Servicios Personalizados
Revista
Articulo
Indicadores
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Salud Pública de México
versión impresa ISSN 0036-3634
Salud pública Méx vol.50 supl.4 Cuernavaca ene. 2008
ARTÍCULO DE REVISIÓN
Medicines in Mexico, 1990-2004: systematic review of research on access and use
Medicamentos en México, 1990-2004: revisión de investigación sobre acceso y uso
Veronika J Wirtz, PhDI; Michael R Reich, PhDII; René Leyva Flores, PhDI; Anahí Dreser, MSc.I, III
ICentro de Investigación en Sistemas de Salud, Instituto Nacional de Salud Pública. México
IIHarvard School of Public Health, Harvard University. Boston, USA
IIIHealth Services Research Unit, London School of Hygiene and Tropical Medicine. United Kingdom
ABSTRACT
OBJECTIVE: To review original research studies published between 1990 and 2004 on the access and use of medicines in Mexico to assess the knowledge base for reforming Mexico's pharmaceutical policy.
MATERIAL AND METHODS: A literature review using electronic databases was conducted of original studies published in the last 15 years about access and use of medicines in Mexico. In addition, a manual search of six relevant journals was performed. Excluded were publications on herbal, complementary and alternative medicines.
RESULTS: Were identified 108 original articles as being relevant, out of 2289 titles reviewed, highlighting four policy-related problems: irrational prescribing, harmful self-medication, inequitable access, and frequent drug stock shortage in public health centers.
CONCLUSIONS: This review identified two priorities for Mexico's pharmaceutical policy and strategies: tackling the irrational use of medicines and the inadequate access of medicines. These are critical priorities for a new national pharmaceutical policy.
Key words: pharmaceutical policy; drug utilization; drug access; Mexico
RESUMEN
OBJETIVO: Revisar estudios de investigaciones originales publicados sobre el acceso y uso de los medicamentos en México de 1990 a 2004, con el fin de evaluar el conocimiento que existe para reformar la política farmacéutica nacional.
MATERIAL Y MÉTODOS: Se condujo una revisión de la literatura sobre estudios originales publicados entre 1990 y 2004 sobre el acceso y uso de medicamentos en México. Además, se revisaron manualmente seis revistas relevantes. Se excluyeron publicaciones sobre herbolaria, medicamentos tradicionales y alternativos.
RESULTADOS: Se revisaron 2 289 artículos e identificaron 108 como relevantes que destacan cuatro problemas importantes relacionados con las políticas farmacéuticas: prescripción inadecuada, automedicación dañina, acceso inequitativo y desabasto de medicamentos en servicios públicos de salud.
CONCLUSIONES: Esta revisión identificó dos prioridades críticas para el desarrollo de una nueva política farmacéutica en México: actuar sobre el uso irracional de medicamentos y sobre el acceso inadecuado a medicamentos.
Palabras clave: política farmacéutica; utilización de medicamentos; acceso a medicamentos; México
This paper provides a systematic review of original research studies of the access to and use of medicines in Mexico published in the last fifteen years. This review is timely, since in October 2005 the Mexican government published a background document for considering a new national pharmaceutical policy.1 One objective of the government's document is to provide a framework for policy development by various stakeholders. Before designing a new policy, however, it is important to identify the major problems that need to be addressed, and outline possible approaches to tackle them. The purpose of this review is to assess the existing research evidence on two key topics -access and use of medicines in Mexico- and to assist in the design of a new national pharmaceutical policy in Mexico. These two areas were chosen according to the core objectives of a national pharmaceutical policy as defined by the World Health Organization (WHO): availability of medicines at affordable prices and rational use of medicines. 2 The study questions for this review are: Are medicines accessible in public and private institutions and are they affordable? What are the consumption and prescription patterns? If problems have been identified in the past, which corrective strategies have been shown to be effective in the Mexican context? We also discuss the implications of this assessment for the reform of Mexico's pharmaceutical policy.
Material and Methods
A literature review was conducted of original studies published in English or Spanish over 15 years (1990-2004) about aspects concerning the access to and use of medicines in Mexico, using the following databases: PubMed, Excerpta Medica (EMBASE), Ingenta, LILACS (Latin America and Caribbean Health Science), SciElo (Scientific Electronic Library Online), Indice Bibliografico Español en Ciencias de la Salud (IBECS) and INRUD (International Network of Rational Drug Use). Key search words included "Mexico" alone or in combination with the search terms: "drugs", "pharmaceutical preparation", "drug supply", "prescribing", "adherence" and "pharmacy". Additionally, a manual search of six journals of high relevance was performed (Gaceta Médica de México, Salud Pública de México, Archives of Medical Research, Salud Mental, Boletín Fármacos and Pharmaceutical Care España). For this review use of medicines includes the concepts of prescription, dispensing and consumption of medicines as well as adherence. With access we are referring to drug pricing, distribution and marketing. Excluded were publications on herbal, complementary and alternative medicines, clinical trials assessing the benefits and risks of medicines, and studies on susceptibility and resistance to antibiotics and on drug intoxication. Also excluded were letters to the editor and editorials.
In the review, titles were first checked, and if they were found to be relevant the abstract was retrieved. For all studies included in the review the complete text was obtained for analysis. References of the articles were also searched for titles of other relevant articles.
Our principal aim was to conduct a systematic review -a search and appraisal of the literature under pre-established and explicit criteria. For this we classified the studies according to the following characteristics: topic studied, study site (including the type and level of care), study design, drug group or health condition investigated, and patient group included. This information was used to determine the main strengths and weaknesses of each article and the research base related to access and use of medicines in Mexico. We did not carry out a meta-analysis of the articles identified; that is, we did not synthesize the study results through quantitative analysis. Throughout, we use the terms drugs, medicines and pharmaceutical products interchangeably.
Results
Using the selection methods described above, we found 108 original articles for analysis, out of 2289 titles reviewed from electronic databases. More were reviewed by hand search.
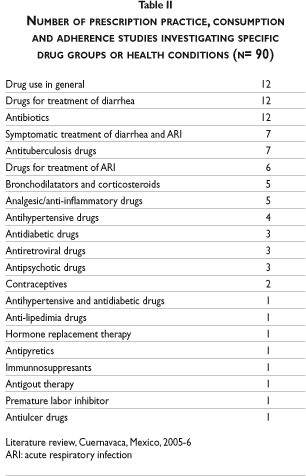
Tables I and II describe the characteristics of the 108 selected studies. In terms of research topics, 52 of the 108 studies investigated the prescribing patterns of medicines; of these, most were carried out in primary care settings (73.1%) and many focused on physicians working in public health care institutions (48.1%). Antibiotics and drugs for the symptomatic treatment of diarrhea were the most investigated drug groups (22%). Twenty-four of the 108 studies analyzed prescription or consumption in patients suffering from acute respiratory infection (ARI) as well as acute diarrhea. With regard to the study population, around one-fifth of the prescribing and consumption studies focused on children under five years old and only three studies examined people over 60 years old. Regarding the study site, more than one third of the studies (35.2%) were carried out in Mexico City. Regarding study methods, 42.6% used surveys as their main instrument of data collection.
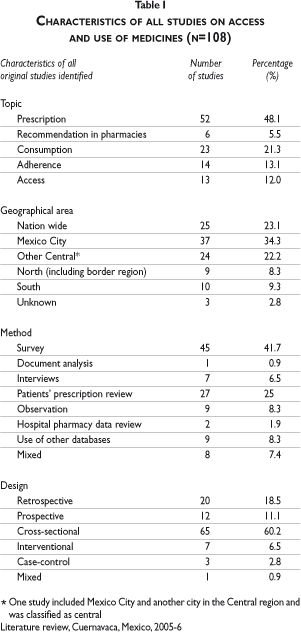
All articles selected were classified in two exclusive categories of use and access. Most research articles addressed use of medicines (95 out of 108 articles). Thirteen studies covered issues of access.
Use of medicines
We divided the studies related to use into four sub-topics: prescription practice (52 articles), advice by pharmacy personnel (6 articles), consumption of medicines (23 articles) and adherence (14 articles).
Prescription practice (n=52)
Investigating how physicians prescribe is one part of analyzing the use of medicines. The objective of pharmacotherapy is a rational utilization of medicines, which means the clinical needs of the patient, individual dose requirements and cost effectiveness are the main criteria for the use of medicines.
All except five studies investigated disease- or drug-specific prescribing practices, most commonly acute respiratory infection (ARI) and/or acute diarrhea (17 out of 52). 3-19 In all of these studies, inappropriate prescribing was identified, mainly due to the use of antibiotics, which is only recommended in a minority of cases of ARI and acute diarrhea. The use of antibiotics was discussed as a risk that could increase the development of bacterial resistance to antibiotics. One study evaluated the use of oral rehydration in the treatment of acute diarrhea.4 Importantly, half of these studies were developed before 1996.
Studies investigating the prescribing practices of public and private physicians for the treatment of conditions other than acute diarrhea and ARI included the following drug groups: antituberculosis drugs,20 antipsychotics and tranquillizers,21, 22 bronchodilatators and corticosteroids,23-26 antihypertensive drugs27-29 and/or antidiabetic drugs,30,31 lipid lowering drugs,32 antigout therapy,33 antiretroviral drugs,34,35 hormone replacement therapy,36 contraceptives,37 and antirheumatic drugs.38 Almost all of these studies concluded that treatment is sub-optimal and sometimes even harmful. Many of the authors attributed this problem to a lack of professional consensus or accepted treatment guidelines. Only one of the studies mentions pharmaceutical promotion as a factor influencing prescribing practice.28
The problem of inadequate treatment was also identified in two studies investigating general prescribing practice.39, 40 These studies detected prescriptions that included harmful medicines, for which the benefits of use were out-weighed by their side effects, and which had already been withdrawn from the pharmaceutical market in some developed countries. In addition to potential harm to individual patients, these medicines contributed to increasing costs to patients.
Three studies investigated prescription costs.41-43 Two studies used household data obtained by the National Health Survey,42,43 the other pharmacy customer data.41 Prescription costs were higher for uninsured individuals, and the use of non-essential drugs was found to impose an economic burden on consumers as it unnecessarily increased costs.
Eleven studies44—55 were carried out in secondary care. Seven studies evaluated antibiotic use, two analgesic use, one antiulcer medication use, and one pharmacotherapy of respiratory infection. The majority of studies concluded that prescribing patterns in secondary care need to be improved.
Overall, these 52 studies showed that prescribing practices for hospitalized and ambulatory patients are often inappropriate, including problems related to harmful prescription behavior, a lack of implementation and monitoring of evidence-based treatment guidelines, and unnecessary costs to patients.
Advice given by pharmacy personnel (n=6)
According to the six studies in this category, trained pharmacy personnel who are able to provide reliable and unbiased information to the consumer are scarce56-61 even though one study found about only 9% of pharmacy customers required advice from pharmacy personnel.56 Three of these studies used undercover researchers posing as customers to investigate the treatment recommendations given by pharmacy clerks mainly regarding treatment of tuberculosis, sexually transmitted infections, contraceptives, acute diarrhea or acute respiratory infections; in more than two-thirds of the cases the advice was either inappropriate or harmful. Three of the five studies did not differentiate between small private and chain pharmacies, and only one compared public and private pharmacies.
Consumption of medicines (n=23)
Consumption is the last stage in the medicines cycle. Twenty-three out of the 108 studies (21.5%) investigated consumption of medicines in the community. The majority of studies used surveys of consumers or patients as their data collection method for investigating the consumption of medicines.
Eleven consumption studies investigated the use of antibiotics62-66 or the use of medicines including antibiotics in the treatment of acute diarrhea in children under five years of age67-70 and ARI.71, 72 All of them reported irrational use of medicines. Four of the studies found that in less than 10% of acute diarrhea cases the use of antibiotics was justified (based on the detection of blood in the stool) and two-thirds of the antibiotics were used for less than five days, which increases the risk of bacterial resistance to antibiotics and unnecessary exposure of patients to side effects.62-65 Other studies found that between 35 and 65% of children below five years of age suffering from diarrhea received medicines, most commonly contra-indicated antibiotics and drugs against diarrhea.67-70
Surveys of either household members or pharmacy customers were used in five studies. These studies found that more than half of the respondents (51 to 61%) bought medicines without a physician's prescription73-75 and between 43 to 55% self-medicated with prescription-only medicines.76,77 Most frequently purchased drugs were antibiotics, analgesics, vitamins and cold and cough preparations. One study found that two-thirds of the antibiotics purchased were broad-spectrum, again indicating an increased risk of the development of bacterial resistance.74
One study on consumption of medicines in patients with fever found that although only 2% of patients were diagnosed with malaria, 37% took anti-malaria medication.78 In contrast to the large proportion of the studies focusing on children under 5, only two studies focused on adults over 60 years.79, 80 They found utilization patterns that are not based on evidence or internationally accepted standard treatment guidelines.66,67
Two studies found inadequate use of NSAID,81,82 one of them concluding that the increasing number of patients presenting with peptic ulcer was potentially related to their increased consumption of these medicines. Using household data from the National Health Survey 2000 it was reported that around half of individuals suffering from hypertension were using medication, but only 20% of them were controlled (<140/90mmHg). 83
Adherence to pharmacotherapy (n=12)
Twelve studies investigated adherence, with half of them on adherence to tuberculosis treatment.84-89 The remaining studies examined adherence to diabetes treatment,90,91 asthma treatment,92 contraception,93 antiretroviral therapy,94 antipsychotic medication,95 pharmacotherapy of infectious diseases and acute diarrhea96 and immunotherapy.97 The majority of these studies investigated factors influencing adherence by using either self-reporting surveys or focus groups. The perception of the disease, education, distance from health centers, living in rural areas, and social support were found to affect treatment adherence. Five studies evaluated the impact of an educational intervention (either patient or physician and patient) on adherence. One study investigated the effect of supervision on adherence. Education and degree of supervision were found to positively influence adherence. In addition to the 12 studies focusing on adherence, one other study examined aspects of adherence to antibiotics along with an analysis of prescribing patterns.19
Access to medicines (n=13)
The studies on access to medicines98-110 show that, first, drug prices in Mexico are higher than in many developed countries when adjusted for income.88-100 For example, with an average salary, an individual in the United States or France is able to buy more medicines than an individual with an average salary in Mexico. Second, the studies reported that access to medicines is hampered due to stock-outs of essential drugs in public health centers.101-106 The absence of medicines is the main reason for not returning to use public health care services.107 Third, there is inequity in access to medicines in Mexico: people from lower income groups spend proportionally more on medicines than individuals with higher income.43, 105 For example, it has been reported that the region with the highest poverty index received the least amount of drugs free of charge from the government.43 A recent study found that in the lowest income groups up to 60% of household health care expenditure is spent on medicines and that 66% of catastrophic health expenditure is due to the purchase of medicines.105
One study analyzed the legal regulations regarding opioid availability in five Latin American countries including Mexico.108 Mexico failed to meet the WHO criteria in adequately regulating access to these products. In addition, Mexico ranked as the Latin American country meeting the lowest number of international standards on opioid availability.
Discussion
Overall, the 108 studies in this review reported significant problems in access and use. The research studies on consumption and prescription patterns show that irrational use of drugs is a frequent problem in all therapeutic fields examined in Mexico, mostly documented in antibiotics and drugs for the symptomatic treatment of diarrhea. Studies on the accessibility and affordability of medicines reported frequent stock-outs of drugs in public health centers and inequitable access to medication (people in lower-income groups spend proportionally more on medicines than people with higher incomes). These findings show that Mexico shares many similarities with other low- and middle-income countries where problems of irrational use and inadequate access to medicines are common.111
The results also show that the research on medicines in Mexico published in peer-reviewed journals listed in the electronic databases and journals reviewed has significant limitations in terms of health problems and study topics, study methods and study sites (table III). Regarding health problems and study topic, the review identifies four areas where little has been published in the literature: 1) the three major causes of mortality in Mexico (cardiovascular diseases, diabetes, oncology); 2) the causes and consequences of irrational use of medicines (e.g., adverse drug events, their magnitude and the strategies to prevent them, and the beliefs, perceptions and attitudes of consumers related to consumption and adherence patterns); 3) the use of medicines in secondary care and rural areas; and 4) access to medicines.
Regarding study methods, longitudinal studies are scarce, which means that changes of prescribing practice or consumption patterns as well as access to medicines over time are not documented. Only five prescription studies were interventional, all developed during the 1990´s to evaluate the impact of educational interventions to improve prescribing practices for children presenting with acute diarrhea and/or ARI.11,15-18 An important theoretical limitation is that in these studies irrational prescribing has been addressed largely as an issue of lack of knowledge. The educational interventions carried out in the studies significantly improved prescribing quality. Questions remain about whether these results are transferable to other drug treatments and secondary care and whether they are sustainable in the long term.
Concerning study location, the majority of studies on prescribing practice were carried out in primary health care settings. Only eleven studies analyzing drug prescriptions were carried out in hospitals.44-55
What do these findings mean for the design and implementation of a new pharmaceutical policy in Mexico? The results indicate three priority areas for a new national pharmaceutical policy to address: a) strategies to combat irrational prescribing and consumption of medicines, b) strategies to improve access to medicines, and c) the promotion of sound nation-wide research on access and use of medicines, in order to inform the development of current and future policies.
Since the results show that irrational use of medicines is widespread among all actors involved in prescribing, dispensing and consumption, strategies to improve rational use need to include all of the actors, in particular medical doctors, pharmacy personnel and consumers. Educational interventions targeted at doctors in public health care institutions have achieved a positive effect on prescribing patterns in Mexico.11, 15-18 Additional studies are needed to explore how to combine educational interventions with other strategies, such as financial incentives for physicians, to influence prescribing practices.
The studies investigating advice received in pharmacies suggest that behavioral change of pharmacy personnel serving costumers can only be achieved through multiple strategies that discourage selling medicines without prescriptions. As Kroeger et al.56 pointed out, the financial profits from the sale of certain medicines are strong incentives for pharmacies and often of primary interest. Hence, strategies are required that include financial incentives for pharmacies to adhere to regulations and standards, for instance, requiring a medical prescription when dispensing prescription-only medicines. Studies are also needed that explore whether the presence of professional pharmacists would improve the quality of services without negatively affecting affordability of medicines due to higher prices, as some authors have suggested.112 At present, Mexican regulations do not require the presence of a professional pharmacist,113 although various groups have called for this policy.
Strategies to address the irrational use of medicines in Mexico also need to consider the role of consumers. This issue has particular relevance in a country such as Mexico where studies report that 51-61% of the population use self-medication as a response to healthcare needs,74-75 and up to 55% of consumers who self-medicate purchase prescription-only medicines without a physician's prescription.77 A public education campaign along with other strategies could help raise awareness among consumers of the potential harmful effects of medicines.
Strategies to improve access must first tackle the root causes of undersupply of medicines in public health care institutions so that individuals can obtain the medicines they require. Addressing the undersupply of drugs in public health care institutions requires a thorough analysis of the current system including financing for an adequate medicine supply. At the same time, health care institutions need to be made more accountable through good governance. Pressure from civil society would be a mechanism to create a more sustainable drug supply. Compared to countries such as Brazil, civil society has played a minor role in shaping health and pharmaceutical policies in Mexico.114
It is important to consider strategies that make medicines in the private market more affordable to people from low income groups. This could be achieved through price regulation in the private sector or by stimulating generic use and competition. The government's recent background document for a new national pharmaceutical policy highlights the importance of using generics to lower pharmaceutical expenditure; currently, however, there is no effective price regulation in Mexico.112 In contrast, in some European countries where the majority of the population have health insurance, price regulation is a central element of national pharmaceutical policy.115, 116
Expanding access will also depend on continued implementation of Mexico's new national health insurance program, the Popular Health Insurance Program [Seguro Popular], which seeks to provide health care and medicines for all Mexicans without health insurance (over half of the population in 2000).117 One study of this major policy reform91 shows that more patients affiliated with Seguro Popular received their prescribed medicines free of charge in comparison to the number of patients covered by other public health insurance schemes. Nonetheless, studies have yet to be published on prescribing patterns within Seguro Popular, consumption of medicines, and effects on health of patients. These are important areas for future policy research.
Conclusions
This first review of published studies on access to and use of medicines in Mexico identified important gaps in the evidence base in four important policy areas, including the use of and access to medicines for chronic diseases and the causes and consequences of irrational use. Research on medicines should be promoted in these areas to help guide the reform of Mexico's pharmaceutical policy. At the same time this review identified two priorities for Mexico's pharmaceutical policy and strategies. The analysis here suggests that tackling the irrational use of medicines and the inadequate access to medicines are critical priorities for a new national pharmaceutical policy. Regulatory changes that only include the government as actor are unlikely to achieve these priorities; instead, multiple strategies and involvement of multiple actors are necessary. Recently, the government launched three new programs, pharmacosurveillance, rational use of medicines, and clinical pharmacists in hospitals of the Ministry of Health; the latter two are specifically intended to improve the quality and cost-effectiveness of pharmacotherapy.118 It will be important to link these programs with other strategies to improve rational use of medicines, and evaluate their impacts over time.
The government's proposal to introduce a new comprehensive pharmaceutical policy comes at a time of major changes in Mexico's health insurance system, specifically the continuing implementation of Seguro Popular - aimed at providing universal coverage by the year 2010.117 These health system changes will have a major impact on access and use of medicines in Mexico. So far, there has been no rigorous analysis of what this means for the country's pharmaceutical policy. This review of published research provides important guidance about major gaps in the knowledge base in Mexico, areas where further research is required, and priority objectives for the design of a new national pharmaceutical policy.
References
1. Secretaría de Salud-COFEPRIS. Towards a comprehensive pharmaceutical policy. México, DF: SSA, 2005. [ Links ]
2. World Health Organization. How to design and implement a national health policy. Geneva: WHO, 2001. [ Links ]
3. Phillips M, Kumate-Rodríguez J, Mota-Hernández F. Costs of treating diarrea in a children´s hospital in Mexico City. Bol Of Sanit Panam 1990; 108: 27-37. [ Links ]
4. Muraira-Gutierrez A, Mendez-Jara A, Ruiz-Villapando G. An evaluation of the 4 years of the Oral Rehydration Service of the Hospital Infantil de Monterrey. Boletín Médico del Hospital Infantil de México 1992; 49: 358-364. (only in Appendix 2, not in text) [ Links ]
5.Gutiérrez G, Guiscafré H, Reyes H, Pérez R, Vega R, Tomé P. Reduction of mortality due to acute diarrheal diseases. Experiences from a research-action program. Salud Publica Mex 1994; 36: 168-179. [ Links ]
6. Torres J, Gonzalez-Arroyo S, Perez R, Muñoz O. Inappropriate treatment in children with bloody diarrhoea: Clinical and microbiological studies. Archive of Medical Research 1995; 26: 23-29. [ Links ]
7. Reyes H, Guiscafré H, Sarti E, Montoya Y, Tapia R, Gutiérrez G. Urban-rural variations in medical care of children with diarrhea in Mexico. Salud Publica Mex 1996; 38: 157-166. [ Links ]
8. Reyes H, Tomé P, Gutiérrez G, Rodríguez L, Orozco M, Guiscafré H. Mortality due to diarrea in Mexico: A problem of accessibility or quality of care? Salud Publica Mex 1998; 40: 316-323. [ Links ]
9. Corral-Terrazas M, Martínez H, Flores-Huerta S, Duque-L X, Turnbull B, Levario-Carrillo M. Beliefs and knowledge of a group of doctors about the nutritional management of the child with acute diarrhea. Salud Publica Mex 2002; 44: 303-314. [ Links ]
10. Cárdenas VM, Koopman JS, Garrido FJ, Bazúa LF, Ibarra JM, Stetler HC. Protective effect of antibtiotics on mortality risk from acute respiratory infections in Mexican Children. Bull Pan Am Health Organ 1992; 26: 108-120. [ Links ]
11. Pérez-Cuevas R, Guiscafré H, Muñoz O, Reyes H, Tomé P, Libreros V, et al. Improving physician prescribing patterns to treat rhinopharyngitis. Intervention strategies in two health systems of Mexico. Soc Sci Med 1996; 42: 1185-1194. [ Links ]
12. Reyes H, Perez-Cuevas R, Salieron J, Tome P, Guiscafre H, Gutierrez G. Infant mortality due to acute respiratory infections: the influence of primary care processes. Health Policy Plan 1997; 12: 214-223. [ Links ]
13. Peláez-Ballestas I, Hernández-Garduño A, Arredondo-García JL, Viramontes-Madrid JL, Aguilar-Chiu A. Use of antibiotics in upper respiratory infections on patients under 16 years old in private ambulatory medicine. Salud Publica Mex 2003; 45: 159-164. [ Links ]
14. Perez-Cuevas R, Muñoz O, Guiscafre H, Reyes H, Tome P, Gutierrez G. Patterns of therapeutic prescription in diarrhea and acute respiratory infections in 2 health care institutions: SS and IMSS. IV Characteristics of the medical prescriptions. Gac Med Mex 1992; 128: 505- 552. [ Links ]
15. Gutiérrez G, Guiscafré H, Bronfman M, Walsh J, Marínez H, Muñoz O. Changing physician prescribing patterns: evaluation of an education strategy for acute diarrhea in Mexico City. Med Care 1994; 32: 436-446. [ Links ]
16. Guiscafre H, Martinez H, Reyes H, Perez-Cuevas R, Castro R, Muñoz O, et al. From research to public health interventions. I. Impact of an educational strategy for physicians to improve treatment practices of common diseases. Arch Med Res 1995; 26 (Suppl): S31-S39. [ Links ]
17. Bojalil R, Guiscafré H, Espinosa P, Martínez H, Palafox M, Romero G, et al. The quality of private and public primary health care management of children with diarrhoea and acute respiratory infections in Tlaxcala, Mexico. Health Policy Plan 1998; 13: 323-331. [ Links ]
18. Bojalil R, Guiscafré H, Espinosa P, Viniegra L, Martínez H, Palafox M, et al. A clinical training unit for diarrhea and acute respiratory infections: an intervention for primary health care physicians in Mexico. Bull World Health Organ 1999; 77: 936-945. [ Links ]
19. Reyes H, Guiscarfe H, Muñoz O, Perez-Cuevas R, Martinez H, Gutierrez G. Antibtiotic noncompliance and waste in upper respiratory infections and acute diarrhea. J Clin Epidemiol 1997; 50: 1297-1304. [ Links ]
20. Alvarez-Gordillo G del C, Halperin-Frisch D, Blancarte-Melendres L, Vazquez-Castellanos JL. Risk factors for the resistance to anti-TB drug in Chiapas, Mexico. Salud Publica Mex 1995; 37: 408-416. [ Links ]
21. Apiquian R, Fresán A, de la Fuente-Sandoval C, Ulloa RE, Nicolini H. Survey on schizophrenia treatment in Mexico: perception and antipsychotic prescription patterns. BMC Psychiatry 2004; 4: 12. [ Links ]
22 Heinze G, Torres M,Cortes J. Attitudes of health professionals towards prescribing psychodrugs. Salud Mental 1999; 22: 1-7. [ Links ]
23. Baeza.Bacab MA, Ocampo-Espinosa CP, Abertos-Alpuche NE. Survey on the knowledge of acute asthma treatment among a group of pediatricians in the city of Merida, Yucatán. Bol Med Hosp Infant Mex 1997; 54: 311-316. [ Links ]
24. Pérez-Padilla JR, Rogelio J. Ketotifen (Zaditen and K-asthmal): Drug with sales out proportion to its demonstrated effectiveness. Gac Med Mex 1999; 135: 165-170. [ Links ]
25. Cavazos-Galván M, Contreras-Castillo J, Martínez-Llano E, Soni-Duque D. A economic study of asthma in Mexico. Rev Alerg Mex 2000; 47: 96-99. [ Links ]
26. Rico-Méndez FG, Hidalgo-Laffaurie G, Ochoa G, Massey LF, Gómez A, Múgica J de J. Treatment of chronic exacerbated bronchitis. Comparative analysis between governmental institutions and private physicians. Revista del Instituto Nacional de Enfermedades Respiratorias 1998; 11: 106-110. [ Links ]
27. García-Peña C, Thorogood M, Reyes S, Salmerón-Castro J, Curán C. The prevalence and treatment of hypertension in the elderly population of the Mexican Institute of Social Security. Salud PublicaMex 2001; 43: 415-420. [ Links ]
28. Goómez B JV, De Denis ME, Pereda-Reyna O. Antihypertensive medications used by specialist physicians and factors that influence the prescription. Revista Mexicana de Ciencias Farmaceuticas 2003; 34: 21-26. [ Links ]
29. Fonseca-Reyes S, Parra-Carrillo JZ, Anguiano AF. Antihypertensive prescription in a primary care center. Medicina Interna de Mexico 2001; 17: 213-217. [ Links ]
30. Robles-Silva L, Alcantara-Hernandez E, Mercado-Martinez FJ. Pattern of medical prescriptions for individuals with diabetes mellitus type II at the primary care level. Salud Publica Mex 1993; 35: 161-168. [ Links ]
31. Olaiz G, Rojas R, Barquera S, Shanah T, Aguilar C, Cravioto P, et al. Nacional Health Survey - Part II - Health of adults. Cuernavaca: Instituto Nacional de Salud Pública, 2003. [ Links ]
32 Meaney E, Vela A, Ramos A, Alemao E, Yin D. Cholesterol goal attainment with lipid lowering drugs. The COMETA Mexico Trial. Gac Med Mex 2004; 140: 493-501. [ Links ]
33 Vázquez-Mellado J, Espinoza J, Hernández-Garduño A, Lino L, Burgos-Vargas R. Diagnosis and treatment of gout in Mexico City. Results from a physician survey. Rev Invest Clin 2003; 55: 621-628. [ Links ]
34. Saavedra-López JA, Magis-Rodríguez C. Costs and expenses in medical care of AIDS in Mexico. México: Consejo Nacional de la Prevention y Control del SIDA, 1997. [ Links ]
35. Ponce de Leon-Rosales S, Rangel-Frausto MS, Váquez de-la Serna A, Huertas M, Martínez-Abaroa C. The quality of medical care in AIDS patients. Gac Med Mex 1998; 132 Suppl 1: 57-61. [ Links ]
36. Mueller KA, Zeron-Sanchez GJ, Sievert LL. Sources of information and HRT prescribing practices among gynaecologist in Puebla, Mexico. Maturitas 2003; 45: 137-144. [ Links ]
37. Rojas-Bernal T, Sojo-Aranda I, Cortéz-Gallegos V. Health conditions in populations using contraceptives. Ginecol Obstet Mex 1997; 65: 461-464. [ Links ]
38. Cardiel MH,Rojas-Serrano J. Community based study to estimate prevalence, burden of illness and help seeking behavior in rheumatic diseases in Mexico City. A COPCORD study. Clin Exp Rheumatol 2002; 20: 617-624. [ Links ]
39. Durán-González LL, Frenk-Mora J, Becerra-Aponte J.The quality of prescritive behavior in primary care. Salud Publica Mex1990; 32: 181-191. [ Links ]
40. Durán-González LL, Becerra-Aponte J, Franco R, Kravzov-Jinich J, Viso-Gurovich F, Frenk-Mora J. Medical main chart use made by the first level attention. Salud Publica Mex 1990; 32: 181-191. [ Links ]
41. Flores W, Ochoa H, Briggs J, Gracia R, Kroeger A. Economic costs associated with inadequate drug prescribing: an exploratory study in Chiapas, Mexico. Acta Tropica 2003; 88: 57-68. [ Links ]
42. Arredondo A, Nájera P, Leyva R. Ambulatory medical care in Mexico: the cost for users. Salud Publica Mex 1999; 41: 18-26. [ Links ]
43. Leyva-Flores R, Eriviti-Erice J, Kageyama-Escobar M L, Arredondo A. Medical prescription, drug access and drug expenditure among health service users in Mexico. Salud Publica Mex 1998; 40: 24-31. [ Links ]
44. Sánchez-Veláquez LD, Huerta-Torrijos J. Evaluation of the use of antibiotics in an intentive care unit. Medicina Crítica y Terapia Intensiva 1997; 11: 112-116 [ Links ]
45. Juárez-Olguín H, Florez-Pérez J, Lares-Asseff I, Montes-Ocampo I . Consumption of antibiotics in a pediatric hospital in Mexico City. Perinatol Reprod Hum 1998; 12: 157-162. [ Links ]
46. Navarrete-Navarro S, Ávila-Figueroa C, Medina-Cuevas F, Santos-Preciado JI. Surveillance and the costs related to the prescription of antimicrobials in a pediatric hospital. Gac Med Mex 1999; 135: 383-389. [ Links ]
47. Hernandez-Illescas JH, Campos-de la Valle G, Cano-Valle F, Olmedo-Canchola VH, Diaz-Muñoz AD, Ronzon B, et al. Management of antibiotics during the first semester of 1999 in the Hospital "Clínical Londres" in Mexico City. Medicina Interna de México 2000; 16: 182-197. [ Links ]
48. Zaidi M, Sifuentes-Osornio J, Rolón AL, Vázquez G, Rosado R, Sánchez M, et al. Inadequate therapy and antibiotic resistance. Risk factors for mortality in the intensive care unit. Arch Med Res 2002; 33: 290-294. [ Links ]
49. Garcia-Rubi E, Sierra-Madero JG, Ponce de Leon-Rosales S. Antibiotic use in outpatients. Rev Invest Clin 1991; 43: 113-118. [ Links ]
50. Velázquez-Armenta Y, Nava-Ocampo AA. Is pharmacy dispensing information useful to identify problems with analgesic prescribing in children? Paediatr Perinat Drug Ther 2003; 5: 135-138. [ Links ]
51. Gonzalez-Gomez LM, Garduno-Espinosa A, Vargas-Gomez M, De Lourdes Guerrero-Almeida M, Garduno-Espinosa J. Patterns of prescription in the pharmacological treatment of pain in hospitalized children. Bol Med Hosp Infant Mex 1995; 52: 98-104. [ Links ]
52. Perez R, Cuevas J, Bojalil R, Guiscafre H. Quality of health care in hospitalized child for acute respiratory infection. Bol Med Hosp Infant Mex 1995; 52: 342-349. [ Links ]
53. Carmona-Sanchez R, Suazo-Barahona J, Gonzalez A, Carmona-Sanchez L, Uscanga-Dominguez L. Use and abuse of histamine H2 receptor blockers in hospitalized patients. Rev Gastroenterol Mex 1997; 62: 84-88. [ Links ]
54. Hernandez-Hernandez DM, Vargas-Rivera MJE, Nava-Ocampo AA, Palma-Aguirre JA, Sumano-Lopez H. Drug therapy and adverse drug reactions to terbutaline in obstetric patients: A prospective cohort study in hospitalized women. BMC Pregnancy Childbirth 2002; 2: 3. [ Links ]
55. Viso-Gurovich F, Gomez-Oliva LM, Gonzalez- Velasco J.M. Assessment of the use of chloramphenicol at the Neonatal Unit of a Mexican Paediatric Hospital. Pharmaceutical Care Espana 2003; 5: 182-185. [ Links ]
56. Kroeger A, Oocha H, Arana B, Diaz A, Rizzo N, García R, et al. Inadequate drug advice in the pharmacies of Guatemala and Mexico: the scale of the problem and explanatory factors. Ann Trop Med Parasitol 2001; 95: 605-616. [ Links ]
57. Gellert G, Pyle N. Pharmacy practice and antibiotic-resistant tuberculosis along the US-Mexico border. JAMA 1994; 271: 1577-1578. [ Links ]
58. Leyva-FR, Bronfman PM, Erviti EJ. Simulated clients in drugstores: Prescriptive behaviour of drugstore attendants. J Soc Adm Pharm 2000; 17: 151-158. [ Links ]
59. Turner AN, Ellertson C, Thomas S, García S. Diagnosis and treatment of presumed STIs at Mexican pharmacies: survey results form a random sample of Mexico City pharmacy attendants. Sex Transm Infect 2003; 79: 224-228. [ Links ]
60. Becker D, Garcia SG, Ellertson C. Do Mexico City workers screen women for health risks when they sell oral contraceptive pills over-the-counter? Contraception 2004; 69: 295-299. [ Links ]
61. Martinez NOG, Piliado MD, Perez AT, Hernandez EZ,Moreno MLR. Evaluation of the patient's information about acquired antibiotics in drugstores. Revista Mexicana de Ciencias Farmaceuticas 1997; 28: 37-39. [ Links ]
62. Calva JJ, Cerón E, Bojalil R, Holbrook A. Antibiotic use in a community in Mexico City. II. A survey of purchases in drugstores. Bol Med Hosp Infant Mex 1993; 50: 145-149. [ Links ]
63. Bojalil R, Calva JJ, Ortega H. Use of antibiotics in a community of MexicoCity. I. Household survey. Bol Med Hosp Infant Mex 1993; 50: 79-87. [ Links ]
64. Bojalil R, Calva JJ. Antibiotic misuse in diarrhea. A household survey in a Mexican community. JClin Epidemiol 1994; 47: 147-156. [ Links ]
65. Calva JJ, Bojalil R. Antibiotic use in a periurban community in Mexico: a household and drugstore survey. Soc Sci Med 1996; 42: 1121-1128. [ Links ]
66. Macías AE, Herrera LE, Muñoz JM, Medina H. Antibiotic-resistant fecal Escherichia coli in healthy children. Induced by the use of antibiotics? Rev Invest Clin 2002; 54: 108-112. [ Links ]
67. Mota-Hernandez F, Tapia-Conyer R, Welti C, Franco A, Gómez-Ugalde J, Garrido MT. The management of diarrhoea at home in some regions of Mexico. Bol Med Hosp Infant Mex 1993; 50: 367-375. [ Links ]
68. Pérez-Cuevas R, Guiscafré H, Romero G, Rodríguez L, Gutiérrez G. Mothers' health-seeking behaviour in acute diarrhoea in Tlaxacala, Mexico. J Diarrhoeal Dis Res 1996; 14: 260-268. [ Links ]
69. Alvarez-Larrauri S. Maternal practice in case of infantile diarrhea and oral rehydration therapy. Salud Publica Mex 1998; 40: 256-264. [ Links ]
70. Martínez H, Ryan GW, Guiscafre H, Gutierrez G. An intercultural comparison of home case management of acute diarrhea in Mexico: implications for program planners. Arch Med Res 1998; 29: 351-360. [ Links ]
71. Castro V, Lopez-Cervantes M, Bronfman M, Tome P, Libreros V, Guiscafre H. Decisive factors of self-medication in two common infectious diseases. Gac Med Mex 1992; 128: 523-529. [ Links ]
72. Martínez H, Suriano K, Ryan G, Pelto GH. Ethnography of acute respiratory illnesses in a rural zone of the Mexican central highlands. Salud Publica Mex 1997; 39: 207-216. [ Links ]
73. Angeles-Chimal P, Medina-Flores ML, Molina-Rodriguez JF. Self-medication in the urban population of Cuernavaca, Morelos. Salud Publica Mex 1992; 34: 554-561. [ Links ]
74. Vicencio-Acevedo D, Alfaro-Valle A, Martínez-Toledo JL. Characteristics of drug acquisition in Morelia (Michoacán), Mexico. Bol Ofic Sanit Panam 1995; 119: 236-242. [ Links ]
75. Leyva-Flores R, Kageyama ML, Eriviti-Erice J. How people respond to illness in Mexico: self-care or medical care. Health Policy 2001; 57: 15-26. [ Links ]
76. Vazquez-Moreno E, Moreno-Santamaria R, Altagracia-Martínez M, Kravzov-Jinich J, Rios-Castaneda C. Self-medication in the Mexican State of Veracruz. Journal of Pharmaceutical Finance, Economics and Policy 2004; 13: 45-63. [ Links ]
77. Altagracia MM, Kravzov JJ, Moreno SR, Rios CC,Vazquez ME. Self-medication in rural and urban communities in the state of Guerrero, Mexico. Revista Mexicana de Ciencias Farmaceuticas 2003; 34: 27-35. [ Links ]
78. Leyva-F R, Eriviti-EJ, Ramsey JM, Gasman N. Medical drug utilization pattern for febrile patients in rural areas of Mexico. J Epidemiol1997; 50: 329-335. [ Links ]
79. Tapia-Conyer R, Cravioto P, Borges-Yáñez A, de la Rosa B. Consumption of prescription drugs in population aged 60 to 65 years in Mexico. Salud Publica Mex 1996; 38: 458-463. [ Links ]
80. Pérez-Guillé G, Camacho-Vieyra A, Toledo-López A, Guillé-Pérez A, Flores-Pérez J, Rodríguez-Pérez R. Patterns of drug consumption in relation with the pathologies of elderly Mexican subjects resident in nursing homes. Journal of Pharmacy and Pharmaceutical Science 2001; 4: 159-166. [ Links ]
81. Bobadilla J, Vargas-Vorácková F, Gómez A, Villaobos J de J. Frequency of peptic ulcer in the Nacional Institute of Nutrition "Salvador Zubirán"; Study of location, associated factors and time trends. Rev Gastroenterol Me 1996; 61: 31-35. [ Links ]
82. Kravzov JJ, Altagracia MM. Situation of NSAIDs in Mexico. Revista Mexicana de Ciencias Farmaceuticas 1997; 28: 14-20. [ Links ]
83. Velazquez-Monroy O, Rosas-Peralta M, Lara-Esqueda A, Pastelin-Hernandez G, Attie F,Tapia-Conyer R. Arterial hypertension in Mexico: results of the National Health Survey 2000. Arch Cardiol Mex 2002; 72: 71-84. [ Links ]
84. Alvarez-Gordillo GC, Dorantes-Jiménez JE. Shortened directly observed treatment of pulmonary tuberculosis. Salud Publica Mex 1998; 40:272-275. [ Links ]
85. Alvarez-Gordillo GC, Alvarez-Gordillo JF, Dorantes-Jimenez JE, Halperin-Frisch D. Perception and practices tuberculosis patients and non-adherence to therapy in Chiapas, Mexico. Salud Publica Mex 2000; 42: 520-528. [ Links ]
86. Alvarez-Gordillo GC, Dorantes-Jiménez JE, Molina-Rosales D. Seeking tuberculosis care in Chiapas, Mexico. Rev Panam Salud Publica 2001; 9: 285-293. [ Links ]
87. Alvarez-Gordillo GC, Alvarez-Gordillo JF, Dorantes-Jiménez JE. Educational strategy for improving patient compliance with the tuberculosis treatment regimen in Chiapas, Mexico. Rev Panam Salud Publica 2003; 14: 402-408. [ Links ]
88. Garcia-Garcia ML, Mayar-Maya ME, Ferreira-Reyes L, Palacios-Martinez M, Alverez-Garcia C, Valdespino-Gomez JL. Eficacia y eficiencia del tratamiento antituberculoso en jurisdicciones sanitarias de Morelos. Salud Publica Mex 1998; 40: 421-429. [ Links ]
89. Manjares-Morales EM, Serrano-Montes V, Cano-Perez G, Verduzco-Guerrero E, Escandon-Romero C, Escobedo-de la Pena J. Main causes of non-compliance with pulmonary tuberculosis treatment. Gac Med Mex 1993; 129: 57-62. [ Links ]
90. Garay-Sevilla ME, Nava LE, Malacara JM, Huerta R, Diaz-de Leon J, Mena A, et al. Adherente to treatment and social support in patients with non-insulin dependent diabetes mellitus. J Diabetes Complications 1995; 9: 81-86. [ Links ]
91. Hernandez-Ronquillo L, Tellez-Zenteno JF, Garduno-Espinoza J, Gonzalez-Acevez E. Factors associated with therapy noncompliance in type-2 diabetes patients. Salud Publica Mex 2003; 45: 191-197. [ Links ]
92 Segura-Mendez N, Espinola-Reyna G, Juarez-Morales D, Vazquez-Estupiñán F, Salas-Ramirez M, Ortiz-Vazquez JU, et al. Results of an educational program for adults with asthma. Rev Alerg Mex 2001; 48: 152-155. [ Links ]
93. Canto-De Centina TE, Canto P, Ordoñez-Luna M. Effect of counseling to improve compliance in Mexican women receiving depot-medrocyprogesterone acetate. Contraception 2001; 63: 143-146. [ Links ]
94. Escobar IT, Arguelles GR. Intervention for improving compliance to treatment in HIV+ patients. Medicina Interna de Mexico 2003; 19: 280-285 [ Links ]
95. Valencia-Collazos M, Ortega-Soto HA, Rascon-Gasca ML, Gomez-Caudillo L. Evaluation of the combination of psychosocial and pharmacological treatment in schizophrenic patients. Actas Esp Psiquiatr 2002; 30: 358-369 [ Links ]
96. Bronfman M, Castro R, Castro V, Guiscafre H, Monez O, Gutierrez G. Medical prescription and treatment compliance in acute infectious diarrhea: indirect impact o fan educational intervention. Salud Publica Mex 1991; 33: 568-575. [ Links ]
97. Ruiz FJ, Jiménez A, Cocoletzi J, Duran E. Compliance with and abandonment of immunotherapy. Rev Alerg Mex 1997; 44: 42-44. [ Links ]
98. Molina-Salazar R, Rivas-Vilchis JF. Overpricing and affordability of drugs: the case of essential drugs in Mexico. Cad Saude Publica 1998; 14: 501-506. [ Links ]
99. Calvo-Vargas CG, Parra-Carrillo JZ, Grover-Páez F, Fonseca-Reyes S. Changes in the costs of antihypertensive medications in a developing country. A study in Mexico comparing 1990 and 1996. American Journal of Hypertension 1998; 11: 487-493. [ Links ]
100. Danzon PM, Furukawa MF. Prices and availability of pharmaceuticals: Evidence from nine countries. Health Affairs 2003; October Suppl Web Exclusives:W3-521-36. [ Links ]
101. Ruiz-Gonzalez C, Vargas-Vorackova F, Castillo-Renteria C, Perez-Pimentel L, Martinez-Mata RA. What patients think of the services of the Instituto Nacional de la Nutricion "Salvador Zubiran". Rev Invest Clin 1990; 42: 298-311. [ Links ]
102. Bronfman M, Castro R, Zúñica E, Miranda C. "We do what we can": health service providers facing the utilization problem. Salud Publica Mex 1997; 39: 546-553. [ Links ]
103. Reséndez C, Garrido F, Gómez-Dantés O. Availability of essential drugs in Ministry of Health first level health care units in Tamaulipas, Mexico. Salud Publica Mex 2000; 42: 298-308. [ Links ]
104. Gómez-Dantés O, Garrido-Latorre F, Tirado-Gómez LL, Ramírez D, Macías C. Drug supplies in primary care units at the Ministry of Health in México. Salud Publica Mex 2001; 43: 224-232. [ Links ]
105. Nigenda G, Orozco E, Olaiz G. The importance of medicines in the Popular Health Insurance Program. In: Caleidoscopio de la Salud. Nigenda G, Knaul FM (ed.). Mexico: Fundacion Salud, 2003: 263-273. [ Links ]
106. Kravzov J,Altagracia M. Changes in the Mexican Essential Drug List. Journal of Social and Administrative Pharmacy 1994; 11: 153-158. [ Links ]
107. Ramírez-Sánchez TJ, Nájera-Aguilar P, Nigenda-López G. Quality of health services in Mexico as perceived by users. Salud Publica Mex 1998; 40: 3-10. [ Links ]
108. De Lima MHA, Sakowski JA, Hill CS, Bruera E. Legislation analysis according to WHO and INCB criteria on opioid availability: a comparative study of 5 countries and the state of Texas. Health Policy 2001; 56: 99-110. [ Links ]
109. Comité Terapéutico y Farmacológico de México. Open formulary proposal for a manager care program in Mexico. Salud Publica Mex 1999; 41: 27-41. [ Links ]
110. Altagracia M, Laska L, Jinich JK, Castilo R, Espejo O, Wertheimer AI, et al. An examination of market share trends within the ACE Inhibitor therapeutic category: Implications for cost containment efforts in Mexico. Journal of Social and Administrative Pharmacy 2000; 17: 34-44. [ Links ]
111. WHO. Medicines strategy: 2004-2007. Countries at the core. Geneva: World Health Organization, 2004. [ Links ]
112. OECD. Health working papers. Pharmaceutical pricing and reimbursement policies in Mexico. OECD: Paris, 2007. [ Links ]
113 SSA-COFEPRIS. Pharmacopeia of the United States of Mexico. Supplement for the establishments dedicating to the selling and dispensing of medicines and other health inputs. Regulation of the inputs for health. Article 127. Third edition. Mexico, DF: SSA-COFEPRIS, 2005. [ Links ]
114. Trostle J, Bronfman M, Langer A. How do researchers influence decision-makers? Case studies of Mexican policies. Health Policy and Planning 1999;14(2):103-114. [ Links ]
115. Ghislandi S, Krulichova I, Garattini L. Pharmaceutical policy in Italy: towards a structural change? Health Policy 2005;72:53-63. [ Links ]
116 Mossialos E, Oliver A. An overview of pharmaceutical policy in four countries: France, Germany, the Netherlands and the United Kingdom. International Journal of Health Planning and Management 2005;20:291-306. [ Links ]
117. Knaul FM, Frenk J. Health Insurance in Mexico: Achieving universal coverage through structural reform. Health Affairs 2005; 24, 1467-1476. [ Links ]
118. SSA. Rational use of medicines. 5th National Forum on Healthcare Quality, September 2007. Availabre form: http://www.salud.gob.mx/dirgrss/foro2007/page/mesas_tema/mesa5_p2.ppt [ Links ]
Received on: August 9, 2007
Accepted on: April 18, 2008
Address reprint requests to: Dr. Michael R. Reich. Harvard School of Public Health. 677 Huntington Avenue, Boston, MA 02115, USA.
E-mail: michael_reich@harvard.edu














