INTRODUCTION
Among the various contraceptive methods, emergency hormonal contraception (EHC) represents an option for preventing unwanted pregnancies, particularly in cases of violence and/or unprotected intercourse1. EHC is the combined administration of estradiol (E2) and progesterone (P4), or the administration of a P4 agonist alone, such as levonorgestrel (LNG), within the first 72 h after intercourse. The effectiveness of the method depends on the phase of the cycle in which intercourse and treatment take place. The efficacy is estimated in 75%, that is, three out of every four expected pregnancies are prevented. The delay in treatment significantly decreases the effectiveness of the method2-4. Regarding the mechanisms of action of the EHC, most studies indicate that the efficacy of LNG lies in its ability to affect ovulation, which paradoxically is greater in the days of the cycle with less probability of pregnancy and lower in the phase with greater probabilities5,6. It is, therefore, possible that other mechanisms, in addition to the inhibition of ovulation, may explain its contraceptive effect. The primary objective of this study was to evaluate the effects of LNG given at the time of follicular rupture (FR) on gene expression in endometrial biopsies obtained in the luteal phase of the menstrual cycle at the time of endometrial receptivity and implantation.
METHODS
This study was approved by the Ethics and Research Committees of the National Institute of Medical Sciences and Nutrition Salvador Zubirán and all volunteers signed an informed written consent form. Ten healthy volunteer women under non-hormonal contraception (bilateral tubal ligation, vasectomized couple, or sexual abstinence) aged between 22 and 35 years (29 ± 4.5) were included in the study. All women had regular menstrual cycles lasting 28.5 ± 3.95 days (24-37 days) with no history of contraceptive or other medication use in the past 6 months before the study. Volunteers were studied during two consecutive menstrual cycles, one control (C) and one randomized treated (T). In the last cycle, on the day of FR, they received LNG (T-LNG) at the dose of 1.5 mg (5 women) or mifepristone (T-MFP) at the dose of 50 mg (5 women) orally. FR (ovulation) was documented by serial transvaginal ultrasounds performed by a single observer with an ACUSON × 150 Siemens device (Siemens Medical Solutions CA, EU). Six days after FR, samples of endometrial tissue were obtained from the C and T groups under local anesthesia for further processing and identified as C-LH+ 7 (n = 10), T-LNG (n = 5), and T-MFP (n = 5), respectively. Endometrial samples were taken from the posterior wall of the uterine cavity, and blood samples from the antecubital vein for the quantification of serum P4 and E2. The endometrial tissue was processed for histological analysis using the criteria described by Noyes7, as well as for the extraction of total RNA.
Hormonal determinations
Serum levels of luteinizing hormone (LH), E2, and P4 were quantified in blood samples obtained during the follicular and luteal phases of the menstrual cycle by employing specific immunoassays, as previously reported8,9.
RNA isolation, preparation
RNA was obtained by the TRIzol method (Invitrogen, Carlsbad, CA, USA) and processed as previously described10, briefly: each endometrial tissue sample was homogenized in 1 mL of TRIzol and 200 μL of chloroform were added. The aqueous phase was purified on Qiagen RNeasy columns, according to the manufacturers instructions (QIAGEN LLC, MD, US). The RNA samples were treated with DNAse 1 (Ambion-Applied Biosystems, CA, US) and their quality was determined using an Agilent 2100 Bioanalyzer (Agilent Technologies, Inc., Palo Alto, CA, USA), and total RNA quantified in an ND-1000 Spectrophotometer (NanoDrop Technologies, Wilmington, DE, USA). To generate amplified and biotilinated cDNA fragments for analysis in expression microarrays, the Affymetrix GeneChip Whole-Transcript PLUS (WT) Sense Target Labeling Case (Affymetrix, Inc., Santa Clara, CA, USA) was used. The biotin-labeled fragments were hybridized to the GeneChip Human Genome U133 Plus 2.0 microarrays (Affymetrix, Inc.) for 17 h at 45°C. After the incubation period, the microarrays were washed with high and low astringency solutions for streptavidin-phycoerythrin staining, according to the Affymetrix Fluidics Station 450 FS450_0007 protocol. Fluorescence signals were analyzed using the GeneChip Scanner 3000 7G (Affymetrix, Inc.), whose intensity and quality data were obtained with the Expression Console Program (Affymetrix, Inc.).
Microarray data analysis
Microarray raw data were background-corrected using Robust Multiarray Average method11 and normalized using quantile Normalization12. Analyses involved two contrasts (T-LNG vs. C-LH+ 7 and T-MFP vs. C-LH+ 7) and the differentially expressed genes (DEG) were determined by linear statistical models with arbitrary coefficients using the Bioconductor library Limma13,14. Correction for multiple hypotheses was applied using a false discovery rate (FDR). The up- and down-DEG were selected on the basis of a fold-change |FC| > 2 and a p < 0.01. All analyses were performed using R software (v. 3.2.1, 2015).
Gene ontology (GO) analysis was done by FatiGO of Babelomics 5.0 (http://babelomics.bioinfo.cipf.es) using GO terms generated by the list of DEG, as previously described15,16. The nomenclature of the enriched biological processes (BPs) included terms of the GO Consortium17. The adjusted p-values were calculated by an exact Fisher test that evaluated the significant overrepresentation of functional terms in the list of DEG with respect to the rest of the human genome after correcting for multiple tests (multiple hypotheses, one for every functional term) using FDR18.
The analysis of selected bio-functions associated with endometrial receptivity was conducted using Ingenuity Pathway Analysis (IPA; http://www.ingenuity.com). Bio-functions were considered relevant when an absolute z-score > 2 and a p < 10E-3 were fulfilled. Furthermore, based on a hypothesis-driven approach of the DEG in T-LNG versus C-LH+ 7 contrast on endometrial receptivity bio-functions, a molecule activity prediction (MAP) analysis by IPA was done as previously described8,19,20.
Real-time quantitative polymerase chain reaction (RT-qPCR)
The cDNA was obtained from 1 μg of total RNA by reverse transcription and oligo-dT following the indications of the Roche First Strand cDNA Synthesis Transcriptor commercial kit (Roche, Mannheim, Germany) in a LightCycler® 2.0 thermal cycler (Roche). For the total RNA RT-qPCR, the LightCycler TaqMan Master of Roche and TaqMan Assay (Ambion-Applied Biosystems) commercial kits were used. The cDNAs were amplified in a final volume of 10 μL, using a reaction mixture containing PCR grade water, cDNA, initiators, and TaqMan DNA polymerase (Master Mix, 5X). The glass capillary mixture was then centrifuged and placed in the thermal cycler. The oligonucleotides for real-time amplification of each gene were designed based on the manufacturers online program (Universal Probe Library Assay Design Center (https://www.roche-applied-science.com) and their alignment with the sequence of interest was verified in the NCBI Blast page (http://www.ncbi.nlm.nih.gov/BLAST/); TaqMan hydrolysis probes (Universal Probe Library, Roche) were used. Parallel incubations in the presence of double-distilled water or in the absence of reverse transcriptase were included as negative controls.
The relative abundance of gene expression was obtained by the formula 2-ΔΔCT using GAPDH as the constitutive gene for results normalization21.
RESULTS
Characteristics of the menstrual cycle
Based on the serum LH, E2, and P4 levels throughout the menstrual cycle (data not shown), and menstrual cycle characteristics of volunteers in Groups C and T (Table 1), the presence of ovulatory cycles was established. All groups presented the mid-cycle LH peak with E2 and P4 serum levels at mid-luteal phase (LH+ 7) above 100 pg/mL and 8 ng/mL, respectively, and as shown in table 1, similar luteal phase duration in all groups was observed.
Table 1 Clinical characteristics of menstrual cycles C (control) and T (treated)
| Cycle variable | C-LH+ 7 (n = 10) | T-LNG (n = 5) | T-MFP (n = 5) |
|---|---|---|---|
| Follicular phase (days) | 15.60 ± 4.27 [14] | 12.80 ± 1.92* [5] | 15.60 ± 2.70 [6] |
| Luteal phase (days) | 12.90 ± 0.99 [7] | 12.60 ± 0.54 [1] | 13.02 ± 2.55 [6] |
| Menstrual cycle length (days) | 28.50 ± 3.95 [13] | 25.40 ± 2.19* [6] | 28.60 ± 2.70 [7] |
| Maximum follicular diameter (mm) | 22.61 ± 1.41 [5.1] | 19.12 ± 7.15 [17] | 22.88 ± 2.53 [6] |
| Follicule rupture (day) | 14.70 ± 2.94 [9] | 12.80 ± 1.92 [5] | 15.60 ± 2.70 [6] |
Results are expressed as the mean ± SD; [Range];
*p < 0.05 versus C-LH+ 7.
Profile of differential expression of genes in endometrial tissue of women treated with LNG and MFP
To determine the effects of LNG and MFP on gene expression in endometrial tissue, 20 expression microarrays were used: C-LH+ 7 (ten), T-LNG (five), and T-MFP (five). The unsupervised hierarchical clustering analysis showed the identification of two well-defined groups corresponding to the contrasts T-LNG versus C-LH+ 7 and T-MFP versus C-LH+ 7. In the first group, 1223 genes showed differential expression represented by 599 (48.98%) and 624 (51.02%) up-regulated and down-regulated genes, respectively. The T-MFP versus C-LH+ 7 contrast showed 2334 DEG: 1051 (45.03%) up-regulated and 1283 (54.97%) down-regulated.
Functional analysis of DEG
This analysis, performed with the list of 1223 and 2334 DEG in the endometrium of the T-LNG versus C-LH+ 7 and T-MFP versus C-LH+ 7 contrast, showed 333 and 1763 BP up-regulated, respectively. In both treated groups, the BP embryonic development in utero and female pregnancy showed high statistical significance. On the other hand, other terms of GO significantly associated with endometrial receptivity, such as interleukin (IL)-6 regulation, wound healing, and remodeling of tissues and blood vessels, were identified within the main BPs in group T-LNG. In addition, the inflammatory response was also identified with high statistical significance associated with endometrial receptivity in the T-MFP group, similar to that observed in previous studies from our laboratory8,20.
Results from the IPA of the DEG list in the T-LNG versus C-LH+ 7 contrast showed canonical pathways associated with endometrial receptivity. In the T-MFP versus C-LH+ 7 contrast, this analysis identified several canonical pathways associated with endometrial receptivity such as those for integrins, endothelin-1, chemokines, vascular endothelial growth factor, IL-6, prolactin, and STAT 3, among others. The finding that female pregnancy was one of the BPs derived from the most represented GO terms in both the T-LNG and T-MFP versus C-LH+7 contrast, prompted us to perform an in silico analysis of molecular activity prediction of two important bio-functions associated with endometrial receptivity. Figure 1 shows the prediction analysis of molecular activity (MAP) of the bio-functions uterine decidualization (A) and embryo implantation and the blastocyst (B). In the case of the T-LNG versus C-LH+7 contrast, these three bio-functions significantly associated with endometrial receptivity, and under the control of leukemia inhibitory factor (LIF) expression were predicted as activated, suggesting that the pattern of DEG in the endometrium during the mid-luteal phase of the menstrual cycle (implantation phase) was compatible with implantation. In the case of the T-MFP versus C-LH+ 7 contrast, the MAP analysis of the three bio functions was predicted as inhibited mainly through the down-regulation of STAT3.
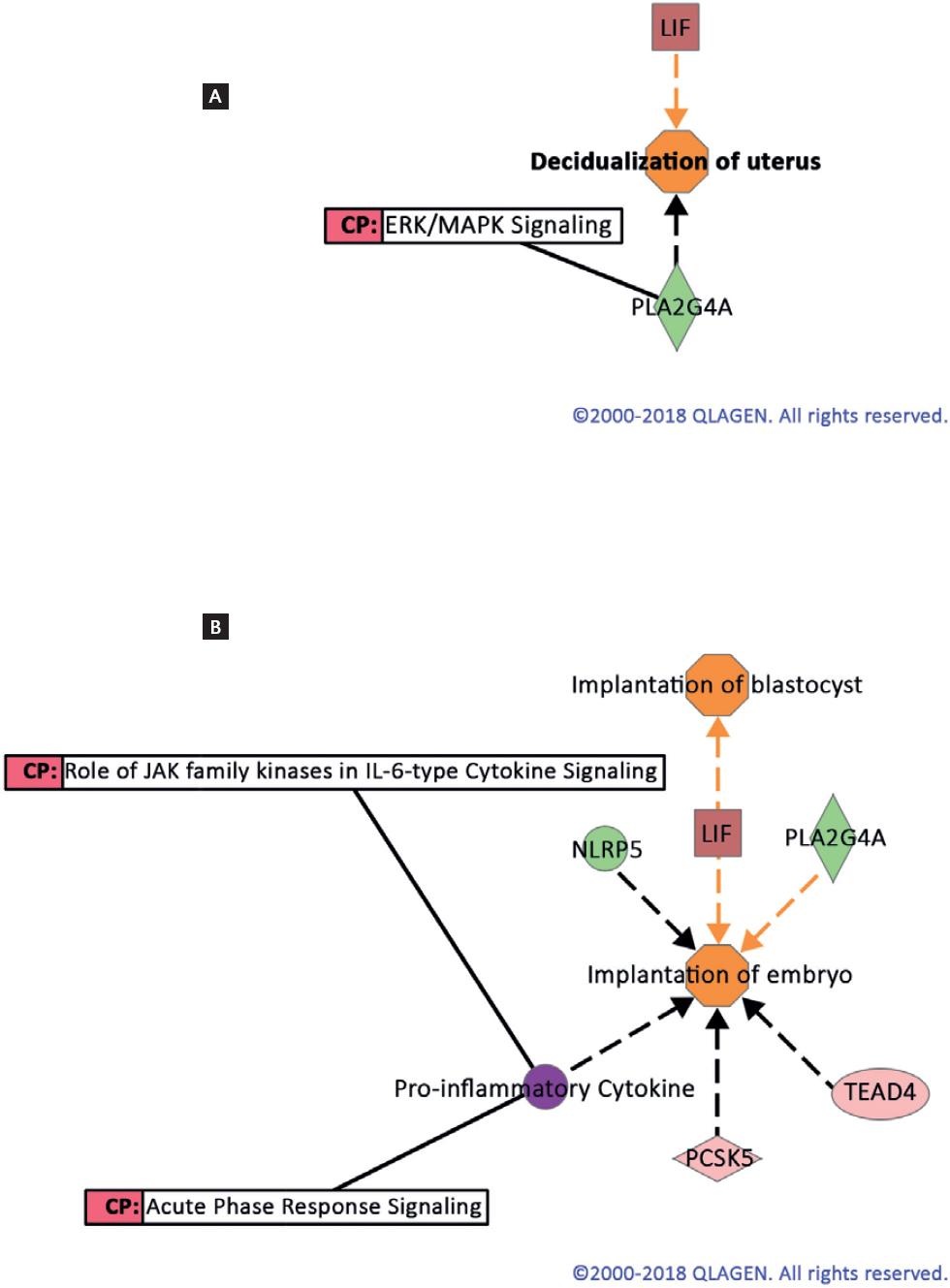
Figure 1 Molecule activity prediction of the bio-functions decidualization of uterus (A) and implantation of embryo (B) using differentially expressed genes in endometrial biopsies at LH+7 from LNG treated versus control cycles. ERK/MAPK signaling was a canonical pathway (CP) involved in the decidualization process. Colors indicated predicted relationships of gene expression levels and bio-functions: Green: down-regulated genes; Red: up-regulated genes; Orange: bio-function activated; Orange line: leads to activation; Gray line: effect not predicted; solid Gray lines: gene associated with CP (http://ingenuity.com).
Biological relevance of the effects of LNG and MFP on the overall expression of the endometrial genome
Figure 2 shows Venn diagrams obtained from the results of the present study (T-LNG and T-MFP versus C-LH+ 7) and those from the so-called endometrial receptivity array (ERA), a customized microarray successfully being used for the diagnosis and treatment of women with infertility22. This analysis shows 60 and 87 common genes when T-LNG or T-MFP were compared with ERA (Fig. 2A and Table S1 and Fig. 2B and Table S2, respectively), as well as 21 common genes among the three DEG lists (Fig. 2C). Table 2 shows the transcription rate and gene directionality of the 21 common genes among the three DEG lists. As shown, 11 common genes up-regulated in ERA showed the same gene expression directionality in T-LNG, whereas these genes were down-regulated in T-MFP. As shown in Table S1 and S2, all genes up-regulated in ERA were up-regulated in T-LNG and vice versa when compared to T-MFP.
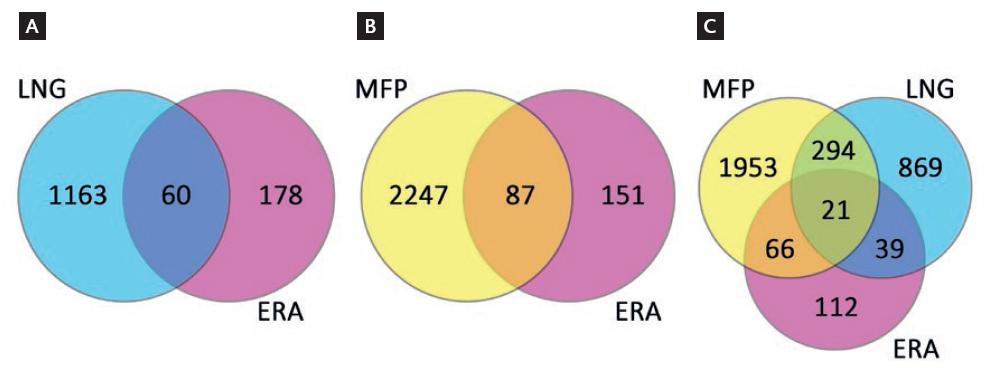
Figure 2 Venn diagrams of endometrial microarrays from T-LNG (A) and T-MFP (B) compared with ERA. Panel C shows common genes among the three microarrays. Intersections show the number of common genes. ERA: endometrial receptivity array22; LNG: levonorgestrel; MFP: mifepristone.
Table 2 Comparison of gene expression directionality of common up- and down- differentially expressed genes in endometrial biopsies from T-LNG, T-MFP and ERA
| Gene symbol | Gene name | ERA (FC) | T-LNG (FC) | T-MFP (FC) |
|---|---|---|---|---|
| C4BPA | Complement component 4 binding protein, alpha | 13.14 | 2.64 | −5.43 |
| GADD45A | Growth arrest and DNA damage-inducible, alpha | 8.25 | 1.69 | −1.66 |
| NNMT | Nicotinamide N−methyltransferase | 7.74 | 1.62 | −1.85 |
| SNX10 | Sorting nexin 10 | 6.56 | 1.93 | −1.53 |
| CP | Ceruloplasmin (ferroxidase) | 6.34 | 1.64 | −3.60 |
| TAGLN | Transgelin | 5.71 | 1.61 | −1.64 |
| THBS2 | Thrombospondin 2 | 5.17 | 1.60 | −2.55 |
| SERPING1 | Serpin peptidase inhibitor, clade G (C1 inhibitor), member 1, (angioedema, hereditary), transcript variant 1 | 5.16 | 1.72 | −2.38 |
| HABP2 | Hyaluronan-binding protein 2 | 4.09 | 5.20 | −1.72 |
| COTL1 | Coactosin-like 1 (Dictyostelium) | 3.58 | 1.52 | −1.42 |
| PROS1 | Homo sapiens protein S (alpha) | 3.28 | 1.45 | −1.40 |
| HLA-DOB | Major histocompatibility complex, class II, DO | −11.06 | −2.35 | −2.55 |
| NR4A2 | Nuclear receptor subfamily 4, group A, member 2, transcript variant 1 | −7.05 | −2.00 | −2.17 |
| DUOX1 | Dual oxidase 1, transcript variant 1 | −6.61 | −3.05 | −3.51 |
| EDN3 | Endothelin 3, transcript variant 3 | −5.30 | −1.45 | 1.78 |
| CAPN6 | Calpain 6 (CAPN6) | −4.76 | −5.17 | 2.53 |
| MMP26 | Matrix metallopeptidase 26 | −4.66 | −5.90 | −8.40 |
| LRRC17 | Leucine-rich repeat containing 17, transcript variant 2 | −4.64 | −1.67 | 1.66 |
| ADAMTS8 | A disintegrin-like and metalloprotease (reprolysin type) with thrombospondin type 1 motif, 8 | −3.59 | −2.48 | −3.78 |
| BIRC3 | Baculoviral IAP repeat-containing 3 (BIRC3), transcript variant 1 | −3.58 | −1.75 | −2.50 |
| LRRC1 | Leucine-rich repeat-containing 1 | −3.23 | −3.16 | 1.66 |
FC: fold change; ERA: endometrial receptivity array22. Negative values mean down-regulation.
Validation of microarray gene expression
For this purpose, specific sense and antisense oligonucleotides were designed for each of the genes to be amplified by RT-qPCR. The magnitude and directionality of some up- and down-regulated genes in the microarray and selected on the basis of their relevance in endometrial receptivity were further confirmed by qPCR (Table 3).
DISCUSSION
LNG is a second-generation synthetic progestin derived from 19-nor testosterone with agonist activity through its binding to P4 nuclear receptors23. This compound is widely used in oral, implant, and intrauterine contraception. Its use in emergency contraception has become widespread due to its high efficacy with limited side effects6. The main pre-ovulatory mechanism of contraceptive action of LNG is at the level of the hypothalamic-pituitary-ovarian axis affecting the release of pituitary LH leading to either inhibition or delay of ovulation5,24-26. In this regard, it is well established that LNG inhibits ovulation when administered in the days before the acute release of LH; however, this effect does not occur when LNG is administered immediately before or during ovulation5. This observation suggests that other mechanisms besides those on ovulation contribute to LNG contraceptive efficacy. The objective of this study was the analysis of LNG effects on gene expression in endometrial tissue at the time of implantation. The results were compared with those obtained in this study with MFP, a well-known P4 receptor antagonist, and with those from the already available microarray assay (ERA) to evaluate endometrial receptivity22, both were included as a positive control for a non-receptive and receptive endometrium, respectively. In this study, LNG administered on the day of FR assured us a normal growth and development of follicles, including the release of LH and ovulation. The results indicated that the administration of 1.5 mg of LNG on the day of FR caused changes in endometrial gene expression consistent with those observed during the stages of endometrial transition from the early to the mid-luteal phase of the cycle27,28. These results suggest that the observed effects of LNG on gene transcription did not apparently modify the receptive capacity of the endometrium induced by natural P4. In addition, the effects on gene expression in the endometrium from women treated with MFP were similar to those observed using another P4 receptor antagonist, ulipristal acetate8. The BP embryonic development in utero derived from the GO analysis, together with female pregnancy, were those enriched with high statistical significance. Similarly, other BPs such as IL-6 regulation, wound healing, and remodeling of tissues and blood vessels, significantly associated with endometrial receptivity, were identified as up-regulated in the T-LNG versus C-LH+ 7 contrast.
These results confirm that the effects of natural P4, produced by the corpus luteum after ovulation, are largely responsible for gene expression during the so-called physiological transcriptional awakening of the endometrium29. Given that female pregnancy was one of the bio-processes most up-regulated in both treated groups, the Ingenuity® Knowledge Database (https://ingenuity.net.au/support/knowledge-base/) was consulted. The objective of this analysis was to predict the molecular activity of the bio-functions uterine decidualization and implantation of the embryo and blastocyst in accordance with the lists of DEG obtained from both T-LNG and T-MFP versus C-LH+ 7 contrast, respectively. As expected, these bio-functions were predicted as activated in the LNG-T and inhibited in the T-MFP group based mainly on the up-regulation of LIF and the down-regulation of STAT3, respectively. In this regard, activation through specific receptors for LIF (LIFR) and recruitment of GP130 (IL6ST) were found related to three significant cytokine-dependent pathways, MAPK, JAK-STAT, and cytokines, which are known to be important for embryo implantation30,31. Transgenic mice who do not express LIF or those without the carboxyl-terminal region of gp-130 are infertile, indicating the importance of this pathway in implantation32. These results suggest that LNG acts as an agonist in the mechanisms of endometrial receptivity through stimulating the expression of relevant genes such as LIF, including its corresponding intracellular signaling components. In the present study, administration of MFP led to inhibition of genes involved in the processes of decidualization and implantation of the embryo, particularly of STAT3, in which the canonical signaling pathways ERK/MAPK, JAK/Stat, IL-15, and the acute phase signaling are shown to be associated with endometrial receptivity20. These observations agree with three previously published studies, in which the post-ovulatory administration of LNG did not change the expected pregnancy rate, nor the expression of genes involved in implantation2,33,34.
In conclusion, this study, the second on this particular issue as far as we know, indicates that LNG did not affect the expression of gene sets that, in the form of a process called transcriptional awakening (implantation), define those P4-driven molecular events involved in developing a receptive phenotype of the human endometrium29,35. In summary, this study further supports that LNG given post-ovulatory might not affect, as MFP does, embryo implantation when required for EHC. (Supplementary Table 1)











 nueva página del texto (beta)
nueva página del texto (beta)


