Servicios Personalizados
Revista
Articulo
Indicadores
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Salud Pública de México
versión impresa ISSN 0036-3634
Salud pública Méx vol.49 no.3 Cuernavaca may./jul. 2007
ARTÍCULO ORIGINAL
Prevalence of hepatitis C virus and HIV infection among injection drug users in two Mexican cities bordering the U.S
Prevalencia de los virus de la hepatitis C y de la inmunodeficiencia humana entre usuarios de drogas intravenosas, en dos ciudades mexicanas fronterizas con los Estados Unidos de America
Emily Faye White, BAI; Richard S. Garfein, PhD, MPHI; Kimberly C Brouwer, PhDI; Remedios Lozada, MD, MPHII; Rebeca Ramos, MPH, MAIII; Michelle Firestone-Cruz, MHSI; Saida G Pérez, BAI; Carlos Magis-Rodríguez, MD, PhDIV; Carlos J Conde-Glez, MSc, PhDV; Steffanie A Strathdee, PhD.I
IDivision of International Health and Cross-Cultural Medicine, Department of Family and Preventive Medicine, School of Medicine, University of California at San Diego. San Diego, CA, USA
IIPatronato COMUSIDA. Tijuana, Baja California, México
IIIUS México Border Health Association. El Paso, TX, USA
IVCentro Nacional para la Prevención y el Control del VIH/SIDA (CENSIDA). Mexico City, México
VInstituto Nacional de Salud Pública. Cuernavaca, Morelos, México
ABSTRACT
OBJECTIVE: To estimate the prevalence of the hepatitis C virus (HCV) and HIV infection and associated risk behaviors among injection drug users (IDUs) in two northern Mexican cities.
MATERIAL AND METHODS: Between February and April 2005, IDUs were recruited in Tijuana (N=222) and Ciudad Juarez (N=206) using respondent-driven sampling (RDS), a chain referral sampling approach. Interviewer-administered questionnaires assessed drug-using behaviors during the prior six months. Venous blood was collected for immunoassays to detect HIV and HCV antibodies. For HIV, Western blot or immunofluorescence assay was used for confirmatory testing. Final HCV antibody prevalence was estimated using RDS adjustments.
RESULTS: Overall, HCV and HIV prevalence was 96.0% and 2.8%, respectively, and was similar in both cities. Most IDUs (87.5%) reported passing on their used injection equipment to others, and 85.9% had received used equipment from others.
CONCLUSIONS: HIV prevalence was relatively high given the prevalence of HIV in the general population, and HCV prevalence was extremely high among IDUs in Tijuana and Ciudad Juarez. Frequent sharing practices indicate a high potential for continued transmission for both infections. HCV counseling and testing for IDUs in Mexico and interventions to reduce sharing of injection equipment are needed.
Key words: hepatitis C virus; human immunodeficiency virus; injection drug use; Mexico; needle sharing
RESUMEN
OBJETIVO: Estimar las prevalencias de los virus de hepatitis C (VHC) y de VIH y los comportamientos de riesgo asociados con ellos, entre usuarios de drogas inyectables (UDI) en dos ciudades del norte de México.
MATERIAL Y MÉTODOS: Entre febrero y abril de 2005, se reclutaron UDIs en Tijuana (N=222) y en Ciudad Juárez (N=206), mediante un método de muestreo llamado en inglés "respondent-driven sampling" (RDS), lo cual es un sistema basado en cadenas de referencia. Los participantes contestaron una encuesta aplicada por entrevista, la cual indagó acerca de los comportamientos en el uso de drogas durante los seis meses previos. Una muestra de sangre venosa fue colectada de cada individuo, para determinar la presencia de anticuerpos contra VIH y VHC mediante técnicas inmunoenzimáticas. En el caso del VIH la técnica de "Western blot" se aplicó con fines de confirmación. La prevalencia final de anticuerpos contra VHC se hizo mediante un cálculo ajustado, que empleó un estimador poblacional del RDS.
RESULTADOS: Las seroprevalencias globales de VHC y VIH, fueron 96% y 2.8%, respectivamente. Estas frecuencias fueron similares entre las muestras de ambas ciudades. La gran mayoría de los UDI (87.5%) manifestó haber transferido a otros sus equipos de inyección usados y a su vez 85.9% de los participantes declaró haber recibido equipos usados de otros.
CONCLUSIONES: La seroprevalencia encontrada de VIH fue relativamente alta dada la prevalencia de VIH en la población general y la de VHC fue extremadamente alta entre los UDI estudiados en Tijuana y en Ciudad Juárez. Las prácticas frecuentes de compartimiento de equipo señalan hacia un alto potencial que favorece la transmisión de ambas infecciones investigadas. Por tanto, son necesarias actividades de consejería y pruebas de laboratorio para VHC dirigidas a UDI en México y asimismo intervenciones para reducir el uso compartido de equipos de inyección.
Palabras clave: VHC; VIH; UDI; uso compartido de agujas; México; uso de drogas inyectables
It is estimated that 170 million people worldwide are infected with the hepatitis C virus (HCV).1 HCV is a single stranded RNA flavivirus, originally identified in 1989 as the major cause of non-A and non-B hepatitis.2 Although only a small proportion of acute HCV infections are symptomatic, HCV progresses to chronic infection in approximately 80% of cases and is an important cause of chronic liver disease worldwide.3,4 Approximately 15 to 20% of persons who acquire HCV infection progress to potentially serious cirrhosis and end-stage liver disease.5 Mexico has an HCV prevalence of approximately 0.7% to 1.6% in the general population.6-10 These estimates are somewhat lower than the 1.7% HCV prevalence for the Americas, and the global prevalence of 3%.1
HCV is transmitted most effectively through parenteral exposures to infected blood. Prior to the virus' discovery, transfusion of blood or blood products was a major mode of transmission. Since testing of blood supplies began, new cases of transfusion-transmitted hepatitis C has been virtually eliminated. Sexual transmission of HCV appears to be inefficient as most sexual risk behaviors have not been shown to be associated with HCV infection.4,11 Nosocomial transmission of HCV is possible if infection control measures are inadequate, such as the use of multidose vials, dialysis, and colonoscopy.12 HCV is readily transmitted through microtransfusions of infected blood through the shared use of syringes and other injection paraphernalia used to inject illicit drugs. Currently, the major mode of HCV transmission worldwide is injection drug use.11,12 In the United States, at least two-thirds of new HCV infections are associated with injection drug use.13
Injection risk behaviors such as the multi-person use (sharing) of injection equipment (i.e., needles/syringes, cookers, cotton, rinse-water) can transmit HCV, and potentially HIV and other blood-borne pathogens.4,14 HIV seroprevalence in Mexico is currently low, at 0.3% of the general population. However, seroprevalence among injection drug users (IDUs) in northwestern Mexico is significantly higher, and recent studies suggest that IDUs are increasingly practiced behaviors that could increase their risk for HIV.15,16 Since most HCV-infected persons are asymptomatic, serologic studies are needed to describe the epidemiology and develop interventions for HCV infection. Since HCV is about 10 times more infectious than HIV, and is usually the first infection to strike IDU populations,17 estimating the prevalence of HCV infection among IDUs in Mexico can serve as an early indicator of the potential spread of HIV.18
The purpose of this paper is to contribute to the epidemiological profile of HCV among IDUs in Mexico by estimating the HCV prevalence and describing risk behaviors among IDUs in Ciudad Juarez and Tijuana. To the knowledge of the authors of this study, this is the first community-based study of HCV seroprevalence among IDUs in these cities.
Material and Methods
Between February and April, 2005, IDUs were recruited in Tijuana and Ciudad Juarez for a cross-sectional study of behavioral and contextual factors associated with HIV and HCV infections. Eligibility criteria for the study included: having injected illicit drugs within the past month, confirmed by inspection of injection stigmata ('track marks'); aged 18 years or older; ability to speak Spanish; willingness and ability to provide informed consent; and not having had previously been interviewed for the study. Subjects gave their written informed consent to participate in the study. Study methods were approved by the Institutional Review Board of the University of California, San Diego and the Ethics Board of the Tijuana General Hospital. Programa Compañeros, which is a trusted and well-respected non-governmental organization (NGO) that has been providing services to and conducting studies of IDUs in Ciudad Juarez for decades, reviewed the protocol as it pertained to this city and approved it on ethical grounds.
Respondent-driven sampling (RDS), a chain referral sampling approach, was used to recruit participants.19,20 Briefly, a diverse group of "seeds" (heterogeneous in age, gender, drug of choice, and recruitment venue) were selected to initiate the process. The seeds were current IDUs who project outreach workers identified as having large social networks and were popular among their peers. Although individuals tend to recruit participants similar to themselves, studies of RDS have shown that balance is reached within approximately four to five waves of recruitment, ensuring that bias introduced from initial seed selection is eliminated.19 After providing informed consent, seeds were interviewed, briefly educated on how to refer other eligible IDUs, and then given three uniquely coded coupons for referring their peers. Waves of recruitment continued as subjects returning with coupons were given three coupons to recruit members from their own social network. The study name, locations where subjects could participate, and a brief explanation were printed on each coupon.
In Ciudad Juarez, interviews were conducted at a clinic run by Programa Compañeros. In Tijuana, CIRAD, an NGO started in 1991 to work with drug users, enrolled participants during weekly trips to three geographically diverse neighborhoods in the city: Zona Norte, Grupo Mexico, and Sepanal. Recruitment at these three sites was facilitated through the use of a modified recreational vehicle that operated as a mobile clinic (the "Prevemovihl").
Upon enrollment, trained staff administered quantitative surveys eliciting information on socio-demographic and behavioral characteristics. The questionnaire included: age at first injection; years of injecting; the number of times in the last six months that the interviewee let another person use their needle, cooker, cotton filter, or rinse water after they themselves had used it (classified as distributive sharing); the number of times in the last six months that the interviewee had used any of these items after they were used by another IDU (classified as receptive sharing); and the number of times in the last six months that the interviewee had divided drugs by filling one syringe with drug solution and then expelling a portion into the hub (frontloading) or barrel (backloading) of a second syringe. Participants were also asked about lifetime history of HCV and HIV testing ("Have you ever been tested for hepatitis C/HIV before today?"), hepatitis C knowledge ("Do you know what hepatitis C is?"), and HIV knowledge ("Do you know how HIV/AIDS is spread?"). All interviews and test results were given in Spanish, by staff members trained in obtaining informed consent and conducting interviews on HIV-related risk factors. Recruitment and data collection were conducted under the supervision of study investigators and senior field survey supervisors.
Blood samples were obtained by venipuncture by a certified nurse trained in collecting and handling biological specimens, and serum was stored at the municipal health clinic before being shipped frozen to either the New Mexico State Laboratory or the San Diego County Health and Human Services Laboratory where antibody testing was performed. HCV antibody testing was performed using an enzyme immunoassay (EIA) test (Ortho Diagnostic Systems EIA 3.0, Raritan, NJ, USA). Reactive specimens were retested in duplicate using EIA and determined to be positive if either or both of the repeat tests were reactive. This method has been used in prior studies and found to be reliable due to the high positive predictive value of this testing algorithm when used in high prevalence populations.21-23 Pre- and post-test counseling and referral to treatment, where indicated, was provided to all participants, consistent with guidelines published in the U.S. and applicable in Mexico.12,24 All participants testing anti-HCV positive were counseled about avoiding behaviors such as sharing injection equipment that could spread HCV, avoiding use of alcohol and drugs that could accelerate liver damage, availability of treatment for HCV infection, and recommendations to be evaluated by a physician if they had hepatitis symptoms.
All participants were screened on-site in Mexico for HIV with the Determine rapid test (Abbott Laboratories, Abbott Park, IL, USA). Samples that were positive or had uncertain results from the rapid test were sent to laboratories in the U.S. for confirmatory testing. Tijuana samples were tested by the San Diego County Health and Human Services Agency using HIV EIA followed by Western blot and HIV immunofluorescence assay on reactive samples. Ciudad Juárez samples were tested by the New Mexico State Department of Health using HIV EIA followed by Western blot on reactive samples. The determine rapid test has a very high sensitivity (100%) and specificity (99.4% - 100%).25-27 Furthermore, there was 100% concordance between rapid test and confirmatory test results in the current study, reassurance of the Determine assay's high positive predictive value in this population. All participants with confirmed HIV-positive results were referred to a doctor at the municipal health clinic in their city for clinical evaluation. Pre- and post-test counseling and referral to treatment, where indicated, was provided to all participants.
Adjusted HCV prevalence was calculated using the RDS population estimator which accounts and adjusts for three types of potential biases: i) what the sample composition would have been if all IDU seeds were recruited equally; ii) if the group of recruits referred by each wave of seeds had possessed equal homophily (i.e., the tendency of individuals to refer others similar to themselves); and iii) if all of these groups had equal network sizes.19,28
Results
Of the 428 IDUs who completed the survey and provided a blood sample, 222 (51.9%) were recruited in Tijuana and 206 (48.1%) were recruited in Ciudad Juarez. The majority of the study participants were men (91.8%), which was similar across cities. One quarter of the study participants drank alcohol at least once a week and 6.7% drank alcohol every day (table I). The median age of first injection was 19.5 years (interquartile range [IQR]: 16-25 years), and the median number of years since first injection was 12.5 years (IQR: 8-19 years).
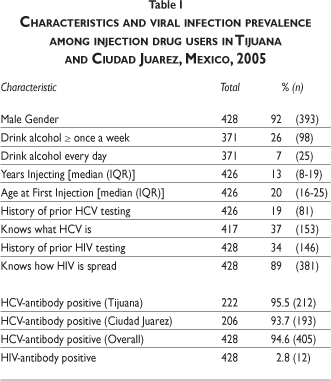
Overall, only 19.0% reported a prior history of HCV testing and 36.7% said that they knew what hepatitis C was. A higher percentage of IDUs (34%) reported having been previously tested for HIV, and 89% of participants said that they knew how HIV/AIDS was spread.
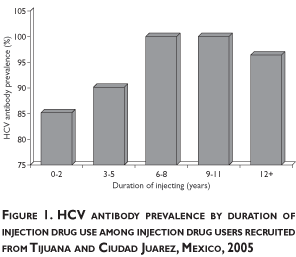
Overall HCV and HIV antibody prevalence was 94.6% and 2.8%, respectively. RDS adjusted HCV prevalence results (96.0%) were similar to crude results. HCV prevalence increased with greater duration of injection drug use, yet even among IDUs who had injected for less than three years, HCV prevalence was 84.6% (figure 1).
The prevalence of all HCV-associated risk behaviors was extremely high among the IDUs surveyed (table II). There was a high percentage of involvement in both distributive (87.3%) and receptive sharing (85.3%) of injection equipment in the last six months. The extremely high anti-HCV prevalence precluded extensive analysis of associations between risk behaviors and infection; however, the difference in the prevalence of receptive sharing of any injection equipment between anti-HCV-positive (85.9%) and anti-HCV-negative (70.6%) participants was marginally significant (p=0.08) even though the statistical power to detect a difference between these groups was only 16% (data not shown).

Discussion
Among IDUs in Tijuana and Ciudad Juarez –two Mexican cities that border the U.S.– a relatively high prevalence of HIV in comparison to the general population prevalence was observed, as well as an extremely high prevalence of HCV infection. Given the fact that nearly all IDUs in both cities were HCV-positive, it was not possible to identify statistically significant associations between HCV infection and injection behaviors known to be risk factors for blood-borne viral infection.23,29 Nonetheless, the observed prevalence of all of these behaviors was extremely high and greater among HCV-positive compared with HCV-negative participants. The potential for continued high prevalence of HCV and increased transmission of HIV and other blood-borne infections is high in these cities. Tijuana is reported to have the highest prevalence of drug use in Mexico and has a large number of "picaderos," or shooting galleries, which are noted for their high-risk syringe-sharing practices.15,30 In 2003, an estimated 6 000-10 000 IDUs in Tijuana injected in "picaderos."31,32 Ciudad Juarez is believed to have the second highest prevalence of drug use in Mexico, with an estimated 6 000 IDUs in the city.31 These findings suggest that HIV infection could spread rapidly among IDUs in Mexican cities bordering the U.S., as has been observed in other parts of the world,33,34 unless interventions to decrease risky injection behaviors are rapidly implemented.
HCV prevalence estimates among IDUs worldwide range from 55 to 95% and approaches 100% seropositivity in long-term users.18,35-37 While anti-HCV prevalence as high as 95% has been observed in U.S. urban IDU populations, lower prevalence has been observed in Latin American countries. A recent study of IDUs in Argentina found an anti-HCV prevalence of 55%, and studies from Brazil have reported anti-HCV prevalence estimates ranging from 53 to 75%.38-41 However, in a 1999 study of prison inmates in Ciudad Juarez, Mexico, anti-HCV prevalence was found to be 100%.42 The high HCV prevalence found in the inmates in the Ciudad Juarez study, along with the high prevalence found in our study, reflects how closely associated injection drug use and HCV seropositivity can be. In the past, blood transfusion was the major mode of transmission and most prevention efforts were aimed at blood banks. From more recent studies, including the one presented herein, it is clear that injection drug use is now the major mode of transmission, and therefore, prevention efforts need to be targeted appropriately.
Treatment for HCV is expensive, complicated, and not always effective. Currently, the best treatment available consists of pegylated interferon alpha plus ribavarin, which produces a sustained virologic response in 30 to 80% of treated patients.43 Response rates vary depending on the HCV genotype of the patient, with lower response rates of 30 to 40% seen in patients infected with genotype 1, which is the most common genotype that has been found in patients in Mexico.9,44,45 In Mexico, treatment with pegylated alpha 2a interferon costs $3 000 pesos per week ($270US) and needs to be taken weekly for six months to one year.46 Side effects of therapy can be severe, with patients experiencing flu-like symptoms, fatigue, and bone-marrow suppression. In addition, treatment may result in neuropsychiatric effects, such as apathy, irritability, and depression, which may be of special concern to IDUs, many of whom likely already suffer from such symptoms.47 Furthermore, the need for weekly interferon injections could be a psychological "trigger" for relapsing into injection drug use for patients who have stopped injecting. Therefore, drug abuse treatment should be a part of the treatment plan during and following treatment for HCV infection.
Due to the high expense and frequent complications of HCV treatment, measures aimed at preventing HCV transmission and reducing HCV disease progression should be expanded. However, only one-fifth of the IDUs in the study presented herein had ever been previously tested for HCV, and nearly two-thirds did not know what HCV was, illustrating the need to improve availability of HCV testing and education. Even in the absence of HCV treatment, screening for HCV is useful as a secondary prevention measure for educating infected persons about steps they can take to reduce their risk of developing end-stage liver disease and avoiding further transmission of the virus.
Secondary prevention measures for HCV-infected IDUs include vaccination against the hepatitis A and B virus, as co-infection has been shown to contribute to the severity of hepatocellular damage.48-50 In addition, patients chronically infected with HCV should be encouraged to avoid consuming alcohol, as even moderate intakes have been shown to enhance disease progression.51 In this study, over one-quarter of participants consumed alcohol at least once a week.
When accompanied by appropriate counseling, HCV antibody testing may also encourage behavioral risk reductions, thereby preventing transmission of HCV, HIV and other blood-borne pathogens. Primary prevention measures for anti-HCV-negative IDUs should include access to sterile injection equipment and education on proper injection practices. The use of any injection equipment that has been contaminated with HCV-infected blood, such as needles, syringes, cottons, and cookers, has been shown to be an independent risk factor for seroconversion.23,52 Not surprisingly, in this study the receptive sharing of injection equipment was associated with HCV infection and there was a high prevalence of sharing of all types of injection equipment. Despite the fact that a statistically significant difference in risk behaviors between the HCV antibody-negative and -positive groups could not be detected, due to the overall high prevalence of HCV infection in the population, the prevalence of each risk behavior was higher in the HCV positive group, indicating that there is much room for behavioral change. This is consistent with what has been found in studies in other parts of the world.23,29,52 Some studies have found the sharing of cookers that are used in the heating of drug preparations to be the single most important risk factor for seroconversion, after controlling for syringe sharing.23,53 Therefore, measures aimed at reducing HCV infection in IDUs who cannot or will not stop injecting drugs should not only focus on encouraging sterile syringe usage but also on the use of sterile cottons and cookers. Currently, Tijuana has one small needle exchange program and only two methadone clinics, both of which are privately operated. IDUs should be advised on where to obtain sterile equipment and should be shown how to properly sterilize equipment with bleach when new equipment is not available, as this may reduce the risk of HCV transmission.54 Since the study present here and others show that HCV infection occurs very soon after initiating injection drug use, a special effort needs to be made to reach out to new and short-term injection drug users.18,35
The high prevalence of anti-HCV found in this study shows the potential for other blood-borne infections to spread in this population. Of particular concern is HIV, which has been shown to spread rapidly amongst IDUs in other settings.33,34,55 Mexico is considered by UNAIDS to be a country of low HIV prevalence but high risk, ranking 23rd in the Americas in HIV prevalence but third in total number of cases.56 The prevalence of HIV in IDUs has thus far remained low for this risk group; however, current estimates in the study presented herein show it is over nine times more prevalent than in the general population, and the near saturation of HCV infection among IDUs in these cities may signal a looming HIV epidemic and a window of opportunity in which to prevent that from occurring. HCV infection has been found to be a significant independent risk factor for HIV seroconversion among IDUs.57 In many studies, over 90% of HIV-infected IDUs are coinfected with HCV.58,59 Furthermore, HIV infection causes HCV infection to progress more rapidly, and end stage liver disease is now the leading cause of death in HIV-HCV coinfected patients.58,60 HIV infection is also associated with increased sexual transmission of HCV, which could promote HCV spread to the non-IDU population.35,61
A few limitations of this study must be acknowledged. All risk behaviors were self-reported and possibly subject to bias from recall and socially desirable responding. IDUs may have tried to downplay the frequency of behaviors that they feel are socially undesirable, such as sharing injection equipment, on behavioral surveys. To minimize this possibility, indigenous outreach workers were used to conduct the interviews, and participants were assured that their responses would be kept confidential. Given the high proportion of participants who reported sharing injection equipment, it appears that socially desirable responding did not greatly influence this study's results, possibly because the IDUs in this study were not aware that sharing injection equipment is risky. Disease knowledge was also self-reported, although almost all of those claiming to know what HIV is correctly identified ways in which HIV is transmitted. Another limitation of this study is that, of those testing anti-HCV-positive, the proportion of actively infected individuals was not measured, as viral RNA was not determined by RT-PCR among seropositive subjects.
The high prevalence of anti-HCV found in this study illustrates the great potential for the parenteral spread of HIV and other blood-borne viruses among IDUs in Tijuana and Ciudad Juarez, Mexico. This study indicates that there is an urgent need to expand HCV counseling and testing for IDUs in Mexico, and to implement interventions that will decrease HCV- and HIV-associated injection risk behaviors in order to prevent a possible surge in the incidence of HIV infection in this population.
Acknowledgement
The authors would like to express their sincere thanks to the study participants for making this project feasible and successful. The authors also gratefully acknowledge support from the National Institute on Drug Abuse through grants DA09225-S11 and DA019829, donor support for the Harold Simon Chair in International Health and Cross-Cultural Medicine, the UCSD Center for AIDS Research (P30 AI36214-06), and training grants supporting Dr. Brouwer (NIDA grant K01DA020364 and an NIH Ruth L. Kirschstein NRSA (5 T32 AI07384) administered through the UCSD Center for AIDS Research). We are extremely thankful to the staff of Proyecto El Cuete, CIRAD, COMUSIDA and Programa Compañeros, as well as to Dr. Peter Hartsock.
References
1. Global surveillance and control of hepatitis C. Report of a WHO Consultation organized in collaboration with the Viral Hepatitis Prevention Board, Antwerp, Belgium. J Viral Hepat 1999;6:35-47. [ Links ]
2. Choo QL, Kuo G, Weiner AJ, Overby LR, Bradley DW, Houghton M. Isolation of a cDNA clone derived from a blood-borne non-A, non-B viral hepatitis genome. Science 1989;244:359-362. [ Links ]
3. Alter MJ. Hepatitis C virus infection in the United States. J Hepatol 1999;31 Suppl 1:88-91. [ Links ]
4. Hagan H, Des Jarlais DC. HIV and HCV infection among injecting drug users. Mt Sinai J Med 2000;67:423-428. [ Links ]
5. Liang TJ, Rehermann B, Seeff LB, Hoofnagle JH: Pathogenesis, natural history, treatment, and prevention of hepatitis C. Ann Intern Med 2000;132:296-305. [ Links ]
6. Vivas-Arceo C, Benavides SA, De Jesus Trujillo J, Panduro A, Rivas-Estilla AM. Hepatitis C virus: prevalence and routes of infection among blood donors of West Mexico. Hepatol Res 2003;25:115-123. [ Links ]
7. Carreto-Vélez MA, Carrada-Bravo T, Martínez-Magdaleno A. Seroprevalence of HBV, HCV, and HIV among blood donors in Irapuato, Mexico. Salud Publica Mex 2003;45 Supp 5:S690-S693. [ Links ]
8. Guerrero-Romero JF, Castañeda A, Rodríguez-Moran M. Prevalence of risk factors associated with hepatitis C in blood donors in the municipality of Durango, Mexico. Salud Publica Mex 1996;38:94-100. [ Links ]
9. Álvarez-Muñoz MT, Vences-Avilés MA, Damacio L, Vázquez-Rosales G, Torres J, González-Bravo F, Muñoz O. Hepatitis C virus RNA (HCV-RNA) in blood donors and family members seropositive for anti-HCV antibodies. Arch Med Res 2001;32:442-445. [ Links ]
10. Vera de León L, Juárez Navarro JA, Díaz Gómez M, Méndez Navarro J, Chirino Sprung RA, Dehesa Violante M, et al. Epidemiologic and situational panorama of hepatitis C in Mexico. Rev Gastroenterol Mex 2005;70:25-32. [ Links ]
11. Sulkowski MS, Thomas DL. Epidemiology and natural history of hepatitis C virus infection in injection drug users: implications for treatment. Clin Infect Dis 2005;40 Suppl 5:S263-S269. [ Links ]
12. Recommendations for prevention and control of hepatitis C virus (HCV) infection and HCV-related chronic disease. Centers for Disease Control and Prevention. MMWR Recomm Rep 1998;47:1-39. [ Links ]
13. Alter MJ. Prevention of spread of hepatitis C. Hepatology 2002;36:S93-S98. [ Links ]
14. Alter MJ, Moyer LA. The importance of preventing hepatitis C virus infection among injection drug users in the United States. J Acquir Immune Defic Syndr Hum Retrovirol 1998;18 Suppl 1:S6-S10. [ Links ]
15. Strathdee SA, Fraga WD, Case P, Firestone M, Brouwer KC, Perez SG, et al. Vivo para consumirla y la consumo para vivir [I live to inject and inject to live]: high-risk injection behaviors in Tijuana, Mexico. J Urban Health 2005;82:iv58-73. [ Links ]
16. UNAIDS. 2006 Report on the global AIDS epidemic. Available at: http://www.unaids.org/en/HIV_data/2006GlobalReport/default.asp. Accessed 2007. [ Links ]
17. Coppola RC, Manconi PE, Piro R, Di Martino ML, Masia G. HCV, HIV, HBV and HDV infections in intravenous drug addicts. Eur J Epidemiol 1994;10:279-283. [ Links ]
18. Garfein RS, Vlahov D, Galai N, Doherty MC, Nelson KE. Viral infections in short-term injection drug users: the prevalence of the hepatitis C, hepatitis B, human immunodeficiency, and human T-lymphotropic viruses. Am J Public Health 1996;86:655-661. [ Links ]
19. Heckathorn D. Respondent Driven Sampling: A New Approach to the Study of Hidden Populations. Social Problems 1997;44:174-199. [ Links ]
20. Frost SD, Brouwer KC, Firestone Cruz MA, Ramos R, Ramos ME, Lozada RM, et al. Respondent-Driven Sampling of Injection Drug Users in Two U.S.-Mexico Border Cities: Recruitment Dynamics and Impact on Estimates of HIV and Syphilis Prevalence. J Urban Health 2006;83 Suppl 7:83-97. [ Links ]
21. Alter MJ, Kuhnert WL, Finelli L. Guidelines for laboratory testing and result reporting of antibody to hepatitis C virus. Centers for Disease Control and Prevention. MMWR Recomm Rep 2003;52:1-13, 15. [ Links ]
22. Thomas DL, Shih JW, Alter HJ, Vlahov D, Cohn S, Hoover DR, et al. Effect of human immunodeficiency virus on hepatitis C virus infection among injecting drug users. J Infect Dis 1996;174:690-695. [ Links ]
23. Thorpe LE, Ouellet LJ, Hershow R, Bailey SL, Williams IT, Williamson J, et al. Risk of hepatitis C virus infection among young adult injection drug users who share injection equipment. Am J Epidemiol 2002;155:645-653. [ Links ]
24. NIH Consensus Statement on Management of Hepatitis C: 2002. NIH Consens State Sci Statements 2002;19:1-46. [ Links ]
25. Aidoo S, Ampofo WK, Brandful JA, Nuvor SV, Ansah JK, Nii-Trebi N, et al. Suitability of a rapid immunochromatographic test for detection of antibodies to human immunodeficiency virus in Ghana, West Africa. J Clin Microbiol 2001;39:2572-2575. [ Links ]
26. Arai H, Petchclai B, Khupulsup K, Kurimura T, Takeda K. Evaluation of a rapid immunochromatographic test for detection of antibodies to human immunodeficiency virus. J Clin Microbiol 1999;37:367-370. [ Links ]
27. Koblavi-Deme S, Maurice C, Yavo D, Sibailly TS, N'Guessan K, Kamelan-Tano Y, et al. Sensitivity and specificity of human immunodeficiency virus rapid serologic assays and testing algorithms in an antenatal clinic in Abidjan, Ivory Coast. J Clin Microbiol 2001;39:1808-1812. [ Links ]
28. Heckathorn DD. Respondent-Driven Sampling II: Deriving Valid Population Estimates from Chain-Referral Samples of Hidden Populations. Social Problems 2002;49:11-34. [ Links ]
29. Thorpe LE, Ouellet LJ, Levy JR, Williams IT, Monterroso ER. Hepatitis C virus infection: prevalence, risk factors, and prevention opportunities among young injection drug users in Chicago, 1997-1999. J Infect Dis 2000;182:1588-1594. [ Links ]
30. Needle RH, Coyle S, Cesari H, Trotter R, Clatts M, Koester S, et al. HIV risk behaviors associated with the injection process: multiperson use of drug injection equipment and paraphernalia in injection drug user networks. Subst Use Misuse 1998;33:2403-2423. [ Links ]
31. Bucardo J, Brouwer KC, Magis-Rodriguez C, Ramos R, Fraga M, Perez SG, et al. Historical trends in the production and consumption of illicit drugs in Mexico: implications for the prevention of blood borne infections. Drug Alcohol Depend 2005;79:281-293. [ Links ]
32. Brouwer KC, Case P, Ramos R, Magis-Rodriguez C, Bucardo J, Patterson TL, et al. Trends in production, trafficking, and consumption of methamphetamine and cocaine in Mexico. Subst Use Misuse 2006; 41:707-727. [ Links ]
33. Choopanya K, Des Jarlais DC, Vanichseni S, Mock PA, Kitayaporn D, Sangkhum U, et al. HIV risk reduction in a cohort of injecting drug users in Bangkok, Thailand. J Acquir Immune Defic Syndr 2003;33:88-95. [ Links ]
34. Des Jarlais DC, Friedman SR, Novick DM, Sotheran JL, Thomas P, Yancovitz SR, et al. HIV-1 infection among intravenous drug users in Manhattan, New York City, from 1977 through 1987. JAMA 1989;261:1008-1012. [ Links ]
35. Thomas DL, Vlahov D, Solomon L, Cohn S, Taylor E, Garfein R, et al. Correlates of hepatitis C virus infections among injection drug users. Medicine (Baltimore) 1995;74:212-220. [ Links ]
36. Bolumar F, Hernández-Aguado I, Ferrer L, Ruiz I, Avino MJ, Rebagliato M. Prevalence of antibiotics to hepatitis C in a population of intravenous drug users in Valencia, Spain, 1990-1992. Int J Epidemiol 1996;25:204-209. [ Links ]
37. Lorvick J, Kral AH, Seal K, Gee L, Edlin BR. Prevalence and duration of hepatitis C among injection drug users in San Francisco, Calif. Am J Public Health 2001;91:46-47. [ Links ]
38. Weissenbacher M, Rossi D, Radulich G, Sosa-Estani S, Vila M, Vivas E, et al. High seroprevalence of bloodborne viruses among street-recruited injection drug users from Buenos Aires, Argentina. Clin Infect Dis 2003;37 Suppl 5:S348-S352. [ Links ]
39. Caiaffa WT, Mingoti SA, Proietti FA, Carneiro-Proietti AB, Silva RC, Lopes AC, et al. Estimation of the number of injecting drug users attending an outreach syringe-exchange program and infection with human immunodeficiency virus (HIV) and hepatitis C virus: the AjUDE-Brasil project. J Urban Health 2003;80:106-114. [ Links ]
40. de Carvalho HB, Mesquita F, Massad E, Bueno RC, Lopes GT, Ruiz MA, et al. HIV and infections of similar transmission patterns in a drug injectors community of Santos, Brazil. J Acquir Immune Defic Syndr Hum Retrovirol 1996;12:84-92. [ Links ]
41. Oliveira ML, Bastos FI, Telles PR, Yoshida CF, Schatzmayr HG, Paetzold U, et al. Prevalence and risk factors for HBV, HCV and HDV infections among injecting drug users from Rio de Janeiro, Brazil. Braz J Med Biol Res 1999;32:1107-1114. [ Links ]
42. Rodríguez CM, Marques LF, Touze G. HIV and injection drug use in Latin America. Aids 2002;16 Suppl 3:S34-S41. [ Links ]
43. Fried MW, Shiffman ML, Reddy KR, Smith C, Marinos G, Goncales FL Jr, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med 2002;347:975-982. [ Links ]
44. Méndez-Sánchez N, Motola-Kuba D, Chávez-Tapia NC, Bahena J, Correa-Rotter R, Uribe M. Prevalence of hepatitis C virus infection among hemodialysis patients at a tertiary-care hospital in Mexico City, Mexico. J Clin Microbiol 2004;42:4321-4322. [ Links ]
45. Jarrett M, Cox P. Hepatitis C virus. Nurs Clin North Am 2004;39: 219-229. [ Links ]
46. Wong JB. Cost-effectiveness of treatments for chronic hepatitis C. Am J Med 1999;107:S74-S78. [ Links ]
47. Golub ET, Latka M, Hagan H, Havens JR, Hudson SM, Kapadia F, et al. Screening for depressive symptoms among HCV-infected injection drug users: examination of the utility of the CES-D and the Beck Depression Inventory. J Urban Health 2004;81:278-290. [ Links ]
48. Maayan S, Shufman EN, Engelhard D, Shouval D. Exposure to hepatitis B and C and to HTLV-1 and 2 among Israeli drug abusers in Jerusalem. Addiction 1994;89:869-874. [ Links ]
49. Koff RS. Risks associated with hepatitis A and hepatitis B in patients with hepatitis C. J Clin Gastroenterol 2001;33:20-26. [ Links ]
50. Prevention of hepatitis A through active or passive immunization: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 1999;48:1-37. [ Links ]
51. Szabo G, Aloman C, Polyak SJ, Weinman SA, Wands J, Zakhari S. Hepatitis C infection and alcohol use: A dangerous mix for the liver and antiviral immunity. Alcohol Clin Exp Res 2006;30:709-719. [ Links ]
52. Hagan H, Thiede H, Weiss NS, Hopkins SG, Duchin JS, Alexander ER. Sharing of drug preparation equipment as a risk factor for hepatitis C. Am J Public Health 2001;91:42-46. [ Links ]
53. Thorpe L, Ouellet L, Hershow R, Bailey S, Williams II, Monerosso E. The multiperson use of non-syringe injection equipment and risk of hepatitis C infection in a cohort of young adult injection drug users, chicago 1997-1999. Ann Epidemiol 2000;10:472-473. [ Links ]
54. Kapadia F, Vlahov D, Des Jarlais DC, Strathdee SA, Ouellet L, Kerndt P, et al. Does bleach disinfection of syringes protect against hepatitis C infection among young adult injection drug users? Epidemiology 2002;13:738-741. [ Links ]
55. Burns SM, Brettle RP, Gore SM, Peutherer JF, Robertson JR. The epidemiology of HIV infection in Edinburgh related to the injecting of drugs: an historical perspective and new insight regarding the past incidence of HIV infection derived from retrospective HIV antibody testing of stored samples of serum. J Infect 1996;32:53-62. [ Links ]
56. UNAIDS/WHO. México: Epidemiological Facts Sheets on HIV/AIDS and Sexually Transmitted Infections. Geneva, Switzerland: Joint United Nations Programme on HIV/AIDS; 2004 January 9. Accessed 2007. Available at: www.who.int/GlobalAtlas/predefinedReports/EFS2004/EFS_PDFs/EFS2004_MX.pdf. [ Links ]
57. Backmund M, Meyer K, Henkel C, Reimer J, Wachtler M, Schutz CG. Risk Factors and Predictors of Human Immunodeficiency Virus Infection among Injection Drug Users. Eur Addict Res 2005;11:138-144. [ Links ]
58. Maier I, Wu GY. Hepatitis C and HIV co-infection: a review. World J Gastroenterol 2002;8:577-579. [ Links ]
59. Miller CL, Wood E, Spittal PM, Li K, Frankish JC, Braitstein P, et al. The Future Face of Coinfection: Prevalence and Incidence of HIV and Hepatitis C Virus Coinfection Among Young Injection Drug Users. J Acquir Immune Defic Syndr 2004;36:743-749. [ Links ]
60. Salmon-Ceron D, Lewden C, Morlat P, Bevilacqua S, Jougla E, Bonnet F, et al. Liver disease as a major cause of death among HIV infected patients: role of hepatitis C and B viruses and alcohol. J Hepatol 2005;42:799-805. [ Links ]
61. Eyster ME, Alter HJ, Aledort LM, Quan S, Hatzakis A, Goedert JJ. Heterosexual co-transmission of hepatitis C virus (HCV) and human immunodeficiency virus (HIV). Ann Intern Med 1991;115:764-768. [ Links ]
Received on: August 25, 2006
Accepted on: February 14, 2007
Address reprint requests to: Dr. Richard S. Garfein. Division of International Health & Cross Cultural Medicine, Department of Family and Preventive Medicine, University of California, San Diego School of Medicine. 9500 Gilman Drive, mailstop 0622 San Diego, CA 92093 USA. E-mail: rgarfein@ucsd.edu














