Three decades ago it was estimated that in the United States over 30% of cancers could be prevented by modification of dietary habits.1 In 2007, the World Cancer Research Fund and the American Institute for Cancer Research (WCRF/AICR) conducted an extensive literature review of epidemiological studies for dietary exposures in relation to cancer risks: Second Expert Report (SER).2 The report led to the formulation of cancer prevention recommendations on food, nutrition and physical activity, based on epidemiological evidence linking dietary or nutritional components with individual cancer risks. Some dietary factors are believed to play a direct role in cancer prevention; while others are more likely to act through obesity-related mechanisms.2 Despite the growing body of evidence, uncertainty remains for some foods and nutrients; subsequent updates of the 2007 report (Continuous Update Projects, CUP) have further reviewed the evidence for individual cancer sites.3 The aim of this review was to combine the evidence for individual cancers, discuss and update existing data for the dietary factors rated as having strong (convincing or probable) or limited-suggestive evidence in relation to cancer prevention for at least one cancer. In the present revision recent reviews and key papers were searched for in PubMed with relevant keywords (cancer site/exposure) since the year when the relevant updated report was published, until June 2015. However, this paper did not involve a full systematic review. The review focuses on thirteen cancers with the highest incidence in men and women worldwide listed by Globocan 20124 breast, prostate, lung, colorectal, cervix uteri, stomach, liver, corpus uteri, ovary, oesophagus, bladder, kidney, pancreas. Cancers of non-Hodgkin's lymphoma and leukaemia were excluded due to limited evidence for individual cancers; the WCRF/AICR report considered them jointly and concluded that more research is needed in order to be able to draw any conclusions.2
Foods items that presented a protective effect in more than one cancer site were: fruits, vegetables, coffee and milk (figure 1). Consequently, nutrients for which the main dietary sources are fruit, vegetables or milk had an inverse association with cancer, including foods containing dietary fibre, selenium, calcium, some carotenoids, vitamins C, D, E, B6, folates and flavonoids.
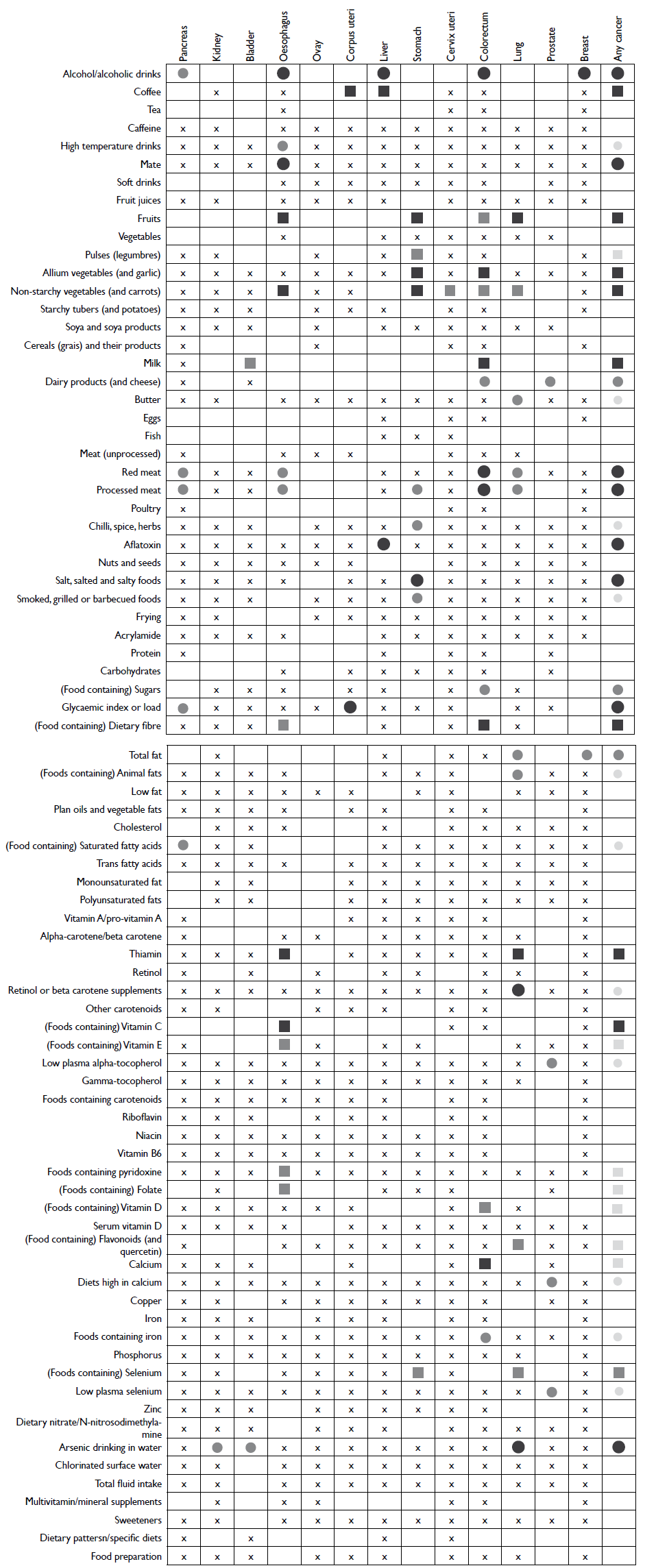
Circles indicate a positive and squares a negative association. Shape size indicates the strength of an association for individual cancers.
For 'any cancer' row the shape size is related to the numbers of cancers that exhibited strong or suggestive association with an exposure of interest.
Empty cells indicate limited evidence for each exposure to draw any conclusion in relation to the cancer of interests. The cross signalises lack of sufficient number of studies to study the association.
Figure I Summary of the evidence evaluated by WCRF / AICR for each cancer individually and the summary for any cancer in relation to dietary exposures that were evaluated in the expert report2
Food items that presented an increased risk at several cancers sites were: alcoholic drinks, red and processed meat. Foods rich in animal fat, saturated fatty acids and iron were correspondingly positively related to cancer. Other factors may also play a role as a cancer trigger, such as food preparation (smoking, grilling or barbecuing), the use of partial hydrogenation of vegetable oils (shortenings containing trans fatty acids), dietary habits (salt addition), storage (aflatoxin), sourcing (arsenic in water) and preservation (salted foods). In addition, foods high in sugar or high glycaemic load have also been positively related to some cancers.
Foods and nutrients with a potential cancer protective effect
Fruits, vegetables and their constituents
There is epidemiologic evidence supporting inverse associations between fruits and/or vegetables intake and several cancers risk.5 According to WCRF/AICR, fruits were inversely associated with oesophageal cancer (based on case-control studies), stomach cancer (based on cohort studies) and lung cancer (based on cohort studies) (table I). There was also a suggestive, yet inconsistent, evidence for a protective role of fruits intake against colorectal cancer. Vegetables were also negatively linked to colorectal and lung cancers; however most of the evidence was rated as limited-suggestive. A non-significantly decreased risk of oesophageal and stomach cancers was observed for intake of non-starchy vegetables (including green, leafy, cruciferous and allium vegetables) suggesting a strong probable association (table I).6-29
Table I Relative risks (RR) and 95% confidence intervals (95%CI) for individual cancer sites in relation to food items that exhibited strong (convincing or probable) or limited-suggestive effect according to the Second Expert Report,2 its updates or most recent meta-analyses
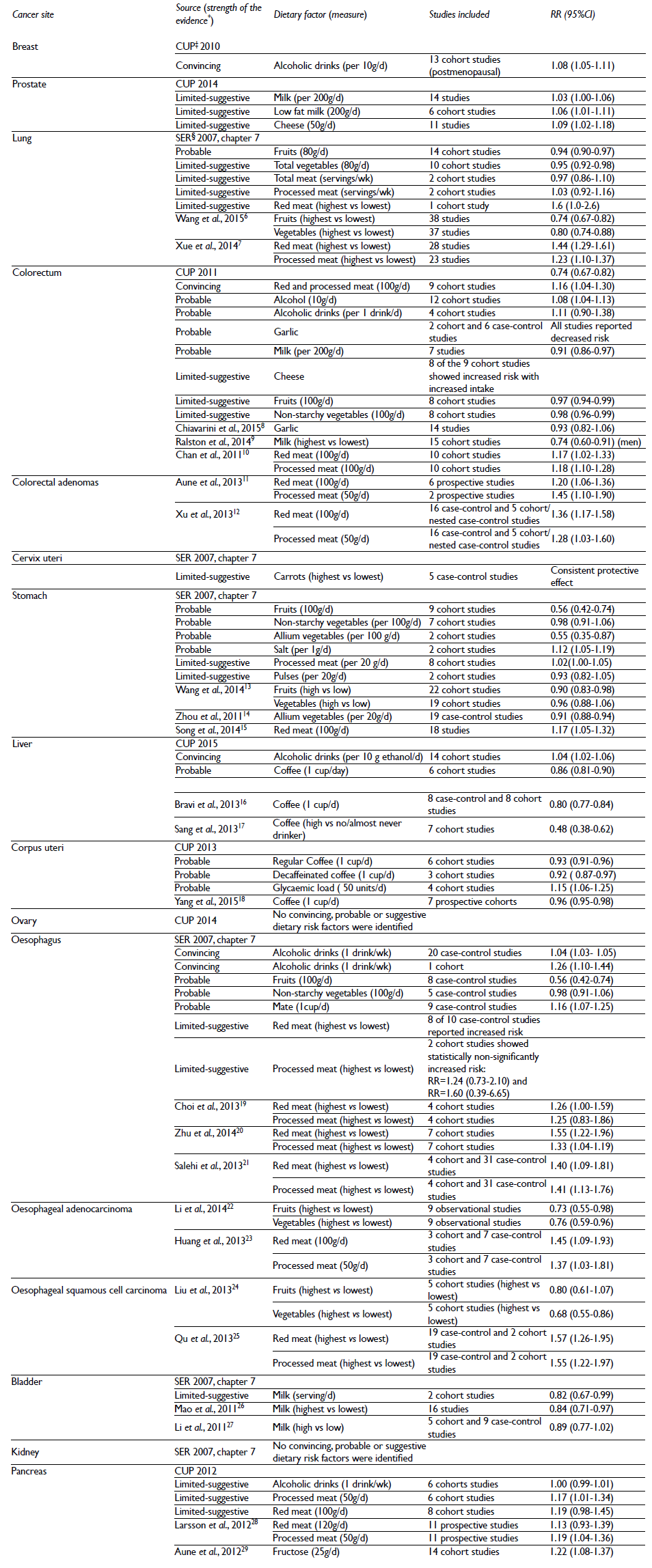
* Convincing evidence (evidence strong enough to support a judgment of a convincing effect or causal relationship, which justifies goals and recommendations, requires RCT evidence), probable evidence (evidence strong enough to support a judgment of a probable effect or causal relationship), limited-suggestive evidence (evidence that is too limited to permit a probable or convincing judgement, but where there is evidence suggestive of a direction of effect).
‡ CUP: Continuous Update Project.3
§ SER: Second Expert Report 2007.2
Note: for some dietary factors meta-analysis of existing studies was not possible and a conclusive expert summary is provided here
More recent data support some of these associations but not all. A meta-analysis of five cohort studies suggested an inverse association of fruits and vegetables with oesophageal cancer risk (RRfruits=0.80; 95%CI 0.61-1.07 and RRvegetables=0.68; 95%CI 0.55-0.86).24 Similar results were obtained based on nine cohorts for oesophageal adenocarcinoma (RRfruits=0.73; 95%CI 0.55-0.98 and RRvegetables=0.76, 95%CI 0.59-0.96)22 and for lung cancer (RRfruits=0.80; 95%CI 0.74-0.88 and RRvegetables=0.74; 95%CI 0.67-0.82 and based on 30 case-control and cohort studies;6 however, divergent results were observed in the subgroup analysis by sex for vegetables, possibly due to confounding by smoking. In a recent meta-analysis of cohort studies a significant reduction in stomach cancer risk for fruits (RR=0.90; 95%CI 0.83-0.98, 22 studies) but not vegetables intake (RR=0.96; 95%CI 0.88-1.06, 19 studies).13 For colorectal cancers, recent results from three additional individual cohorts did not support a clear association with fruits,30 vegetables31 and fruits or vegetables.32
Some individual vegetables were also listed by the report to have limited-suggestive protective effect (table I). Carrots were the only factor that showed consistent protective evidence against cervical cancer. Allium vegetables were linked to lower risk of both stomach14 and colorectal cancers.3 Based on 2011 CUP, garlic was indicated as a probable colorectal cancer protective vegetable; two cohort and six case-control studies reported a non-significantly decreased risk (table I). However, in recent meta-analysis no significant association for garlic and colorectal cancers was observed (OR=0.93; 95%CI 0.82-1.06).8
In the large European Prospective Investigation into Cancer and Nutrition (EPIC) of 10 European countries and close to 500 000 subjects analyses for fruit intake and total cancer risk showed a borderline protective effect (HR=0.99; 95%CI 0.98-1.00, per 100g/d).33 For vegetables this effect was slightly stronger (HR=0.98; 95%CI 0.97-0.99). When cancers were considered separately, significant inverse trends were observed only for fruit but not vegetable intake and oesophageal and lung (in smokers only) cancers.34 A significant trend across quintiles was observed for combined fruit and vegetable intake for the risk of colorectal cancer (RR for highest vs lowest quintile=0.86; 95%CI 0.75-1.00; P-trend=0.04).34
Similarly, foods containing carotenoids (lung) or beta carotene (oesophagus), vitamin C (oesophagus), selenium (lung, stomach) and dietary fibre (colorectum, oesophagus) appeared as cancer protective food constituents (figure 1). A meta-analysis of prospective cohorts indicated an inverse correlation for carotenoidrich vegetables with lung cancer risk (RR=0.79; 95%CI 0.71-0.87).35 Selenium, carotenoids and vitamin C, but also flavonoids and vitamin E, are recognized antioxidants that naturally occur in fruits and vegetables. These micronutrients may play a protective role on cancer by trapping free radicals, preventing lipid oxidation and protecting against DNA damage.36 Epidemiological evidence supports a protective role by dietary fibre against colorectal and oesophageal cancers (figure 1). Dietary fibre decreases transit time and dilutes potentially toxic substances,37 limiting exposure of gastrointestinal tract to their possible carcinogenic effects. Another important cancer preventive characteristic of fibre could be its effect on modifying gut microbiota and alteration of microbial metabolites.37 For example, high fibre intake may increase production of short chain fatty acids with antiapoptotic properties. In addition, binding or diluting bile acids may reduce the potential carcinogenic effect of their metabolites.37 It has been suggested that the source of fibre may have divergent effects. For colorectal cancers, a meta-analysis of twenty-five prospective studies indicated a significant inverse association only for fibre coming from cereals (RR=0.90; 95%CI 0.83-0.97) or whole grains (RR=0.83; 95%CI 0.78-0.89), but not fruit, vegetable or legume fibre.38 However, as indicated by the expert report, no conclusion could be drawn based on the existing epidemiological evidence for cereal and grain products and cancer risk.2
A controversy pertains to the use of nutritional supplements as cancer-preventive measure. Epidemiological evidence indicates beta-carotene/retinol supplementation as risk factors during supplementation for lung cancer in a specific population of heavy smokers or subjects exposed to asbestos.39,40 A meta-analysis of randomized controlled trials concluded that there is no clinical evidence to support preventive capacity of antioxidant supplements on cancer.41 Multivitamins were evaluated by WCRF/AICR for several other cancer sites (figure 1), but no sufficient evidence was present to draw any conclusion.
Coffee
Substantial amount of epidemiological evidence existed to suggest a strong probable dose-response relationship between coffee and endometrial and liver cancers (table I). Both regular and decaffeinated coffee showed a protective effect against cancer development in endometrium. Three recent meta-analyses of observational studies confirmed this association.18,42,43 For liver cancers, meta-analysis based on six studies indicated 14% reduced risk with regular coffee intake (table I). Further three individually published meta-analyses found even stronger protective effect,16,17,44 while in the EPIC cohort a 72% risk reduction for hepatocellular carcinoma (HCC) for highest vs lowest quintile of regular coffee drinkers was observed (HR=0.28; 95%CI 0.16-0.50).45 A divergent effect in subgroup analyses was observed: significantly inverse association was observed only for caffeinated coffee. Considering combined cancer sites a meta-analysis indicated that intake of regular coffee may significantly reduce the risk of total cancers (RR=0.97; 95%CI 0.96-0.98, per one cup/day).46
The probable cancer protective effect of coffee may be attributed to caffeine but also other bioactive coffee components (e.g. chlorogenic acid, polyphenols and their metabolites).47 Coffee constituents may have antioxidant properties, play a role in the regulation of DNA repair, apoptosis and inflammation,47 have effects on hormonal activity,48-50 glucose tolerance51 and circulating levels of immune and inflammatory markers.52
Milk
Milk consumption has been shown to have a protective effect on colorectal and bladder cancers (table I). For colorectal cancer the inverse association for milk was confirmed in four meta- or pooled-analyses of observational studies.9,53-55 Higher calcium and vitamin D intake with milk may partly explain these findings by their action on inhibiting colonic neoplasia.56 Indeed, both dietary calcium and foods containing vitamin D had a protective effect for colorectal cancer (figure 1). Meta-analysis for both dietary calcium (RR=0.94, 95%CI 0.93-0.96; per 200 mg/day, five studies) and vitamin D (RR=0.95, 95%CI 0.93-0.98; per 100 IU/d, ten studies) showed reduced risk of colorectal cancer.3 For bladder cancer a recent meta-analysis of sixteen studies supported a reduced risk with high milk intake (SRR=0.84; 95%CI 0.71-0.97).26 However, these results were not confirmed by another meta-analysis.27 Stronger heterogeneity in case-control studies and male subset and differences by geographic region were observed.
Dietary calcium together with vitamin D and other bioactive milk components (e.g. lactoferrin, glycans) may mediate the protective effect of milk for some cancers through their antiproliferative, anti-inflammatory, anti-viral and antimicrobial properties.56,57 However, for some other cancer sites some milk constituents may have an adverse effect. There was a limited-suggestive positive association for low fat milk and cheese and the risk of prostate cancer (table I) . In a recent study from EPIC, high dairy, milk, cheese and calcium, vitamin D, fat and protein coming from dairy but no other sources were all positively associated with liver cancer (HCC) risk.58 This could be related to the effect of high calcium intake on lowering circulating vitamin D59 or milk on increasing circulating Insulin-like growth factor 1 (IGF-1) levels associated with both HCC and prostate cancers.58,60 Interestingly, high IGF-1 levels were also linked to increased colorectal cancer risk,61 while milk had a protective effect, suggesting a more complex role of milk constituents on carcinogenesis processes at different sites.
Foods and nutrients with a potential cancer promotive effect
Alcoholic drinks
Alcohol is an established independent risk factor for cancers in liver (HCC), female breast, colorectal, oesophagus, oral cavity, pharynx and larynx.62 Based on fourteen cohorts, the estimate from a meta-analysis for liver cancers showed a 4% risk increase (per 10g/day = approximately 1 alcoholic drink) (table I). Chronic alcohol consumption may lead to systemic inflammation and metabolic dysregulation, leading to liver damage and progression to cirrhosis, key risk factors for liver cancers. Twenty case-control studies showed a 4% increased risk of oesophagus cancer for an increment of one drink a week. The estimate for one cohort analysed separately was stronger (table I). Dose-response analyses for twelve cohorts showed an 8% increased risk of colorectal cancer, per 10g/day of ethanol, while each additional alcoholic drink a day was associated with a non-significant 11% increased risk (table I). The results for breast cancer are partly dependent on menopausal or hormonal status.63 Meta-analysis of thirteen studies indicated a dose-response 8% increased risk of postmenopausal breast cancer per 10g/day increase in alcohol consumption (table I). A dose-response meta-analysis of ten cohort and case-control studies indicated that ER- breast cancers may be less dependent on alcohol exposure than the hormone-positive cancers.64 However, recent data from the EPIC cohort reported a linear dose response relationship for alcohol intake and breast cancer with a 4.2% (95%CI 2.7%-5.8%, per 10 g/d) risk increase. Positive association was observed for both pre- and postmenopausal women and all types of receptor status. Exposure prior to first term pregnancy was related to a higher risk.65 Only heavy drinking was associated with an increased pancreatic cancer risk (RR=1.30; 95%CI 1.09-1.54), but no clear dose-response was observed (table I). However, for some cancers adverse effect of alcohol consumption was observed also for light drinking (≤12.5 g ethanol; ≤1 drink).66
According to the International Agency for Research on Cancer (IARC) alcohol is classified as a group 1 carcinogen to humans.67 Multiple biological mechanisms are proposed for the role of ethanol in the process of carcinogenesis. Ethanol and its intermediate metabolites demonstrated direct carcinogenic and/or genotoxic properties. Ethanol modulates activity of enzymes responsible for its detoxification and may act as a solvent for other carcinogenic molecules improving their cellular infiltration.68 Ethanol interacts with folate and tobacco affecting methylation processes and/or DNA modification by promoting genetic mutations.69 Ethanol is also a systemic pro-oxidant, leading to lipid peroxidation, production of prostaglandins, generation of free radicals and modulation of cellular regeneration.68
Animal products: red and processed meat and products high in fat
High intake of red or processed meat was associated with cancers in several sites (table I). Convincing and generally consistent evidence existed for increased risk of colorectal cancer. In dose-response analysis from nine cohort studies, 16% increased risk for each 100g/d increase in red and processed meat was observed and agreed with some other published meta-analyses.10,70-74 However uncertainties for a clear positive dose-response association exist. One study found evidence for a non-linear shape association,11 confirmed by two other studies, but only for intakes higher than 90 or 140g/d, respectively.10,12 While for processed meat evidence is stronger, two recent comprehensive analyses by Alexander and colleagues put a doubt on a clear dose-response relationship between specifically red meat consumption and colorectal cancer risk,75,76 raising concerns on methodological differences between existing studies.
For lung, stomach, oesophagus and pancreas limited suggestive evidence was present (table I). For the association between red meat and lung cancer seven of nine case-control studies and a cohort study reported increased risk with increasing intakes, while a two-fold increased risk in highest vs lowest analyses based on three cohorts was observed (RR=2.10; 95%CI 1.00-4.42).2 A more recent meta-analysis (33 studies) showed a positive effect of both red and processed meat on lung cancer development.7 Stomach cancer was also linked to processed but not red meat intake (table I). A recent meta-analysis including eighteen studies indicated that each 100 g/day increment in red meat intake was associated with 17% increased risk of gastric cancer.15 For oesophageal cancer two cohort studies suggested a non-significant positive association with processed meat in extreme categories analyses, while for red meat the conclusions came from case-control studies, for which eight of ten found a positive association (table I). Another four meta-analyses confirmed an increased risk of both squamous and adenocarcinoma with higher red and/or processed meat intake,19,20,23,25 however, there is evidence for heterogeneity by the cancer subtype.21 For pancreatic cancer, red meat intake indicated a non-significantly increased risk, while for processed meat the association was significant (table I). This was confirmed in a meta-analysis based on eleven prospective studies.28
The definition of processed meat refers to meats preserved by smoking, curing, or salting, or addition of chemical preservatives. Therefore possible underlying mechanisms for cancer development may refer to the effect of carcinogenic compounds that are produced at high temperature (heterocyclic amines and polycyclic aromatic hydrocarbons).77 Meats are often preserved with salt. Salt was positively associated with the risk of stomach cancer (table I). Seven prospective studies investigated salt intake from processed meat and accordingly found a statistically positive association (RR=1.24; 95%CI 1.06-1.46; for high consumers).78 Haem present in red meat may undergo conversion to other compounds with cancer-promotive effects, N-nitroso compounds and cytotoxic alkenals.77,79 Their precursor, nitrate, added as preservative may enhance this effect.80 Evidence from animal and in vitro studies suggests the involvement of iron in gastrointestinal tract cancer development, caused by oxidative damage to colonic cells and promotion of cell growth.81,82
Some animal products (meat, cheese) high in fats were shown to increase risk of pancreatic cancer.83 It is plausible that for pancreatic cancer dietary saturated fatty acids may be an important factor for cancer development.84 Total fat was also considered as a limited-suggestive risk factor for postmenopausal breast cancer (RR=1.06; 95%CI 0.99-1.14; per 20g/day; based on five cohort studies). For both cheese and animal fat positive association (suggestive) was found for colorectal cancer (figure 1).
Trans fatty acids
Some recent epidemiological studies indicate that a high intake of Industrial trans fatty acids (ITFA) from industrially-produced hydrogenated vegetable oils may increase risks of different types of cancer, but studies are still scarce. These studies reported evidence of increased risks of postmenopausal breast,85,85) prostate,87 ovarian,88 distal colorectal89 cancers and colorectal adenomas90 associated with increasing dietary intake or biomarkers of ITFA. Thus, prevention of cancer should consider suppressing ITFA in highly processed foods.
Glycaemic index/load or high in sugar foods
An important role in most cancers' development is attributed to body fatness.91 Diets high in some fats, but also sugars postprandially converted to fat may indirectly contribute to increased body adipose tissue content. Diet high in sugar may results in hyperinsulinemia and increased levels of circulating tumour promoter IGF-1.92 Sugar (including sucrose and fructose) and/or high glycaemic index (GI) or load (GL) that reflect postprandial increase in blood sugar levels, were positively associated with some IGF-1-related cancers.92 Notably, pancreatic cancer that is strongly related to body fatness showed some evidence for association with foods and beverages containing fructose; 22% statistically significant increased risk of pancreatic cancer per 25g/day of fructose was observed in a meta-analysis of six cohort studies (RR=1.22; 95%CI 1.08-1.37).2 Foods containing sugars were also listed as limited-suggestive risk factor for colorectal cancer (figure 1), however no significant associations were reported between GI or GL and cancers of digestive tract, including colorectal.29,93 In turn, based on summary report of four cohort studies, GL was positively associated with endometrial cancer risk (table I). Lower but statistically significant estimates were obtained in another study (RR=1.06; 95%CI 1.01-1.11; per 50 unit/day).94 High GL was related to postmenopausal breast cancer in the EPIC cohort study. A stronger effect was observed among ER- tumors (HR= 1.36; 95%CI 1.02-1.82; 5th vs 1st quintile).95 Nowadays popular sources of fructose include sugary drinks. A suggestive, modest positive association between sugary drinks and pancreatic cancer was observed in a pooled analysis of fourteen cohort studies (RR=1.19; 95%CI 0.98-1.46; comparing ≥250 to 0 g/d).96 Recent evidence also suggests a possible risk association between liver cancer (HCC) and both sugar and sugary drinks intake.97,98
Direct mechanisms for carcinogenesis related to high dietary sugar intake and high glycemic load may be related to insulin resistance,99 promotion of systemic inflammation and increased levels of pro-inflammatory factors,100 that may promote cell proliferation (e.g. tumor necrosis factor alpha, interleukin-6 and C-reactive protein).91
Indices of lifestyle, dietary patterns and cancer risk
Foods are consumed in combination and several factors may affect consumer's dietary choices. Important impact on the foods consumed may derive from geographical location, cultural dietary habits, socioeconomic status, religion and underlying diseases.101 WCRF/AICR report investigated the relationship of several dietary patterns and specific diets but no conclusion could be reached in relation to any of the cancers studied. However, more recent evidence form cohort studies suggested protective effect of: a) healthy lifestyle index score, combining healthy diet, physical activity, low alcohol intake, no smoking and healthy BMI, on gastric adenocarcinoma, breast (in postmenopausal women) and colorectal cancers;102,104 b) adherence to the Mediterranean dietary pattern on overall cancer risk;105 c) healthy dietary pattern derived from factor analysis on breast106 and colorectal cancer risk,107 and d) concordance with a score based on WCRF/AICR recommendations on total, colorectal, stomach, breast, endometrium, lung, kidney, upper aerodigestive tract, liver, and esophagus cancers.108
Limitations and conclusion
In this report we updated the epidemiological evidence on the association of dietary factors and cancer. However, as new studies are conducted some of the results may be modified in particular with regards to factors with probable or limited evidence. In addition, it is important to remember that the estimation of the true intake of some nutrients is limited by imprecise dietary measurements, Other factors including potential recall bias in case-control studies or in more health conscious populations, unmeasured or residual confounding factors (such as food processing or preparation) may also alter the results. Dietary pattern analyses may partly deal with interaction between dietary nutrients but are difficult to interpret in etiological studies.
Taken together, favourable diet in relation to cancer prevention should be based on plant foods (fruits and vegetables) and be rich in dietary fibre and naturally occurring antioxidants. More evidence is needed on the protective effects of cereal products, legumes and coffee. Consumption of red and processed meat, energy-dense and high in sugars foods, as well as alcoholic and possibly sugary drinks should be limited. Adherence to dietary recommendations should be complemented by lifestyle modification, such as maintaining healthy body weight and being physically active. It is also important that the recommendations be applicable considering geographical region, economic development, and dietary cultural habits. Lastly, diet-gene interactions should be taken into account and personalised nutrition considered, especially in high risk individuals.109
In 2014, the 4th edition of European Code Against Cancer was launched by IARC indicating twelve lifestyle behaviours to lower the risk of developing cancer.110 One of the recommendations refers to maintaining a healthy diet: eating of plenty of whole grains, pulses, vegetable and fruits, limiting intake of red meat, high-calorie and high in salt foods, and avoiding sugary drinks and processed meat. Additional evidence, particularly based on existing cohorts, is continuously accumulating and changing, especially concerning the strength of observed associations. New WCRF/AICR global evaluation is ongoing and expected to be released in 2017.











 nova página do texto(beta)
nova página do texto(beta)


