology surgeon, medical oncologist, radiologist, and pathologist) in order to standardize the initial treatment (surgery or chemotherapy).
The diagnosis of advanced disease (stages III and IV) worldwide is 75%,36 but the survival for stage III is different, depending on the clinical stage IIIA, IIIB or IIIC. For stage IIIA, survival is 60% to five years; while for IIIB is 50 and 35% to IIIC.37
On the whole, our patients are at stages IIIc and IV. They represent 85% of all the patients who visit the doctor for the first time. They already have extensive disease in a clinical entity known as abdominal carcinomatosis, that is to say, disease in the abdominal wall and in the peritoneal surface (oment take), which makes impossible to perform a first surgery. Therefore, the patients are given induction chemotherapy (neoadjuvant chemotherapy) during three cycles; then, they will be submitted to a surgery known as interval debulking surgery, and then continue with three additional cycles of adjuvant chemotherapy, for a total of six cycles.38
Conversely, patients in stages I, IIa, IIb (and a few in IIIc) are treated with exploratory surgery known as exploratory laparotomy, which is cytoreductive and will stage the disease. It consists of performing panhysterectomy, omentectomy, pelvic and para-aortic lymphadenectomy, and cheking parietocolic gutters, pelvic floor, and diaphragmatic dome.
Surgery begins with an incision from the xiphoid appendix until the symphysis pubis. The purpose is to remove the visible disease from the abdominal and pelvic cavity where the prognosis is much more favorable. If that is not possible, the smaller the implants which remain, the better. The implants are measured in ≤.5 cm, >.5 cm, < 1cm, >1cm, and <2 cm.39
After surgery, patients receive six cycles of adjuvant chemotherapy. The standard chemotherapy is based on six cycles of carboplatin + paclitaxel. Every cycle is administered every three weeks.40,41 It is also possible to administer a weekly regimen of paclitaxel with the carboplatin administered every three weeks because this is a well-tolerated regimen.42 If the patient presents residual disease (implants) ≥1 cm, it is possible to add bevacizumab, a monoclonal antibody targeted to the vascular endothelial growth factor. This is administered during chemotherapy and it will be also indicated as maintenance treatment.43
As has been stated above, patients with EOC have a high relapse rate, even 75%, especially in the more advanced stages (IIIc and IV). Obviously, it is important to consider the disease free interval. Patients who relapse in less than six months or progress during chemotherapy are classified as resistant and refractory. They also have poor prognosis. The patients who relapse between 6 and 12 months have an intermediate prognosis; and the ones who relapse after 12 months have better prognosis. Overall, patients who relapse after 6 months may also be treated with the same regimen of carboplatin + paclitaxel. If the patient had neuropathy symptoms, secondary to paclitaxel, the rescue regimen may be carboplatin + gemcitabine.
Patients who have BRCA 1 and 2 mutations are considered more sensitive to chemotherapy; thus the use of PARP (Poly ADP-ribose Polymerase) inhibitors like olaparib may be assessed in their clinical condition, after chemotherapy as maintenance therapy for a year because PARP inhibitors have been approved for patients in the firs relapse occurred after six months.44
The issue is that patients with EOC and who relapse for the first time remain disease-free approximately for two years or more and relapse again in a shorter time; so, the time to relapse begin to diminish. The free-progression median is 2.5 years and the survival median with several treatment lines is four years.45
At Instituto Nacional de Cancerología we have a prevention, timely detection, and access to treatment program since 2011. We have received 1 758 women, 25% of them did not have cancer. There were 1 319 patients with ovarian cancer, 1 121 of them with epithelial type (EOC), 672 of them did not receive previous treatment. Although data is not accurate, we have estimated that the overall survival is 5.1 years. Nowadays, we want to run clinical trials including the use of immune action drugs like metformin, COX2 inhibitors, and antibodies like anti PDL1 and 2, anti CTLA4 targeted to proteins which are expressed on the surface of the tumor cells.
This is a very promising area to foster the immune response of the patient against the tumor or the sub-clinic disease in order to achieve more sustained responses with chemotherapy, avoid resistance to drugs, and prevent recurrence.
Discussion
Since EOC is the most common of all the ovarian cancers, our efforts must be focused on the epithelial type. Thus, we have to consider it as a neoplasm whose clinical behavior is not only confined to the pelvis but to gastrointestinal symptoms like colitis, which now are more noticeable for primary care physicians, especially gastroenterologists, general surgeons, and gynecologists.
Thus, it is imperative to include the cancer diseases in women in the Secretaría de Salud prevention programs in order to make women to be aware that their ovaries may develop cancer, especially in women who are going through menopause or whose direct-lined female relatives have had breast or ovarian cancer.
Besides having a pap smear test, which women think identify any type of gynecological cancer, it is crucial to detect infertility, endometriosis, polycystic ovaries, and use of hormonal replacement therapy.
Women who have BRCA 1 and 2 mutations merit that their direct-lined female relatives receive genetic advice, be educated on preventive measures, and be attended by a multidisciplinary team which includes a genetist and a psychologist to discuss issues like satisfied parity and the acceptance of bilateral salpingo-oophorectomy.
Nevertheless, the early detection by TVUS plus CA 125 tests is not recognized as useful because the screening tests worldwide have been negative or not conclusive. Thus, women should be educated about the risk factors to develop this neoplasm, especially if they have breast or ovarian cancer family background. Likewise, if they have symptoms like persistent colitis; that is to say vague abdominal discomfort, abdominal distention sensation, constipation alternating with diarrhea, rectal tenesmus (frequently regarded as irritable bowel) and persistent urinary discomfort. All of those symptoms must be a reason to perform a TVUS plus CA 125 test.
CA 125 may have high values for different reasons, not only ovarian cancer. Nevertheless, women who have high levels of this antigen must be monitored preferably every three months, even if there is no visible disease at pelvis.
The objective in EOC is establishing a diagnosis of less advanced disease because its early dissemination towards peritoneum is the most common, even with adnexal cyst lesions, which may be small, especially in the serosa papillary type. Diagnosis reached even in the earliest stage III (IIIA or IIIB) is better than stage IIIC, where tumor burden and peritoneal carcinomatosis are generally found. It should be considered that more than clinical stage it is a matter of tumor volume, and that our aim is to diagnose it when there is less tumor volume in order to ensure successful surgeries which remove all the disease and only remain the microscopic disease or implants ≤5 cm, <1 cm, which is better than leaving implants of 2 cm or more. Identifying mucinous and clear cell type cancers are much more difficult to detect, even when they have not come up to the pelvis.
It is essential that if a patient goes to the pre-consultation services of the cancer institutions with an ultrasound or tomography which shows an adnexal (cyst and solid) tumor, she should be admitted without being required a neoplasm report. That would prevent that the patient be treated by non-oncologist surgeons and need to undergo a new surgery, not to mention the time wasted (from 6 to 12 months), while the disease progress towards an advanced stage.
It is also imperative to implement educative programs to identify the ovarian cyst lesions as well as gathering radiologic data by means of ultrasound which confirms the presence of cancer, based on the radiologists' expertise.
Surgery is the key treatment for EOC and includes the ovary, Fallopian tubes, and uterus resection (panhysterectomy), besides the pelvic nodes and para-aortic chains resection. Likewise, it includes the omentum resection, with an incision from the xiphoid appendix until the symphysis pubis which enables an appropriate vision of the abdominal cavity and a revision of the diaphragmatic domes, parietocolic gutters and pelvic floor.
Since EOC is a very sensitive neoplasm to chemotherapy is also treated with antibodies like bevacizumab as maintenance therapy for patients with residual disease (implants > 1 cm) after surgery and PARP inhibitors, such as olaparib. This is only recommended to women who have BRCA 1 and 2 mutations, responsible of the DNA reparation. However, its use is limited to patients who have experienced their first relapse.
Unfortunately, EOC has a very high recurrence pattern. The time to relapse is crucial for the prognosis; so it is better when it occurs after more than 12 months, when it may be capable of a new surgery, followed by chemotherapy.
Overall survival of advanced ovarian cancer has reached 4.9 years. During this time, it is possible that the patients have received several chemotherapy regimens (one to six lines). This may weaken the patient and shorten the free-disease intervals; thus, the disease may become more resistant.
As the relapse pattern is very high is imperative to implement protocols based on immune-oncologic treatments which are promising because they avoid the patients to undergo several lines of chemotherapy, which result in a higher resistant to drugs. Such drugs improve the immunologic response and enable that the cancer stem cell be identified by dendritic cells, by CD4 and CD8 T cells, and by T cell natural killer, where anti PLD1 and 2 as well as anti CTLA4 antibodies are used along with other drugs like metformin, COX2 inhibitors, among others, plus the conventional chemotherapy to achieve longer free-disease periods and relapses prevention.
Conclusions
The most common type of ovarian cancer is epithelial; thus, it deserves all our attention. Eight women die daily from it, so it is more lethal than the breast or cervical cancer. Therefore, women and physicians should be alert to identify persistent gastrointestinal symptoms, which are the most common in the clinical manifestation of the ovarian cancer.
Regarding its dissemination pattern, our objectives must be establishing a diagnosis in less advanced stages and warrantee that the patient be admitted in cancer centers only with and ultrasound or tomography which shows suspicion of ovarian cancer. This is important because as the diagnosis is surgical, we need to avoid the patient visit several hospitals and physicians before confirming the diagnosis, which also implies the disease progression (advanced stages and extensive tumor volume).
Even though the suitable surgical treatment, chemotherapy, and molecular therapy have achieved a 50% survival in five years, we believe we could obtained better results regarding the immunogenic nature of the ovarian cancer. In other words, as ovarian cancer is an immunogenic neoplasm is capable of being treated with a protocol based on immune-oncologic therapy.











 nueva página del texto (beta)
nueva página del texto (beta)


