Introduction
The species included in the genus Pisolithus Alb. & Schwein. (Boletales: Sclerodermataceae) present globose, reniform, or pyriform basidiomes characterized by a peridium with irregular dehiscence and an alveolar gleba conformed by peridioles, which become pulverulent at maturity, with globose, echinulate basidiospores (Burgess et al., 1995). The genus is widely distributed in temperate and tropical regions, forming ectomycorrhizal associations with a wide range of woody plants, highlighting species from the families Pinaceae, Myrtaceae, Fagaceae, Dipterocarpaceae, and Cistaceae (Marx, 1977).
In Mexico, two species of Pisolithus have been cited: P. arhizus (Scop.) Rauschert. and P. tinctorius (Mont.) E. Fisch. The first was reported from the states of Sonora (Esqueda et al., 1990, 1998, 2000; Moreno et al., 2010; Hernández-Navarro et al., 2015), Chihuahua (Moreno et al., 2010), Hidalgo (Bautista-Hernández et al., 2018) and Jalisco (Rodríguez Alcántar et al., 2019), while the second was reported from the states of Aguascalientes (Pardavé, 1991), Chihuahua, Querétaro, Guerrero, Coahuila (Guzmán and Herrera, 1969; 1973), Mexico City (Herrera, 1964), Oaxaca (Welden and Guzmán, 1978), Veracruz (Welden and Guzmán, 1978), Hidalgo (Frutis and Guzmán, 1983), Baja California (Ayala and Guzmán, 1984), Michoacán (Díaz-Barriga et al., 1988), Chiapas (Herrera et al., 1989), Durango (Herrera et al., 1989), Morelos (Herrera et al., 1989), Sonora (Pérez-Silva et al., 1994), Mexico State (Nava Mora and Valenzuela Garza, 1997; Zarco, 1986), Nayarit, Tabasco (García-Rodríguez et al., 2006), Nuevo León (Urista et al., 1985; Garza Ocañas et al., 2023), Tamaulipas (Garza Ocañas et al., 2023), and Zacatecas (Herrera, 1964). However, in all records, the determination of the species was based exclusively on the morphological characteristics of the basidiome, considering the validity status of the species cited at the time. For this reason, both names have been indiscriminately cited worldwide (Chambers and Cairney, 1999).
The description of new species of the genus and its delimitation has been carried out using ITS barcode sequences (Martin et al., 2002, 2013; Reddy et al., 2005; Phosri et al., 2012; Rusevska et al., 2015; Lebel et al., 2018; Abel-Aziz and Bakhit, 2023; Crous et al., 2024). Moreover, based on ITS sequences, it has been reported that P. arhizus is primarily distributed in Eurasia and has been introduced to South Africa (Phosri et al., 2012; Rusevka et al., 2015; Lebel et al., 2018), while P. tinctorius s.s. is distributed in Europe, Canada, Kenya, Nicaragua, and the USA (Díez et al., 2001; Martin et al., 2002; Lebel et al., 2018). Most records of Pisolithus from Mexico correspond to Pinus-Quercus associations (Calonge et al., 2004), with a few records from Pecan Orchards (Carya illinoinensis (Wangenh.) K. Koch) (Sáenz-Hidalgo et al., 2023) and Eucalyptus L’Her. (García Rodríguez et al., 2006). However, neither P. arhizus nor P. tinctorius are associated with Eucalyptus, leading to the need to confirm the Mexican specimens already cited under the name “P. arhizus”.
Our objective was to morphologically and molecularly characterize Pisolithus species based on collections from temperate and arid zones of Mexico preserved in the fungal collections of the herbarium MEXU. In addition, soil metabarcoding sequences were mined to integrate additional information that will contribute to the taxonomy and phylogeny of Pisolithus in Mexico.
Materials and Methods
Fungal material and morphological characterization
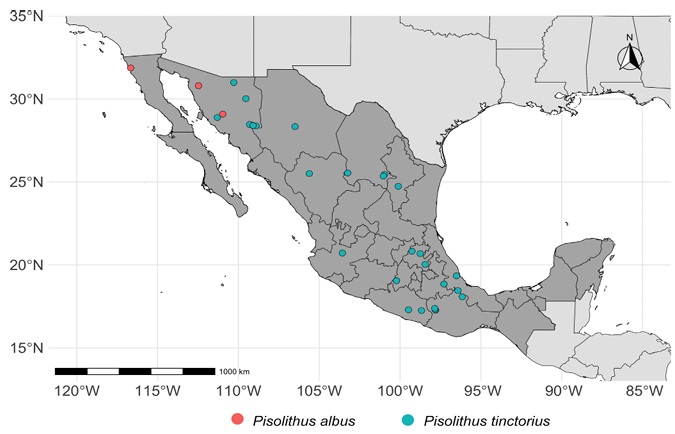
Thirty-two collections labeled as Pisolithus and preserved in MEXU (Appendix 1) were examined. The habitat, potential host, and collection date mentioned in the text are based on field notes or data annotated on the herbarium labels, and a distribution map (Fig. 1) was created using the ggplot2 package in R (Wickham and Chang, 2008). The specimens were photographed, measured, and characterized following the suggestions by Cifuentes et al. (1986).
Portions of the gleba were mounted in 10% KOH and observed under an OLYMPUS IX81 optical microscope (Tokyo, Japan). A portion of the gleba was metalized and visualized under Scanning Electron Microscopy (SEM) in a HITACHI SU 1510 microscope (Hitachi, Japan) in the Laboratorio de Microscopía y Fotografía de la Biodiversidad, of the Laboratorio Nacional de la Biodiversidad (LaNaBio), Instituto de Biología, Universidad Nacional Autónoma de México (IBUNAM).
DNA extraction, PCR amplification, and sequencing
Genomic DNA was isolated from a small portion of the basidiome of 32 species (Appendix 1), placed in a 2 ml tube with a sterilized Tungsten sphere, frozen in liquid nitrogen, and ground with a TissueLyser Lt (QIAGEN, Hilden, Germany). After adding 500 μl of CTAB and 2 μl of β-mercaptoethanol per sample, the tubes were incubated in an Eppendorf ThermoMixer C (Hamburg, Germany) at 45 °C for 30 minutes at 300 rpm. Next, 500 μl of SEVAG (chloroform: isoamyl alcohol; 24:1) was added and incubated in the Thermo Fisher Scientific Clinical Rotator (Massachusetts, USA) for 30 minutes at 85 rpm.
The mixture was centrifuged at 13,000 × g for 10 minutes in an Eppendorf centrifuge 5424R (Hamburg, Germany). The supernatant was transferred to a 1.5 ml tube, and 500 μl of isopropanol was added. The mixture was gently mixed and stored at -20 °C for one hour. Subsequently, the mixture was centrifuged for 10 minutes at 12,000 × g, and the supernatant was discarded. The pellet was washed with 70% EtOH at -20 °C, vacuum-dried for five minutes, and resuspended in 50 μl of ultrapure water. The sample was quantified using a NanoDrop 2000 instrument (Thermofisher Scientific, Waltham, Massachusetts, USA), and its integrity was visually verified on a 1% agarose gel stained with RedGel™.
The ITS5 and ITS4 primer pairs were used to amplify the complete ITS1-5.8S-ITS2 region (Schoch et al., 2012) using the PCR Mix (5’BIO, Mexico). The PCR amplicons were visualized on a 1% agarose gel stained with RedGel™. The successful amplicons underwent treatment with ExoSAP-IT™ according to the manufacturer's instructions. Clean PCR reactions were sequenced at both ends in the Laboratorio de Secuenciación Genómica of LaNaBio-IBUNAM.
Phylogenetic analysis
Twenty new sequences of Pisolithus, and one of Scleroderma albidum Pat. & Trab. were generated (Appendix 1). The ITS rDNA dataset consisted of 127 sequences and 824 positions, of which 150 were conserved, and 614 variable, 211 were singletons, and 401 were informative. For the metabarcoding soil sequences, the search was conducted in the Global Soil Mycobiome Consortium database (Tedersoo et al., 2021) using the awk command to retrieve all rows annotated as Pisolithus in the genus column (column 10). Only two OTU sequences were recovered: 2904dd2b5e2b1bd4c81f5402c904a0e93246127e (SOIL 2904 from Nuevo León (24°43'48.0"N, 100°06'36.0"W)) and 44b8a06bdba1c02587989f9e3302edb4a542d7ae0 (SOIL44b8 from Coahuila (25°21'00.0"N, 101°01'48.0"W)).
The remaining sequences were downloaded from the NCBI database (NCBI, 2024) (Appendix 2); 99 belonged to different species of Pisolithus, four to Scleroderma Pers., and one to Astraeus hygrometricus (Pers.) Morgan as outgroups (Anderson et al., 1998, 2001; Gomes et al., 2000; Díez et al., 2001; Malajczuk y Dunstan, 2002; Martin et al., 2002, 2013; Kanchanaprayudh et al., 2003; Thomas et al., 2003; Reddy et al., 2005; Palmer et al., 2008; Phosri et al., 2009, 2012, 2013; Jourand et al., 2010; Kasuya et al., 2008; Hitchcock et al., 2011; Wilson et al., 2011; Montagner et al., 2015; Crous et al., 2016, 2024; Lebel et al., 2018; Abdel-Aziz and Bakhit, 2023; Patel y Rajput, 2023).
The newly generated sequences were manually curated by inspecting their chromatograms in Geneious Prime v. 2025.0.3 (Geneious Prime, 2025). Sequences were aligned using the online version of MAFFT v. 7 (Katoh et al., 2019). The alignments were reviewed in MESQUITE v. 3.81 (Maddison and Maddison, 2023), followed by minor manual adjustments to ensure character homology among the taxa. Phylogenetic inferences were estimated using the Maximum Likelihood Method in W-IQ-TREE (Trifinopoulos et al., 2016), and ModelFinder (Kalyaanamoorthy et al., 2017) was used to select the best substitution model. Bayesian inference analysis was conducted using MrBayes v. 3.2.6 × 64 (Huelsenbeck and Ronquist, 2001).
The information block for the matrix included two simultaneous runs, four Monte Carlo chains, a temperature set at 0.2, and a sampling of 10 million generations (standard deviation ≤0.1) with trees sampled every 1000 generations. The two simultaneous Bayesian runs continued until convergence parameters were met, and the standard deviation fell below 0.0001 after 10 million generations. The final tree was edited using FigTree v. 1.4.4 (Rambaut, 2018).
Results
The 32 collections studied and preserved under Pisolithus come from 23 municipalities of 12 states of the Mexican Republic (Fig. 1, Appendix 1). After examining the collections, we determined that the specimens correspond to two well-defined species: Pisolithus albus (Cooke & Massee) Priest, a new record for Mexico, associated with Eucalyptus in the semiarid zones of Northwest Mexico, and P. tinctorius associated with Pinus spp., Quercus spp., and Carya illinoinensis, with a broader distribution in our country.
We successfully amplified and sequenced the ITS region of 21 Pisolithus collections, and the data of Tedersoo et al. (2021) resulted in two consensus sequences of P. tinctorius from Coahuila and Nuevo León. One collection (MEXU 11889) deposited under the name “P. arhizus” was analyzed using both molecular and morphological methods. After our examination, we concluded that this collection corresponds to Scleroderma albidum.
The result of Posterior Probability (PP) from Bayesian inference and bootstrap from maximum likelihood (BS) generated trees with similar topologies; the topology of the Bayesian analysis is shown (Fig. 2). The genus Pisolithus recovered as monophyletic (PP/BS=0.70/92). It presents three distinct lineages: the first comprises seven species (PP/BS=0.74/90): P. arhizus, P. capsulifer (Sowerby) Watling, Phosri & M.P. Martín, P. calongei M.P. Martín, Phosri & Watling, P. hypogaeus S.R. Thomas, Dell & Trappe, P. marmoratus (Berk.) E. Fisch, P. orientalis Watling, Phosri & M.P. Martín and P. tinctorius. Most sequences generated in this work grouped with P. tinctorius with high support (PP/BS=0.99/100) and are associated with Pinus spp., Quercus spp., and Carya illinoinensis. The second lineage includes five species (PP/BS=0.97/ 95): P. albus, P. croceorrhizus P. Leonard & McMull.-Fish., P. microcarpus (Cooke & Massee) G. Cunn., P. thermaeus T. Lebel, Pennycook & Beever, and P. tympanobaculus T. Lebel & M.D. Barrett. Three sequences generated in this study grouped with P. albus (PP/BS=0.99/95), a new record for Mexico. Two species form an unresolved polytomy: P. abditus Kanch., Sihan., Hogetsu & Watling, and P. madagascariensis Rivas-Ferreiro, Dentinger, Suz & A.M. Ainsw. The third lineage (PP/BS=0.99/94) is composed of P. aureosericeus M.P. Martín, Kaewgraj., Phosri & Watling, and P. aurantioscabrosus Watling. The P. indicus sequence was grouped within the Scleroderma genus (PP/BS=0.6/100).
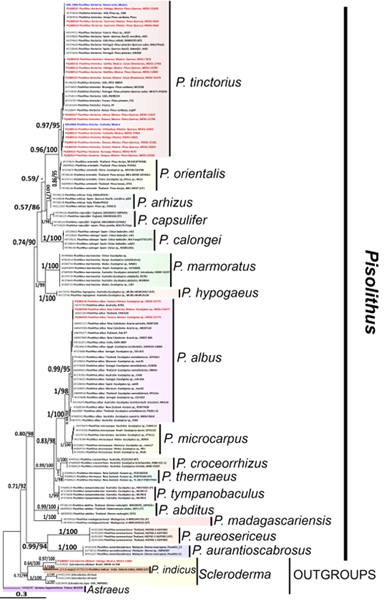
Figure 2: Phylogram of Bayesian inference (BI) tree from the ITS sequence data of Pisolithus Alb. & Schwein. The numbers above branches represent Bayesian Posterior Probabilities (PP=0-1), and Bootstrap Values (BS=0-100) for Maximum Likelihood. The scale bar represents the expected number of nucleotide substitutions per site. The taxa whose sequences have been obtained in this study are marked in bold red. Metagenomic soil sequences are marked in blue. Accession numbers of NCBI (2024) are indicated in each sequence.
Taxonomy
Basidiomycota
Agaricomycetes
Boletales
Sclerodermataceae
Pisolithus albus (Cooke & Massee) Priest, in Lebel, Pennycook and Barrett, Phytotaxa 348(3): 167. 2018. Fig. 3.
≡ Polysaccum albumCooke and Massee, Grevillea 20 (no.94): 36. 1891.
TYPE: AUSTRALIA. Queensland, Dundoo, on the ground, undated, Martin 916 (holotype K!).
Basidiome epigeous or semi-epigeous, 45-110 mm wide × 65-190 mm tall × 30-120 mm diameter, subglobose, ovoid to pyriform, with a robust and rooted base; peridium thin, smooth, cracking into irregular segments from top to bottom at maturity, whitish when young, cream or beige when mature; gleba conformed into peridioles 2-5 mm diameter, subglobose to ovoid, cream-colored when young and turning ochre to olivaceous when mature; basidiospores 7-11 µm, globose, echinulate, bright yellowish, in SEM isolated straight to slightly curved spines up to 1 µm high; basidia not observed.
Habit: on soil, associated with Eucalyptus spp.
Distribution: Africa: Egypt (Abdel-Aziz and Bakhit, 2023), Morocco (Eddine Bakkali Yakhlef et al., 2009), Senegal (Duponnois and Bâ, 1999), Tunisia (Jaouani et al., 2015). America: Mexico (this study), USA (Phosri et al., 2012). Eurasia: India (Singla et al., 2004), Italy (Gargano et al., 2018), Malaysia (Martin et al., 2002), Spain (Díez et al., 2001), Thailand (Kanchanaprayudh et al., 2003). Oceania: Australia (Lebel et al., 2018), New Caledonia (Hosaka, 2009), New Zealand (Moyersoen et al., 2003).
Studied material: MEXICO. Baja California Norte, municipio Ensenada, 31°52'12.9"N, 116°39'59.0"W, under Eucalyptus sp., 25.I.2019, E. Hernández-Navarro 774 (MEXU 31677). Sonora, municipio Caborca, 30°47´46"N, 112°27´56"W, under Eucalyptus sp., 18.IV.2019, R. Gutierrez and A. Gutierrez s.n. (MEXU 31773). Municipio Hermosillo, 29°04´53"N, 110°58´07"W, under Eucalyptus sp., 06.IX.1995, M. Esqueda s.n. (MEXU 31772).
Notes: Pisolithus albus is recognized by its white exoperidium, olivaceous gleba, and basidiospores 7-11 µm diameter with isolated, low, straight spines. The P. albus specimens in this study were always collected under Eucalyptus sp.; however, this species is also associated with Acacia s.l. and Kunzea Benth. (Abdel-Aziz and Bakhit, 2023). This is the first record of P. albus from Mexico. Mexican specimens, mainly from Caborca, Sonora, are stouter (up to 19 × 11 cm diameter) than reported in the holotype (5-6 cm diameter) (Cooke, 1891). However, materials from Italy are 3-20 cm diameter (Gargano et al., 2018), while those from Tunisia are 3-12 cm diameter (Jaouani et al., 2015), and those from Egypt are 4.5-15 × 6-18 cm diameter (Abdel-Aziz and Bakhit, 2023). Additionally, in the holotype, basidiospores are 9-10 µm, while our materials are 7-11 µm diameter; however, it matches with Egyptian materials (7-11 µm diameter), while Tunisians are 9-12 μm diameter and Italians are 8.75-10.25 µm diameter (Jaouani et al., 2015; Gargano et al., 2018; Abdel-Aziz and Bakhit, 2023).
Young specimens of P. albus may recall P. croceorrhizus, as both species are commonly associated with Eucalyptus trees. However, P. croceorrhizus has a distribution practically restricted to Australia (Southeast Queensland) and New Caledonia. Moreover, P. albus presents larger basidiospores (7-12 μm diameter) with irregular pyramidal spines. In contrast, P. croceorrhizus presents smaller basidiospores (5.3-6.9 μm diameter) densely covered with pyramidal spines connected basally in a low reticulum (Leonard et al., 2013).

Figure 3: Pisolithus albus (Cooke & Massee) Priest. A. specimen in its habitat (MEXU 31677); B. peridioles (MEXU 31677); C-D. herbarium specimens (MEXU 31772); E. basidiospores in light microscopy (MEXU 31772); F.-G. basidiospores in SEM (MEXU 31677).
Pisolithus tinctorius (Mont.) E. Fisch., in Engler and Prantl, Nat. Pflanzenfam., Teil I (Leipzig) 1: 338 (1900). Fig. 4.
=Polysaccum tinctorium Mont., in Webb and Berthelot, Hist. nat. Iles Canar. (París) 3(2): 87. 1840.
=Lycoperdodes canariense Kuntze, Revis. gen. pl. (Leipzig) 2: 859. 1891.
TYPE: SPAIN. Canary Islands, without precise location, at the roots of Cistus, s.f., coll. Despréaux s.n. (holotype: not located).
Basidiomes epigeous, globose, claviform to irregularly shaped, subglobose, ovoid or pyriform, 35-90 mm wide × 55-150 mm high × 30-120 mm diam, with a robust or sometimes sessile base; peridium thin, papery, smooth, and brittle, yellowish-whitish when young, brown when mature; dehiscense through irregular cracks; gleba reddish-brown to dark brown, structured in peridioles that become powdery with maturity; peridioles 2-5 mm, subglobose, ovoid or irregular and somewhat angular, creamy when young and becoming yellowish when mature, smaller and more compact towards the stipe; basidiospores 8.5-12 µm, globose, strongly echinulate, dark ochre; in SEM spines primarily straight, coalescent, some slightly curved towards the apex; basidia not observed.
Habit: on soil, associated with Pinus-Quercus and Carya illinoinensis.
Distribution: Africa: Kenya (Martin et al., 1998), South Africa (Martin et al., 2002). America: USA (Gomes et al., 2000; Martin et al., 2002; Phosri et al., 2012), Mexico (Herrera, 1964; this study), Nicaragua (Martin et al., 2002). Eurasia: France (Gomes et al., 2000), Philippines (Anderson et al., 2001), Portugal (Martin et al., 2002), Spain (Díez et al., 2001; Phosri et al., 2012).
Studied material: MEXICO. Chihuahua, municipio Cd. Cuauhtémoc, km 67 Chihuahua - Ciudad Cuauhtémoc road, 28°26'57.8"N, 106°52'10.1"W, under Quercus, 25.IX.1978, E. Pérez-Silva y R. Hernández s.n. (MEXU 12463). Ciudad de México, alcaldía Cuajimalpa de Morelos, Desierto de los Leones, 2900 m, 19°17'51.4"N, 99°18'56.0"W, under Pinus sp., 02.VII.1950, T. Herrera s.n. (MEXU 1409). Coahuila, municipio Saltillo, 25°26'56.3"N, 100°57'44.4"W, under Carya illinoinensis, 10.VIII.1982, T. Herrera s.n. (MEXU 17901). Municipio Torreón km 22 Torreón - Matamoros road, 25°32'18.9"N, 103°14'13.3"W, agricultural land, 12.VIII.1982, T. Herrera s.n. (MEXU 17902). Durango, municipio Tepehuanes, Sierra de la Candela, 25°30'10.6"N, 105°36'56.9"W, 17.IX.1960, F. Sánchez s.n. (MEXU 4570). Estado de México, municipio Temascaltepec, 1100 m, 19°02'37.1"N, 100°13'30.2"W, in Pinus-Quercus forest, 12.I.1978, R. Hernández s.n. (MEXU 12005). Guerrero, municipio Chilpancingo de los Bravo, Rincón Viejo, 17°14'40.5"N, 98°40'30.2"W, in Pinus-Quercus forest, 13.VI.1963, H. Kruse s.n. (MEXU 4569); 17°17'28.8"N, 99°28'46.0"W, in Pinus-Quercus forest, 21.VI.1963, H. Kruse s.n. (MEXU 4566). Hidalgo, municipio Cardonal, 45 km east of Ixmiquilpan, 2000 m, 20°38'53.4"N, 98°57'52.5"W, 02.X.1980, R. Hernández y D. Rodríguez s.n. (MEXU 16021). Municipio Metztitlán, 20°40´30" N, 98°45´09"W, in Pinus-Quercus forest, 11.X.2023, E. Hernandez-Navarro 720 (MEXU 31650). Municipio Santiago Tulantepec de Lugo Guerrero, 2370 m, 20°03'35.5"N, 98°26'07.3"W, in Pinus-Quercus forest, 03.VII.1979, R. Hernández s.n. (MEXU 13200). Municipio Zimapán, La Majada, 20°49'08.6"N, 99°15'33.6"W, in Pinus-Quercus forest, 06.X.1979, R. Hernández s.n. (MEXU 13790). Jalisco, municipio Zapopan, Cañón de las Flores, km 25 Guadalajara - Tequila road, 1500 m, 20°40'27.9"N, 103°23'37.1"W, in Pinus-Quercus forest, 25.VIII.1977, R. Hernandez s.n. (MEXU 11637). Oaxaca, municipio Acatlán de Pérez Figueroa, 2100 m, 18°26'44.8"N, 96°25'18.0"W, in Pinus-Quercus forest, 23.IV.1976, E. Pérez-Silva et al. s.n. (MEXU 10485); La Carbonera, 19°02'37.1"N, 100°13'30.2"W, in Pinus-Quercus forest, 23.VI.1976, E. Pérez-Silva et al. s.n. (MEXU 10996). Municipio San Juan Mixtepec, 8 km from la Cañada El Águila, 1900-2250 m, 17°16'09.6"N, 97°48'07.2"W, in Pinus-Quercus forest, 03.X.1989, E. Pérez-Silva et al. s.n. (MEXU 22336); 17°18'20.5"N, 97°49'54.7"W, under Pinus-Quercus, 02.X.1988, J. Reyes-Santiago s.n. (MEXU 21301); Cerro del Rio Azucena, 2000 m, 17°22'52.1"N, 97°51'20.9"W, Pinus-Quercus forest, 21.VII.1989, J. Reyes Santiago s.n. (MEXU 21768). Municipio Tuxtepec, 18°04'20.3"N, 96°08'52.8"W, under Pinus caribaea, 31.I.1974, R. Singer s.n. (MEXU 8969). Sonora, municipio Cananea, 30°59'23.6"N, 110°17'19.9"W, in Pinus-Quercus forest, 10.IX.1993, E. Pérez-Silva y M. Esqueda s.n. (MEXU 23820); km 18 Santa Rosa - Yécora road, 28°26'03.8"N, 109°10'34.3"W, under Quercus sp., 05.VIII.1989, A. Aparicio s.n. (MEXU 22604). Municipio Hermosillo, 28°52'48.8"N, 111°18'14.2"W, under Carya illinoinensis, 03.X.2019, E. Hernández-Navarro 746 (MEXU 31676). Municipio Yécora, Rincón de Güerigo, 28°26'03.8"N, 109°10'34.3"W, under Quercus sp., 26.VII.1989, A. Aparicio s.n. (MEXU 22605); 28°23´41"N, 109°05´26"W), under Pinus sp., 10.IX.1995, M. Esqueda et al. s.n. (MEXU 31778); 28°23´41"N, 109°05´26"W, Pinus-Quercus forest, 13.IX.1996, M. Esqueda et al. s.n. (MEXU 31777). Veracruz, municipio Maltrata, Cumbres de Maltrata, a 1700 m, 18°50'21.2"N, 97°17'08.8"W, under Quercus, 28.VIII.1969, R. Hernandez y R.C. Trigos s.n. (MEXU 7877); Cumbres de Maltrata, a 1600 m, 18°50'21.2"N, 97°17'08.8"W, under Quercus, 28.VIII.1969, R. Hernández y R.C. Trigos s.n. (MEXU 7876). Municipio Emiliano Zapata, 950 m, 19°20'29.3"N, 96°30'33.0"W, under Quercus, 10.VII.1974, R. Hernández s.n. (MEXU 9575).

Figure 4: Pisolithus tinctorius (Mont.) E. Fisch. A. specimen in its habitat (MEXU 31650); B-C. herbarium specimens (MEXU 4569, MEXU 13790); D. basidiospores in light microscopy (MEXU17902); E-F. basidiospores in SEM (MEXU-31650).
Notes: Pisolithus tinctorius could be confused with P. arhizus, which has the same spore size and is associated with Pinus L. and Quercus L. plants. Rusevska et al. (2015) mention that P. arhizus and P. tinctorius are so similar that it is almost impossible to separate these species morphologically. However, based on materials from Macedonia, Rusevska et al. (2015) mention that P. tinctorius presents a shorter pseudostipe (40-50 × 20-25 mm), a very thin yellowish olivaceous peridium, and basidiospores with spines on average of 1.6 μm long, more or less isolated; while P. arhizus presents a larger pseudostipe (≤95 mm × (20-)25-40(-50) mm), pale ochre to brown black peridium, and the spines of the basidiospores are smaller in average (1.2 μm), isolated but very compact, in parts forming small pyramids (Rusevska et al., 2015). Nevertheless, molecular data support two distinct taxa restricted in distribution in the northern hemisphere: P. tinctorius s.s. is distributed in Europe and North America, while P. arhizus occurs in Eurasia and has been introduced in South Africa (Lebel et al., 2018).
The species P. tinctorius could also be confused with P. marmoratus, as they share the size of their basidiospores and spines. However, the latter is associated with Eucalyptus and is distributed in the southern hemisphere (Martin et al., 2002). Additionally, P. marmoratus presents a peridium with black warts and black pseudostipes (Leonard et al., 2013).
Discussion
We present the first formal taxonomic study of Pisolithus in Mexico based on morphological and molecular data of fungarium specimens. According to Index Fungorum (2024), the genus currently includes 19 valid species. However, some controversies persist regarding the correct identification, nomenclature, and possible synonymy of some species.
Since the description of Pisolithus, with P. arenarius Alb. & Schwein. as the type species, Albertini and Schweinitz (1805) denoted the lack of detailed information on closely related species, then placed in Scleroderma such as Scleroderma arhizum (Scop.) Pers. (=P. arhizus) and S. tinctorium Pers. (=P. tinctorius), to which P. arenarius most closely resembled. Although P. arenarius is a valid species, it still requires more up-to-date information on its macromorphological variation, and specimens in various herbaria have not been reexamined or sequenced for verification.
For a long time, P. arhizus was considered conspecific with P. tinctorius. Consequently, both species were cited interchangeably worldwide (Chambers and Cairney, 1999). Later, phylogenetic studies using ITS barcode sequences confirmed them as distinct species: P. arhizus is primarily distributed in Eusaria and introduced to South Africa, and P. tinctorius s.s. is found in Europe and North America (Leonard et al., 2013; Martín et al., 2013).
Numerous fungal collections and sequences in public databases require updating (Lebel et al., 2018). Pisolithus tinctorius originally was described under the genus Polysaccum Mont., associated with Cistus L. from the Canary Islands; however, global identification of the species has primarily followed Coker and Couch (1928), focusing on samples related to Pinus-Quercus forest, but P. tinctorius (Pers.: Pers.) Coker & Couch turned out to be an illegitimate name for a taxon nowadays recognized as P. arhizus.
In the case of P. kisslingii E. Fisch., no barcode sequences or recent collections exist, while P. indicus Natarajan & Senthil is represented by a single sequence in NCBI (2024) which, in our analyses, grouped with Scleroderma. This species was described from India as having grayish gleba, a characteristic not reported in any other Pisolithus species (Reddy et al. 2005). The combination of characters and the sequence analysis suggests that this could be a Scleroderma species. Additionally, Reddy et al. (2005) did not provide photographs of the peridioles, no sequences of Scleroderma were included in the analysis, and the clade containing P. indicus grouped with the rest of the Pisolithus species with low support (BS= 36). Leonard et al. (2013) included the P. indicus sequence, which was grouped with P. aurantioscabrosus. Nonetheless, no Scleroderma sequences were included.
Lebel et al. (2018) included some LSU sequences of P. albus, P. croceorrhizus, and P. indicus; the latter was placed as a basal taxon, paraphyletic to P. aurantioscabrosus and P. aureosericeus. Nevertheless, they did not include any Scleroderma sequences in the analysis. On the other hand, most published phylogenies present low support values in the deepest branches of the genus, as only ITS sequences have been analyzed, except for the one proposed by Lebel et al. (2018), which included some LSU sequences. Hence, other markers, such as EF1α or RPB1, are needed to resolve these ambiguities within the genus.
In our studied materials, P. tinctorius was primarily collected from Pinus-Quercus, except for two collections found under Carya illinoinensis (MEXU 17901; 31676). Both collections originate from arid zones transformed into commercial pecan agricultural lands with abundant canopy, organic matter, fertilization, and irrigation, which favor the development of P. tinctorius despite the high temperatures of the zone. This aligns with reports that P. tinctorius can be associated with C. illinoinensis (Sáenz-Hidalgo et al., 2023). This is due to soil and plant inoculation practices, as P. tinctorius has historically been used in forestry to promote tree growth (García-Rodríguez et al. 2006). As a result, it has been introduced globally (Lebel et al., 2018). This adaptability explains its presence in diverse environments, including acidic soils and areas with heavy metal contamination (Lampky and Peterson, 1963; Sebastiana et al., 2020).
Regarding P. albus, a new record for Mexico, the specimens from Sonora and Baja California, whose sequences are similar to those from Senegal and Egypt, are also associated with Eucalyptus spp., a widely distributed introduced genus (Abdel-Aziz and Bakhit, 2023). Hence, the presence of other Pisolithus species in unexplored regions of Mexico is possible. For example, García-Rodríguez et al. (2006) reported collections of P. tinctorius from Eucalyptus plantations, but no barcode sequences were generated; ergo, their specimens are likely to correspond with another species, since P. tinctorius does not form associations with Eucalyptus. These reports support the need for further sampling across Mexico, primarily in underexplored arid and semiarid zones where gasteroid fungi are more diverse and abundant, as well as areas with vegetal species that match the known host of the genus.
Of the 32 collections reviewed, 21 were successfully sequenced; however, DNA degradation posed challenges due to specimen age, with collections ranging from 1 to 74 years old (average age: 45 years). Future collections should prioritize conditions that preserve DNA integrity for molecular analyses. This work underscores the need for continued research on Pisolithus to achieve comprehensive species diversity in Mexico.
Conclusions
We confirm the presence of two species of Pisolithus widely distributed in Mexico. Pisolithus tinctorius is mainly associated with Pinus-Quercus trees and Carya illinoinensis trees. Until now, its presence has been molecularly confirmed in the states of Chihuahua, Coahuila, Durango, Guerrero, Hidalgo, Jalisco, the State of Mexico, Oaxaca, Sonora, and Veracruz, while P. albus is associated with Eucalyptus in semiarid zones of northwestern Mexico, in Sonora and Baja California. Among the specimens studied from Mexico and soil metabarcoding sequences, we had no evidence of any that could be identified with P. arhizus.











 nueva página del texto (beta)
nueva página del texto (beta)