INTRODUCTION
The estimated prevalence of Vitamin B12 (VB12) deficiency in the general population varies between 1.5% and 15%1,2. The most common cause of VB12 deficiency in children is inadequate dietary intake3. As VB12 cannot be produced by humans, it should be obtained from animal-based foods, such as meat, milk, eggs, and fish4,5. Therefore, vegetarianism and low socioeconomic conditions are important risk factors for VB12 deficiency6. Other causes are pernicious anemia, gastrectomy, bariatric surgery, gastritis, drugs (proton-pump inhibitors, H2 receptor blockers, antacids, etc.), and Imerslund-Gräsbeck syndrome7,8.
Intramuscular (IM) route is the primary choice of treatment for VB12 deficiency in most countries; however, it has some disadvantages, such as pain, high cost, and poor adherence to treatment9,10. In recent years, because of these disadvantages, new methods are being sought for treating VB12 deficiency.
Dietary VB12 gets absorbed through two different pathways11. Through the first pathway, VB12 binds intrinsic factor (IF), which is released by parietal cells in the stomach, following which the VB12-IF complex gets actively absorbed in the terminal ileum12. Approximately 60% of the absorption of dietary VB12 occurs through this biochemical mechanism12. Through the second pathway, dietary VB12 gets absorbed by simple diffusion (without binding to IF) along the entire intestine or it gets directly absorbed by sublingual (SL) capillaries13. Therefore, SL route for treating VB12 deficiency has several advantages, including potentially good adherence to treatment, safety, and low cost1. In addition, this route enables the treatment of VB12 deficiency in patients with swallowing disorders or malabsorption syndrome (inflammatory intestinal diseases, intestinal surgery, short bowel syndrome, etc.)14,15. In recent years, the importance of SL route has been recognized; however, few studies have evaluated this method, especially in children1,13,16.
In the present study, we aimed to compare SL and IM routes of VB12 administration in children with VB12 deficiency and to compare the efficacy of methylcobalamin and cyanocobalamin therapy in these children.
METHODS
Study population
This retrospective study comprised 129 patients with VB12 deficiency (126 inadequate dietary intake and 3 pernicious anemia) aged 5-18 years, and conducted between January 2017 and December 2019. We obtained the data from the electronic medical record system and patient files at Parkhayat Hospital. Based on the formulations of VB12, we divided the patients into three treatment groups as IM cyanocobalamin, SL cyanocobalamin, and SL methylcobalamin. The study was approved by Kütahya University of Health Sciences Ethics Committee and conducted in accordance with the ethical principles described by the Declaration of Helsinki (2020/05-12).
Inclusion criteria were as follows: (I) aged 5-18 years and (II) VB12 deficiency (serum VB12 level ≤ 200 pg/mL). Exclusion criteria were as follows: (I) patients with chronic diseases that may affect hematologic parameters (sideroblastic anemia, thalassemia, aplastic anemia, etc.), (II) folate deficiency, (III) iron deficiency, (IV) renal disease, (V) using drugs that may affect the absorption of VB12 (metformin, proton pump inhibitors, phenobarbital, etc.), and (VI) having missing data.
Laboratory studies
Complete blood count was measured with automatic blood count device LH 750 (Beckman Coulter, Inc., USA), and VB12 level was measured with automatic biochemistry device Advia Centaur® XPT (Siemens, Berlin, Germany). The lower level of hemoglobin (Hb), white blood cell (WBC), and platelet was determined as follows: < 11 g/dL, < 4000/mm3, and < 150.000/mm3, respectively. VB12 level ≤ 200 pg/mL was described as a deficiency. According to the protocol that is applied in our hospital, the treatment response of VB12 is reevaluated after 4 weeks in every patient.
Treatment protocols
Statistical analysis
The statistical analyses were performed using SPSS version 21 software. Continuous variables were calculated as mean ± standard deviation (SD). A one-way analysis of variance and Chi-square tests were used to assess comparisons between the treatment groups. Post hoc analysis was carried out by Tukey test. The independent t-test was used to compare pre-treatment and post-treatment values. p < 0.05 was considered as statistically significant.
RESULTS
A total number of 129 children with VB12 deficiency were included in this retrospective study (the treatment groups; IM cyanocobalamin group [n = 47], SL cyanocobalamin group [n = 43], and SL methylcobalamin group [n = 39]). Female/male ratio of IM cyanocobalamin, SL cyanocobalamin, and SL methylcobalamin groups was 21/26, 23/20, and 19/20, respectively (p > 0.05), and mean age of the groups was 12.7 ± 5.1, 12.3 ± 2.1, and 11.9 ± 5.6 years, respectively (p > 0.05). The demographic and laboratory characteristics of the study participants are summarized in Table 1.
Table 1 The demographic and laboratory characteristics of the study participants
| IM cyanocobalamin group (n = 47) | SL cyanocobalamin group (n = 43) | SL methylcobalamin group (n = 39) | p | |
|---|---|---|---|---|
| Mean ± SD | ||||
| Age, year | 12.7 ± 5.1 | 12.3 ± 2.1 | 11.9 ± 5.6 | 0.463 |
| Gender, n | 21/26 | 23/20 | 19/20 | 0.139 |
| Female | ||||
| Male | ||||
| Serum vitamin B12, pg/mL | ||||
| Pre-treatment | 147.5 ± 37.7 | 137.2 ± 36.5 | 146.7 ± 40.5 | 0.153 |
| Post-treatment | 602.0 ± 156.1 | 483.4 ± 144.8 | 565.5 ± 108.1 | |
| p | < 0.001 | < 0.001 | < 0.001 | |
| ΔsVB12 levels before and after treatment | 454.5 ± 118.4 | 346.2 ± 108.3 | 418.8 ± 67.6 | < 0.001 |
| Hb, gr/dl | ||||
| Pre-treatment | 10.1 ± 1.3 | 10.4 ± 1.5 | 10.3 ± 1.6 | 0.154 |
| Post-treatment | 11.8 ± 1.2 | 11.3 ± 1.3 | 11.6 ± 1.4 | 0.142 |
| p | < 0.001 | < 0.001 | < 0.001 | |
| MCV, fL | ||||
| Pre-treatment | 82.6 ± 10.8 | 81.1 ± 6.8 | 82.3 ± 7.1 | 0.332 |
| Post-treatment | 78.7 ± 10.2 | 80.1 ± 6.8 | 79.3 ± 15.9 | 0.591 |
| p | < 0.001 | 0.023 | < 0.001 | |
| Anemia, n | ||||
| Pre-treatment | 28 (59.5%) | 23 (53.4%) | 25 (64.1%) | 0.417 |
| Post-treatment | 15 (31.9%) | 16 (37.2%) | 16 (41%) | 0.351 |
| WBC count, /mm3 | ||||
| Pre-treatment | 7266.6 ± 1556.5 | 8612.1 ± 1012.7 | 7877.5 ± 1890.2 | 0.211 |
| Post-treatment | 8924.4 ± 1015.4 | 8904.1 ± 2534.1 | 7351.2 ± 1199.9 | 0.124 |
| p | 0.131 | 0.342 | 0.457 | |
| Platelet count, /mm3 | ||||
| Pre-treatment | 290.833 ± 57.314 | 296.920 ± 95.949 | 291.374 ± 116.925 | 0.927 |
| Post-treatment | 272.142 ± 57.616 | 304.187 ± 89.928 | 299.333 ± 66.525 | 0.573 |
| P | 0.531 | 0.742 | 0.757 | |
Hb: hemoglobin; IM: intramuscular; MCV: mean corpuscular volume; SL: sublingual; sVB12: serum Vitamin B12; WBC: white blood cell.
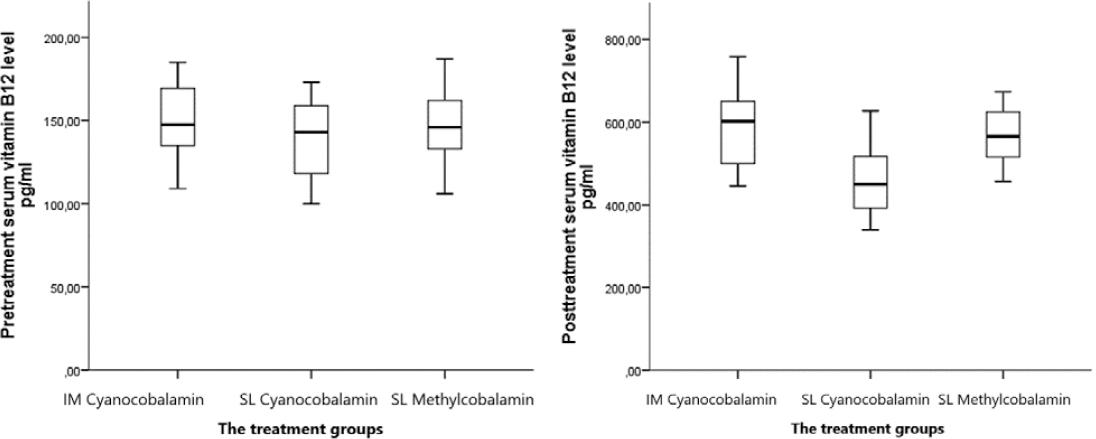
After VB12 therapy, serum VB12 levels increased significantly in all patients, and there was a statistically significant difference between the treatment groups (p < 0.05). Mean pre-treatment VB12 levels of IM cyanocobalamin, SL cyanocobalamin, and SL methylcobalamin groups were found as 147.5 ± 37.7, 137.2 ± 36.5, and 146.7 ± 40.5 pg/mL, respectively (p > 0.05), and after the therapy, the levels were detected as 602.0 ± 156.1, 483.4 ± 144.8, and 565.5 ± 108.1 pg/mL, respectively (p < 0.05) (Table 1 and Fig. 1). In addition, mean ± SD difference between serum VB12 levels before and after VB12 therapy of the treatment groups was found as 454.5 ± 118.4, 346.2 ± 108.3, and 418.8 ± 67.6, respectively (p < 0.05) (Table 1).
Mean ± SD difference between serum VB12 levels before and after VB12 therapy was found as in IM cyanocobalamin group versus SL cyanocobalamin group (454.5 ± 118.4 vs. 346.2 ± 108.3, p < 0.05); in IM cyanocobalamin group versus SL methylcobalamin group (454.5 ± 118.4 vs. 418.8 ± 67.6, p < 0.05); and in SL cyanocobalamin group versus SL methylcobalamin group (346.2 ± 108.3 vs. 418.8 ± 67.6, p < 0.05) (Table 2).
Table 2 Tukeys post hoc analysis of the groups
| IM cyanocobalamin group (n = 47) | SL cyanocobalamin group (n = 43) | p | IM cyanocobalamin group (n = 47) | SL methylcobalamin group (n = 39) | p | SL cyanocobalamin group (n = 43) | SL methylcobalamin group (n = 39) | p | |
|---|---|---|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | |||||||
| ΔsVB12 levels before and after treatment | 454.5 ± 118.4 | 346.2 ± 108.3 | < 0.001 | 454.5 ± 118.4 | 418.8 ± 67.6 | 0.032 | 346.2 ± 108.3 | 418.8 ± 67.6 | <0.001 |
sVB12: serum Vitamin B12; IM: intramuscular; SL: sublingual.
Out of 129 patients, 76 (58.9%) were found to have anemia (Hb < 11 g/dL) before VB12 treatment. After the 1st month of the treatment, the improvement ratios in anemia of the treatment groups (IM cyanocobalamin group, SL cyanocobalamin group, and SL methylcobalamin group) were detected as 13 (46.4%)/28, 7 (30.4%)/23, and 9 (36%)/25, res- pectively (p > 0.05). In addition, WBC and platelet counts did not change significantly either after VB12 therapy or between the treatment groups.
DISCUSSION
IM injection is a traditional choice of treatment for VB12 deficiency due to inadequate dietary intake, pernicious anemia, gastrectomy, ileal resection, or malabsorption syndrome17,18. However, IM method is associated with some disadvantages, such as pain, high cost, poor adherence to treatment, and bleeding in patients with coagulation disorders19. Considering these issues, SL route is now being considered for VB12 administration. SL method enables direct absorption of VB12 under the tongue, bypassing intestinal absorption14. Moreover, this method has several advantages, for example, it is less costly, results in high patient satisfaction, does not require a hospital visit, is not painful, and does not result in injection-related injury16. However, few studies have evaluated the efficacy of SL administration of VB12 and compared it with that of IM administration.
Bensky et al. compared the efficacy of SL and IM administration of VB12 in terms of normalizing serum cyanocobalamin levels. In their study, 3451 patients received SL VB12 (1000 μg/day for 6 months) and 830 patients received 1000 μg/dose IM VB12 (every other day for 2 weeks, followed by once a week for 4 weeks). Pre-treatment mean values of serum cyanocobalamin level were increased from 298 ng/L to 551 ng/L in the SL group, whereas in the IM group, mean value increased from 234 pg/mL to 452 ng/L. The post-treatment values significantly differed between the two groups (p < 0.001). The authors concluded that SL route should be the first-line choice of treatment in patients with VB12 deficiency13. Sharabi et al. compared SL and oral routes in a randomized prospective study of 30 adults with VB12 deficiency. Participants were given one tablet daily of 500 μg cyanocobalamin orally or sublingually or two tablets daily of a VB12 complex (250 μg cobalamin, 100 mg thiamine, and 250 mg pyridoxine) for 8 weeks. After 4 weeks, VB12 level normalized, and serum cyanocobalamin levels were increased from 108 to 286 pmol/L, 94 to 288 pmol/L, and 98 to 293 pmol/L, in SL, oral, and oral B-complex groups, respectively (p = 0.0001). The authors reported that SL route is as effective as the oral route in treating VB12 deficiency20. In a prospective open-labeled study, Delpre et al. tested 18 patients with VB12 deficiency. In their study, the patients received VB12 (2000 μg/day for 7-12 days) by SL route, and serum VB12 level normalized rapidly and significantly in all the patients14.
To the best of our knowledge, this is the first study to compare two treatment methods (SL and IM) and two cobalamin types (cyanocobalamin and methylcobalamin) simultaneously in children with VB12 deficiency. Our results are consistent with those of previous studies. We found that SL cyanocobalamin and methylcobalamin were as effective as IM cyanocobalamin in treating VB12 deficiency in children. The serum VB12 level normalized in all the patients after 4 weeks. The mean VB12 level was found to be the highest in IM cyanocobalamin group and the lowest in SL cyanocobalamin group after the treatment.
Hematologic response time and degree to VB12 treatment vary according to the basal VB12 level of patients21. Anemia (Hb and mean corpuscular volume [MCV]) usually begins to improve in 1-2 weeks and normalizes within 6-8 weeks21,22. Sezer et al. treated 135 children (aged between 1 month and 18 years) with VB12 deficiency. Treatment protocols were as follows: IM cyanocobalamin group 100 μg/day for 1 week, then 1000 μg on alternate days for a week, then 1000 μg 2 times a week for a week and finally once a week and oral group one B-complex tablet (50 mg thiamin, 250 mg pyridoxine, and 1000 μg cyanocobalamin) per day up to 1 month. After the 1st month of treatment, Hb levels normalized and MCV decreased in 34% of patients in IM group and 19% of patients in the oral group23. Verma et al. carried out a prospective study in 28 children (aged between 6 months and 18 years) with macrocytic anemia, and oral methylcobalamin was given at a dosage of 30 μg/kg/day for 1 month. After the treatment, Hb levels normalized in 85.7% of the patients24. In another study conducted by Andres et al., 30 patients were treated with oral VB12 (250-1000 μg/day) for >1 month. After the 1st month of treatment, anemia improved in only 54% of the patients25.
Our results were in line with previous studies. After the 1st month of the treatment, the improvement in anemia was mostly detected in IM cyanocobalamin group (46.4%), whereas the least improvement was observed in SL cyanocobalamin group (30.4%). However, there was no statistical difference between the three groups. We thought that these differences between the studies were probably because of the case selection, follow-up time, treatment method, and drug selection.
The retrospective design is the most limiting factor of the study. Therefore, we did not investigate methylmalonic acid and homocysteine levels. In addition, short follow-up period and relatively small sample size are another limitations of the study.
In conclusion, SL cyanocobalamin and methylcobalamin are as effective as IM cyanocobalamin in correcting serum VB12 levels and hematologic abnormalities in children with VB12 deficiency. However, SL formulations, which are cheaper, safer, painless, and practical, have been less used than IM formulations for decades. Our results suggest that SL formulations can be used as the first-line treatment in children with VB12 deficiency.











 nueva página del texto (beta)
nueva página del texto (beta)