Introduction
Agriculture is a millenary activity that has been perfected over time to treat the soil and cultivate the land with the main objective of producing food and satisfying human needs. Due to the great demand for agricultural products worldwide, natural resources have been exploited, but poor cultivation practices and the massive use of chemical compounds have caused soil erosion and poor-quality products that affect the health of consumers, as well as a decrease in biodiversity, the appearance of resistant pests, imbalances in agroecosystems and harmful effects on the environment (Grageda-Cabrera, Díaz, Peña, and Vera, 2012). In view of this, research has been conducted focused on economically and socially acceptable agricultural production. To achieve this, the production and application of bio-inputs have been proposed (Mamani and Filippone, 2018).
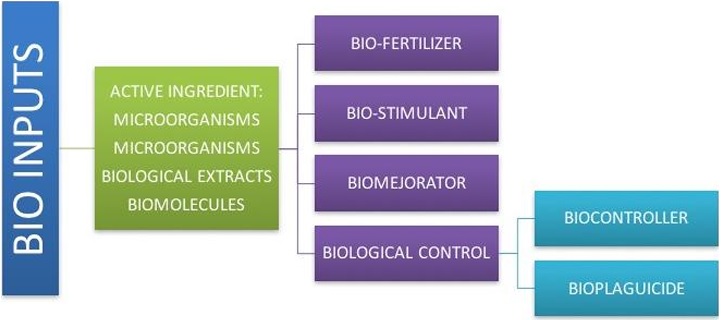
These are biological products formulated from compounds and extracts of microorganisms or plants, or live microorganisms, capable of improving productivity (or yield), quality, and health of soils and crops, resulting in environmentally friendly Figure 1 (Fillipone et al., 2017).
Biotechnology is used for their formulation since they use strategies that arise from the study and characterization of the different interactions of plants with their environment; they can be of plant or microbial origin. Depending on their effect on the plant, they are classified as biofertilizers (biostimulants, microbial inoculants, biostabilizers, organic fertilizers, humus, and guano) and biopesticides (microbiocides, bioinducers of plant defense against pests and diseases, and biorepellents) (Figure 2). In addition to bioinputs, biotechnology has contributed to the in vitro cultivation of plants in laboratories and greenhouses and the commercial exploitation of transgenic plants in different agroecological zones of the world (Christeson and Sims, 2011; Logan and Rabaey, 2012). Molecular engineering in the agricultural area has allowed obtaining plants resistant to insect pests and pathogens; improved products, such as fruits with prolonged storage periods with better nutritional properties (higher content of proteins, oils, amino acids) and industrial improvements (higher content of fruit solids).
The increase in population density has created a challenge for agriculture because it is necessary to supply the growing demand for food products; food security is related to climate change, the conditions of which have worsened, with alterations expected to become increasingly drastic. To address this problem, it is necessary to increase the implementation of sustainable agriculture and generate alternatives or products that allow mitigation of the pollution produced in agriculture using residues and the reduction of the use of chemical fertilizers and pesticides. In addition, it is necessary to generate new plants that are better adapted and more productive (Barajas, 2017; Chaves, 2021).
Therefore, the purpose of this review is to show the advances in the production of bioinputs, their characteristics and the results of their application, and thus expand our knowledge of these alternative biotechnological products for agriculture.
Development
Technological innovations with application in agriculture
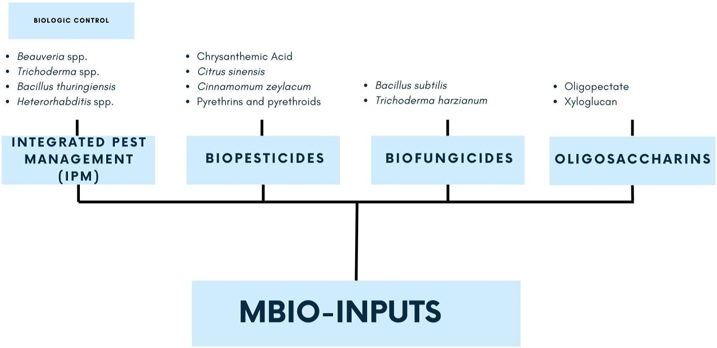
Currently, there are several products on the market derived from biotechnological research that present some activity or improvement in plant development and that are emerging as an alternative in agricultural activities. The use of microorganisms, such as fungi, bacteria, viruses; or their metabolites and insects, to eliminate or reduce the damage caused by harmful organisms that affect crops have been proposed as biological control agents in Integrated Pest Management (IPM), their purpose is to reduce chemical agents, having biofungicides and bioinsecticides as an ecological option (Viera-Arroyo et al., 2020).
Also, carbohydrates have been used, among which are oligosaccharins, Oligogalacturonides, and Xyloglucans, which induce a defensive response and resistance in plants, in addition to stimulating plant growth and development (Falcón-Rodríguez, Costales, González, and Nápoles, 2015; Lara, Costales, and Falcón, 2018).
On the other hand, the use of chitosan stimulates plant root growth, acts as an antimicrobial agent to combat pathogens in the soil, presents an activity to control the deterioration of post-harvest products during storage and the export process; as well as in the elaboration of fertilizer coatings for controlled release (Katiyar, Hemantaranjan, Singh, and Bhanu, 2014; Bauer, Villegas, and Zucchetti, 2022).
Nowadays, co-products, by-products, and solid and liquid organic residues (raw material) from the fishing, agricultural, and industrial sectors (food, sugar, tequila) contain bioactive compounds and minerals of agricultural interest. These residues are used in bioprocesses that employ methods such as aerobic biodegradation, fermentation, hydrolysis, and extraction (macroalgae). The products obtained can be considered as organic fertilizer and biostimulant (depending on their composition of macronutrients, micronutrients, amino acids, microorganisms, and bioactive compounds), whose effect ranges from the improvement of growth and development in plants to the control of biotic and abiotic stress in crops so that they can grow without difficulties (Florez-Jalixto, Roldán, Omote-Sibina, and Molleda, 2021). Each of these bioproducts is detailed below.
Use of biological control in integrated pest management (IPM)
Biological control uses living organisms or their metabolites to eliminate or reduce damage caused by harmful organisms. Biological control agents (BCA), such as fungi, bacteria, viruses, and insects, are being developed worldwide to reduce the population of pests affecting crops (Table 1). The use of entomopathogenic fungi has gained importance in recent years because they do not cause damage to the environment and health (Viera-Arroyo et al., 2020).
Table 1: Main organisms used in biological control with application to crops of agricultural importance.
| Microorganism | Application, action, or effect in agriculture | Reference |
| Beauveria spp. | Management of numerous veterinary and forestry agricultural arthropod pests. It is implemented in one or more flooding applications of large quantities of aerial conidia in dry or liquid formulations. Mass production is mainly practiced by solid-state fermentation to produce hydrophobic aerial conidia, which are the main active ingredient of mycoinsecticides. Infection of the fungus begins with the adhesion of the conidia to the cuticle of the host; when these germinate, they attach to the cuticle, forming a hyphae that allows it to penetrate the insect with the help of hydrolytic enzymes (proteases, lipases, chitinases), mechanical pressure and other factors. The hyphal bodies produce blasto-spores that disperse throughout the host body, destroying tissues, evading the immune system, and producing toxins that contribute to death. It is used in a variety of crops for the control of whiteflies (Bemisia tabaci), mites (Tetranychus urticae), beetles (Gonipterus scutellatus), coffee berry borer (Hypothenemus hampei), banana borer (Cosmopolites sordidus), defoliating worm (Dione juno), thrips, aphids, bugs, leafhoppers. | (Wraight and Ramos, 2002; Humber, 2008; Lacey, Wraight, and Kirk, 2008; Malpartida-Zevallos, Narrea, and Dale, 2013; Ortiz-Urquiza and Keyhani 2013; Jaramillo, Montoya, Benavides, and Góngora, 2015; Mascarin and Jaronski, 2016; Espinel et al., 2018) |
| B. bassiana | ||
| B. brongniartii. | ||
| Trichoderma spp. | The species of this fungus act as biofungicide, biofertilizer, growth promoter, and inducer of natural resistance; its application can be foliar before planting (in seeds or propagation material), incorporated into the soil during planting or transplanting (by means of irrigation). It is the most widely used biological fungicide in agriculture. The fungi live freely in the soil and root ecosystems, possessing antagonistic properties and therefore exerting biocontrol of phytopathogenic fungi such as Fusarium, Rhizoctonia, Pythium, Sclerotium, and Phytophthora, which affect crops of commercial interest such as rice, corn, onion, tomato, bean, wheat, cocoa, and quinoa.Indirectly, it competes for space and nutrients, producing antibiotics or stimulating plant growth and defense mechanisms; directly, it can control mycoparasitosis through the production of different metabolites. | (Woo et al., 2014; Charoenrak and Chamswarng, 2016; Tirado-Gallego, Lopera, and Ríos 2016; Leon-Ttacca, Ortiz, Condori, and Chura, 2018) |
| T. harzianum. | ||
| T. asperellum | ||
| T. viride. | ||
| T. atroviride. | ||
| T. gamsii. | ||
| T. hamatum. | ||
| T. koningii. | ||
| T. lignorum. | ||
| T. polysporum | ||
| Bacillus thuringiensis | Entomopathogenic bacteria must be ingested and act by releasing toxins or penetrating midgut cells before invasion of the hemocele to multiply in the insect carcass. After causing death, the new progeny of the bacteria leave to infect new hosts. Gram-positive bacteria such as Bacillus thuringiensis (Bt) and insect pathogens of the genera Paenibacillus and Lysinibacillus have the ability to form spores. In contrast, Gram-negative bacteria do not form spores and include isolates of the genera Serratia, Yersinia, Photorhabdus, Chromobacterium. Several entomopathogenic bacteria can be produced by fermentation. B. thuringiensis active strains act on Lepidoptera, Coleoptera and Diptera. Due to its greater stability in the production, formulation, and field application processes, using Cry spores and protein crystals as the active ingredient, it is the species most widely used on a large scale for the control of insect pests. Species such as B. subtilis, B. pumilus, B. licheniformis, and B. amyloliquefaciens have been successfully implemented in commercial formulations developed mainly for the control of fungal diseases in grasses, oilseeds, fruit trees, vegetables, and ornamentals. | (Grijalba, Hurst, Ibarra, Jurat, and Jackson, 2018; Villareal-Delgado et al., 2018) |
| Heterorhabditis spp., | Entomopathogenic nematodes are soil-dwelling organisms that interact as important regulators of their ecosystem and are used as controllers of lepidopteran pests and other types of insect pests in commercially important crops. Their use is based on their ability to adapt to new environments and adverse conditions, resistance to chemicals, high specificity for insects, harmlessness to the environment and mammals, and compatibility with other entomopathogens. The species of the genus Heterorhabditis present a cephalic structure like a spike in the juvenile infectives, which gives them an additional way of entering the insect; this characteristic makes them more infective with respect to other species, since in addition to penetrating through the natural openings (anus, mouth, spiracles), they can also do so by perforating the cuticle. | (Castillo, Gallegos, Asaquibay, and Oña 2011; Gianfelici, Bertolotti, and Cagnolo, 2014; López-Llano and Soto-Giraldo, 2016; Rumbos and Athanassiou, 2017; Sánchez, Valle, Pérez, Neira, and Calderón, 2019) |
| Steinernema spp. | ||
| Plant extracts Jatropha curcas, Azadirachta indica, Melia azedarach, Citrus aurantifolia, Eucalyptus globulus, Piper nigrum, Punica granatum, Salix safsaf | They are derived from some parts or active ingredients of plants; among these are attractants, hormones, plant and insect growth regulators, enzymes, and chemical signaling substances of importance in the plant-insect relationship. These pesticides contain phytochemicals or secondary metabolites such as terpenes, phenols (tannins and coumarins), alkaloids, cyanogenic glycosides, sulfur compounds (thiophenes) and flavonoids (rotenone); these substances cause repellency, inappetence, prevent oviposition, act as a barrier by their taste, inhibit growth, are toxic to nematodes, mites and insects, release cyanide, act as enzyme inhibitors. | (Jannet, H-skhiri, Mighri, Simmonds, and Blaney, 2001; Martínez, 2012; Nava-Pérez, García, Camacho, and Vázquez, 2012) |
| Aqueous seaweed extract Padina gymnospora (Kützing) Sonder, Sargassum latifolium (Turner) C. Agardh Hydroclathrus clathratus (C. Agardh) M. Howe Sargassum vulgare | In vivo application of seaweed powder as a soil amendment decreases the percentage of root rot disease caused by Fusarium solani on Solanum melongena L. (eggplant). The antifungal activity of extracts of the algae Laminaria digitata, Undaria pinnatifida (Harvey) Suringar, and Porphyra umbilicalis Kützing against the postharvest pathogens Botrytis cinerea, Monilinia laxa,and Penicillium digitatum in strawberries, peaches and lemons is highlighted by inhibiting mycelial growth and conidial germination. The aqueous and methanolic extract of Sargassum vulgare has shown antifungal potential by inhibiting the mycelial growth of Pythium aphanidermatum, reducing the disease observed in potato tubers cv. Spunta. | De Corato, Salimbeni, De Pretis, Avella, and Patruno, 2017; Ibraheem, Hamed, Abd Elrhman, Farag, and Abdel, 2017; Ammar et al., 2017 |
As a result of the application of biotechnology in agriculture, several products have been developed and placed on the market. The formulations are contemplated in an Integrated Pest Management (IPM) plan, such as biopesticides (insects, mites, nematodes) and biofungicides, or they are incorporated into crops as biofertilizers or biostimulants to promote plant development through efficient assimilation of nutrients.
Biopesticides (Table 2) are products that contain microbial control agents (entomopathogens), entomophagous organisms, organic compounds as the active ingredient, or metabolites of the microorganism that are extracted by procedures that do not alter their composition. These products have demonstrated their toxic activity for pest control, cause minimal environmental damage, and do not leave toxic residues in food, so they are not considered a risk factor in humans (Biokrone, 2023).
Table 2: Biopesticides marketed for pest control.
| Biotechnology product | Active ingredient/biological | Action | Manufacturer or distributor | Reference |
| Cridor® | Chrysanthemic acid and pyritic acid | Chrysanthemum cineraiaefolium extract is effective for the control of pests such as Frankliniella occidentalis, Lygus sp. Pentatrichopus fragaefolii, Scirtotrhips perseae, Idona minuenda, Brevicoryne brassicae, Plutella xylostella, Bemisia tabaci, Bactericera cockerelli in vegetables, strawberries and avocado. | Syngenta Agro, S.A. de C.V. | (Syngenta, 2023) |
| Candor® | Citrus sinensis Cinnamomum zeylacum | Botanical acaricide and insecticide based on citrus and cinnamon extracts that acts on Tetranychus urticae, Bemisia tabaci, Brevicoryne brassicae; used for IPM on vegetables, cucurbits, and strawberries. | Syngenta Agro, S.A. de C.V. | (Syngenta, 2023) |
| Pirecris® | Pyrethrins and pyrethroids | An incapacitating effect known as knock down that acts by contact and ingestion, producing an immediate paralysis of the insect and the death of the pest (decreases the population). It is indicated for the control of pests such as aphids and whiteflies; in avocado, strawberries, vegetables, cucurbits, broccoli, cabbage, cauliflower, rape, kiwi, and grapes. | SEIPASA MEXICO, S.A. DE C.V. | (Seipasa, 2023) |
| Cinnerate™ | Cinnamon extract oil | It acts by contact, penetrating and destroying the soft parts of the body. Insects (avocado thrips) and mites are exposed to loss of body fluids, causing dehydration and death. It is applied on vegetables, cucurbits, strawberries, citrus and avocado. | SEIPASA MEXICO, S.A. DE C.V. | (Seipasa, 2023) |
| BIO BT® | Bacillus thuringiensis var. kurstaki and Bacillus thuringiensis var. aizawai | During the sporulation, it produces delta endotoxins (δ-endotoxins), components of the protein crystals formed, which once activated in the intestine of the insect by enzymatic effect under conditions of alkaline pH of the stomach of the larvae. These cause osmotic imbalances that break the intestinal wall of Helicoverpa zea, causing death due to lack of food or septicemia. | AGRHUSA Agrobiológicos | (AGRHUSA, 2023) |
| AGRONEEM® | Neem seed extract oil (azadirachtin) | The components of neem extract, especially azadirachtin, enter the body of the insect (Bemisia tabaci) either by contact or ingestion, suppressing the activity of ecdysone, preventing the insect from molting or causing malformations that lead to death or sterility. In addition, it has a phagodisuasive effect, discourages oviposition of females, inhibits chitin formation and acts as a repellent of larvae and adult insect pests. | AGRHUSA Agrobiológicos | (AGRHUSA, 2023) |
| Benemite® | Geraniol + Citronellol + Nerolidol + Farnesol | Broad spectrum acaricide contains pheromones that alter sexual and feeding behavior, interferes in the molting process of immature stages such as nymphs and protonymphs, blocks the spiracles causing asphyxia of the mite and decomposition of the chitin (rupture of the exoskeleton of the mite). It can be used on strawberries, vegetables, cucurbits, corn, avocado, and rose. | Arysta LifeScience México, S.A. de C.V. | Arysta LifeScience, 2023 |
| EPA 90® | Soybean vegetable oil | The insecticidal action is based on its property of forming a layer that completely covers nymphs and adults, blocking the spiracles (gas exchange), which causes suffocation or asphyxiation of the insects. As a highly lipophilic product, it alters the chorion, cell membrane, and integument of eggs, nymphs, and adults, causing their death by desiccation. Recommended for Trips tabaci, Bemisia tabaci, Diaphorina citri, Tetranychus urticae, Paraleyrodes perseae, Frankliniella occidentalis; in chives, citrus, strawberries, avocado, and vegetables. | Biokrone, S.A. de C.V. | Biokrone, 2023 |
| NEMOVER | Extracts of fig (Ricinus communis), oregano (Origanum vulgare) and pine (Pinus pinaster) | It acts by contact with a broad spectrum of control and immediate effect on the populations of soil pests and nematodes that attack different vegetable crops. By adhering to the substrate and roots, it impregnates the particles with its active ingredients, inhibiting the presence of eggs on the surface where the product had contact, contains the development of the nematode (Meloidogyne spp.) in the second larval stage and causes intoxication by feeding on the epidermal tissues. Controlling nematodes and soil pests allows healthy and abundant roots to develop. | ECOPROTECTO, S.A. DE C.V. | Agrorganicos nacionales, 2023 |
A biofungicide is a compound whose active material is an antagonistic microorganism, a beneficial microorganism such as fungi or bacteria that acts on pathogenic plant diseases. Biofungicides (Table 3) are an alternative to chemical fungicides (Biokrone, 2023).
Table 3: Biofungicides marketed for the control of phytopathogens.
| Biotechnology product | Active ingredient/biological | Action | Manufacturer or distributor | Reference |
| Cinnerate™ | Cinnamon extract oil | It exerts a curative action, cinnamic aldehyde inhibits mycelial growth and prevents germination of fruiting bodies. It affects mitochondrial oxidative phosphorylation and induces profound changes in the enzymatic activity of the cell membrane of the pathogen (powdery mildew, downy mildew), causing dehydration and the disappearance of foliar symptoms. Crops: Avocado, vegetables, strawberries, citrus, cucurbits, tobacco, solanaceae, kiwifruit, grapes. | SEIPASA MÉXICO, S.A. DE C.V. | Seipasa, 2023 |
| Fungisei® | Bacillus subtilis strain IAB/BS03 | It acts by contact; the selected strain of Bacillus subtilis could produce lipopeptide antibiotics (iturins, surfactins, and phengicins), which prevent pathogenic spores from germinating, altering the growth of the germ tube of the spores and thus inhibiting the attachment of the pathogen (powdery mildew, gray mold) to the leaf; in bell pepper, grape, and blueberry. | SEIPASA MÉXICO, S.A. DE C.V. | Seipasa, 2023 |
| Seipa System | Acacia Extract | Strengthens the plant's defensive system: Together with proteins, it generates self-defense molecules that inhibit the hydrolytic enzymes of phytopathogenic fungi from penetrating plant cells. Mechanical resistance is produced in the plant due to the association of silicon with cell wall components. It controls Fusarium and Botrytis in vegetables, grapes, and strawberries. | SEIPASA MÉXICO, S.A. DE C.V. | Seipasa, 2023 |
| Baktillis® | Bacillus subtilis spores and metabolites | It acts as a physical barrier on the plant surface preventing adherence and germination of phytopathogens. The lipopeptides it contains perforate the mycelial membranes and fungal spores, preventing their development. In addition, the metabolites subtilin, bacitracin, and toximicin act directly on the spores and mycelium of phytopathogens. Recommended for the control of Fusarium, Botrytis, Colletotrichum, among others; present in strawberries, citrus, and vegetables | Biokrone, S.A. de C.V. | Biokrone, 2023 |
| NatuControl® | Spores of Trichoderma harzianum | Directly attacks and parasitizes the structures of phytopathogenic fungi, including those inside the plant. Induces resistance that activates plant defense mechanisms. Produces substances and metabolites that act directly on spores or mycelium preventing the germination and development of phytopathogenic fungi. Rapidly colonizes the rhizosphere of the plant, preventing the germination, growth, and development of phytopathogenic fungi such as Phytophthora, Rhizoctonia, and Fusarium, among others. Recommended for: strawberries, vegetables, citrus, cucurbits. | Biokrone, S.A. de C.V. | Biokrone, 2023 |
| Iodus 40® (Vacciplant) | Laminarin based product (extracted from seaweed) | Activates the intrinsic protection of the plant (strengthens the cell wall, phytoalexin production, and production of PR protein). Recommended for use in vegetables, cucurbits, and strawberries, among others, to control Botrytis, Alternaria, Xanthomonas, ash, mildew, and anthracnose. | Laboratorios GOEMAR SAS (Francia). | Arysta Life Science, 2023 |
Use of Oligosaccharins
Oligosaccharins (Table 4) are natural polysaccharides and oligosaccharides that are part of the cell walls of plants and microorganisms such as fungi; the main sources of raw material for their large-scale preparation are agricultural by-products and the exoskeleton of crustaceans discarded from the fishing industry (Falcón-Rodríguez et al., 2015). They can be endogenous or exogenous.
Table 4 Products or research that has been carried out based on oligosaccharins with applications in the agricultural sector.
| Product | Application, action or effect in agriculture | Reference |
| Oligogalacturonides are located in the pectic portion of the cell wall of plants Oligopectate (pectin-derived oligosaccharide) | Increased color in the fruit of Vitis vinifera var. Flame Seedless (grape), related to increased anthocyanin content and possible induction of PAL (Phenylalanine ammonia lyase) mRNA expression. | (Ochoa‐Villarreal, Vargas, Islas, González, and Martínez, 2011) |
| Xyloglucans: hemicelluloses that make up the primary cell wall structure of dicotyledonous and non-poaceous monocotyledonous plants. In dicotyledon seeds, they are part of the reserve polysaccharides | Induction of defensive response and resistance in plants, influencing plant growth and development. Tamarind seeds are rich in xyloglucans. | (Falcón-Rodríguez, Costales, González, and Nápoles, 2015; Lara, Costales, and Falcón, 2018) |
| Pectimorph®. Composed of biologically active carbohydrates. 55-61 % galacturonic acid, which constitutes vegetable pectin, is present mainly in fruits. | Replaces growth regulators in vitro culture, promotes rooting in cuttings, stimulates crop growth and yield, and accelerates seed germination. Crops: Coffee, sugar cane, garlic, potato, cassava, tomato, banana, radish, carrot, lettuce, chard, spinach, soybean, bean, guava, citrus, grape, peach, fig, cinnamon, laurel, rose, orchid, lily, anthurium, bougainvillea, ficus, areca, thuja. | (Falcón-Rodríguez et al., 2015; Lara et al., 2018; Ramos et al., 2013; INCA, 2023) |
| EcoMic®. Formulation based on Arbuscular Mycorrhizal Fungi, Glomus cubense strain | It increases the absorption capacity of soil nutrients and fertilizers, decreases the damage of some root and foliar pathogens, contributes to improving soil structure, and increases carbon sequestration in agroecosystems. Crops: Beans, corn, bananas, rice, soybeans, sorghum, vegetables, cotton, sunflower, citrus and fruit trees, grass species, flowers, and trees. | (Rivera et al., 2020; INCA, 2023) |
| Quitomax®. Biostimulant based on natural biodegradable polymer, chitosan | It allows a reduction in the application of chemical pesticides on crops and increases from 10 to 30% the yield of plants; its combined application benefits the process of nitrogen fixation and growth in leguminous plants. Crops: corn, sorghum, potato, tomato, bell pepper, garlic, onion, soybean, bean and rice. | (Morales-Guevara, Dell Amico, Jerez, Hernández, and Martín, 2016; Rodríguez-Pedroso et al., 2017; Gustavo-González et al., 2021 ; INCA, 2023) |
| AzoFert®. Biofertilizer based on rhizobia (Rhizobium) and nodulation inducers | Direct nitrogen supply to the plant, promotes plant growth and crop yield, increases the utilization of soil nutrients, as well as the protection of the root system against certain diseases. It has been used in bean and soybean crops. | (Estrada et al., 2017; Hernández and Salido 2019; Calero, Quintero, Pérez, Jiménez, and Castro 2020; INCA, 2023) |
| Coating of seeds with a 2% solution of chitosan | Positive effect on the microbiological development of the Zea mays (maize) plant, acting as a seed protector against pathogenic fungi under abiotic stress conditions (drought, humidity, acid pH, and alkaline pH) | (Lizárraga-Paulín, Torres, Moreno, and Miranda, 2011) |
| Crosslinking of chitosan in hydroxypropyl methylcellulose for the preparation of Neem oil coating for postharvest storage of pitaya (Stenocereus pruinosus) | Application of chitosan-based antimicrobial coatings for postharvest preservation of pitaya (storage at 10 ± 2 °C, relative humidity of 80 ± 5%). This reduced weight loss and fungal contamination, increasing shelf life (up to 15 days). | (Hernández-Valencia, Román, Aguilar, Cira, and Shirai, 2019a) |
Chitosan is also considered as oligosaccharin, it is a natural polymer derived from chitin (the main component of the shell of crustaceans, it is also found in the cell wall of fungi and the exoskeleton of some insects, it is the second most abundant natural biopolymer); It acts as a plant root growth enhancer, an antimicrobial agent to combat pathogens in the soil, a gel that controls the deterioration of post-harvest products during storage and during the export process, as well as in the elaboration of fertilizer coatings for controlled release (Katiyar et al., 2014; Bauer et al., 2022).
Development of biofertilizers and stimulants
The indiscriminate use of chemical fertilizers is one of the main problems in agriculture, leading to the loss of soil fertility and contamination of water bodies. As an alternative, biofertilizers are a biotechnological tool that consists of applying microorganisms that help to improve the availability of nutrients in the soil and thus reduce the effects caused by the excess of fertilizers. Their use in different crops has evidenced positive effects on soil fertility; they are applied to seeds, plant surfaces, or soil to colonize the rhizosphere or the interior of the plant and promote growth by increasing the supply or availability of primary nutrients to the host plant.
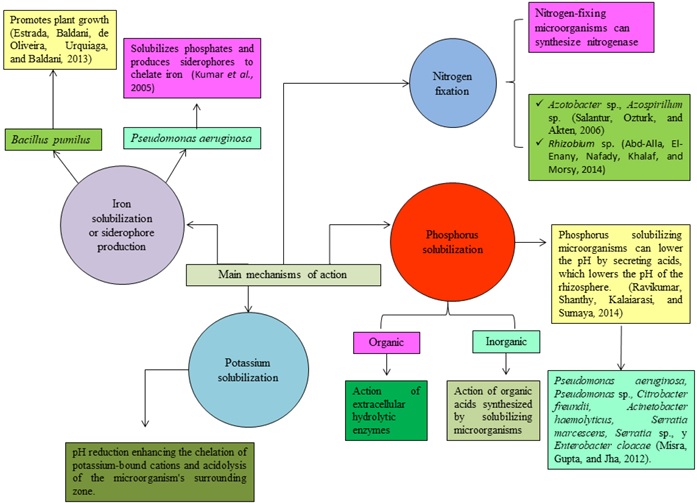
Plant growth-promoting rhizobacteria (rhizosphere-dwelling bacteria) are phytostimulants containing bacterial auxins capable of inducing root elongation. Several genera such as Acetobacter, Acidithiobacillus, Aminobacter, Arthrobacter, Azoarcus, Azospirillum, Azotobacter, Azolla, Bacillus, Burkholderia, Clostridium Enterobacter, Gluconoacetobacter, Pseudomonas, Serratia and Sphingomonas; have demonstrated their growth promoting capacity, so they are among the main genera used for the formulation of commercial biofertilizers, which are applied in cereal and vegetable crops. Their main function is to mobilize nutrient availability based on their biological activity, help recover lost microbiota and generally improve soil health (Malusá, Sas-Paszt, and Ciesielska, 2012; Ismail, Mohamed, Khattab, and Sherif, 2014; Velasco-Jiménez, Castellanos, Acevedo, Aarland, and Rodríguez, 2020).
The impact of chitosan, mycorrhizal fungi, and humic acids on the growth of bell pepper (Capsicum annuum L.) varieties under protected conditions has been reported, finding that the three biostimulants increased seed germination between 11.66 and 16.67%, while emergence was enhanced by humus and chitosan. Humic acids produced taller plants and larger diameter stems. When humic acids were applied, higher yields were achieved due to increased length, diameter, weight, and fruits per plant (Reyes-Pérez et al., 2021). On the other hand, the agronomic response of Vigna unguiculata L. Walp (beans) was greater when seeds were inoculated with a strain of the arbuscular mycorrhizal fungus Rhizophagus irregularis (Tamayo, Riera, Terry, Juárez, and Rodríguez, 2019).
A similar case has been observed in evaluating the effect of different nutritional management on yield and internal and external quality of tomato fruits. The combination of 50% mineral fertilizer - bioproducts (partial organic nutrition treatment), allowed obtaining an adequate tomato yield without affecting the internal and external quality of the harvested fruits. Therefore, the bioproducts contributed to partially replacing the mineral fertilization required by the tomato crop (Terry-Alfonso, Ruiz, and Carrillo, 2018).
The use of mycorrhizae has been related to crops that have a seedling stage, as occurs in horticulture; these fungi are important in nutrient uptake, protection under water stress conditions, and prevention against pathogens. The mycorrhizal association is relevant in soil stability and in reducing the negative effects of microbial competition, in the establishment, survival, and growth of plants in the field, as well as in stimulating crop yields (Plana-Llerena, González, and Soto, 2016).
The use of algal biostimulants in plant crops can generate numerous benefits with reported effects, including improved rooting, higher crop and fruit yields, increased photosynthetic activity, and resistance to fungi, bacteria, and viruses. With Padina gymnospora, the growth of eggplant (Solanum melongena L.) in Fusarium solani-infected soils is improved (Sharma, Fleming, Selby, Rao, and Martin, 2014; Ibraheem, Hamed, Abd Elrhman, Farag, and Abdel, 2017).
Biofortifiers and biostimulants (Table 5) are products based on microorganisms, organic matter, plant extracts, and oils, among others, whose application promotes and improves plant efficiency in the absorption and assimilation of nutrients and contributes to tolerance to biotic or abiotic stresses.
Table 5: Biofertilizers or biostimulants marketed to promote nutrient uptake and assimilation in plants.
| Biotechnology product | Active ingredient/biological | Action | Manufacturer or distributor | Reference |
| Isabion® | Organic matter (mixture of amino acids and peptides) | Fertilizers of natural origin are classified as an organic nutrient that supplies plants with amino acids and peptides to stimulate their vegetative development. Recommended for: vegetables, ornamentals, cucurbits, alliaceae and brassicas | Syngenta Agro, S.A. de C.V. | Syngenta, 2023 |
| ALGACROP® | Seaweed 80%, Mixture of: Ascophyllum nodosum and k sp. | High content of amino acids, phytohormones, carbohydrates, and other organic compounds that promote plant growth and development; prevent abortion in the flowering stage; improve fruit quality and firmness; favor root development; and cover nutritional deficiencies by increasing the availability and absorption of elements. Crops: vegetables and tobacco. | AGRHUSA Agrobiológicos | (AGRHUSA, 2023) |
| ULTRAHUMI® | Humic acids, fulvic acid | Humic acids are the reverse of nutrients necessary for the plant, as these require organic matter as a carbon source. It helps the plant to avoid stress, stimulates bacterial growth in the rhizosphere, acts as a soil improver by increasing moisture retention, stimulates respiration, metabolism, and cation exchange capacity at the cellular level, reduces the harmful effects of toxic agents, favors the recovery of insoluble phosphorus and increases soil microbial life. Crops: vegetables and tobacco. | AGRHUSA Agrobiológicos | (AGRHUSA, 2023) |
| E-Microzyme® | Primary microorganisms: Azotobacter vinelandii, Clostridium pasteurianum | Microbial enzyme complex contains a variety of soil microorganisms that help increase soil fertility and provide vital nutrients to all types of plants and crops. | MYDAGRO LLC. | (PLM® DEAQ/DIPO, 2023) |
| AmiKrone® | L-amino acids, peptides, and polypeptides of natural origin Aspartic Acid, Glutamic Acid, Alanine, Amino Acids, Glycine, Lysine, Manganese, Organic Matter, Methionine, Proline, Zinc. | It is rapidly absorbed by plants either via foliar or root (depending on the form of application), acts as a biostimulant by triggering and enhancing numerous reactions of plant metabolism, provides vitamins and gives resistance in times of stress, also controls the water content inside, which gives it a prolonged life. It can be applied to vegetables, aliaceae, cucurbits, brassicas, legumes, grasses, fruit trees, ornamentals, and strawberries. | BIOKRONE, S.A. DE C.V. | Biokrone, 2023 |
| AZSeed® | Azospirillum brasilense, Azotobacter spp., Rhizobium spp., Bacillus spp., Natural rooting agents (AIA) Inoculant and root bioactivator. | When applied to the seed and deposited on the soil or substrate, the bacteria contained in the product are activated and begin to reproduce rapidly, generating a synergism with the plant roots. The beneficial bacteria fix atmospheric nitrogen and give it to the plant. The metabolites (AIA) it contains ensure uniform seed germination and rapid rooting of the plants, thus generating vigorous plants. It can be used in grasses, vegetables, brassicas, alliaceae, fruit trees, citrus, legumes, oilseeds, strawberries, sugar cane, ornamentals, and agave. | BIOKRONE, S.A. DE C.V. | Biokrone, 2023 |
| GLUMIX® | Spores of Glomus geosporum, G. fasciculatum, G. constrictum, G. tortuosum, G. intraradices, selected strains of vesicular arbuscular mycorrhizal fungi (VAM) | It invades the roots when they emit exudates or chemical compounds that stimulate the germination of the spores; inside the root, a series of dichotomous ramifications begin that give rise to an arbuscule, which occupies the cortical cells of the host. The hyphae and mycelium of the mycorrhizae penetrate the root of the plant, coming into contact with the ribosomes (root cells). Externally to the roots, the mycorrhizae emit large quantities of mycelium or hyphae that extend exploring a considerable volume of soil; in this way, the mycorrhizae contribute to the better development and growth of the plant. It is recommended for use in grasses, vegetables, brassicas, aliaceae, fruit trees, citrus, legumes, oilseeds, strawberries, sugar cane, ornamentals, and agave. | BIOKRONE, S.A. DE C.V. | Biokrone, 2023 |
Main Mechanisms of Action of microorganisms used
In general terms, the mechanisms of action employed by some of the microorganisms used to produce biofertilizers are summarized in Figure 3.
Genetic improvement of crops of agricultural interest
Another way in which biotechnology has contributed to the agricultural sector is the development of in vitro tissue culture methodologies, which allow the massive propagation of individuals of interest, making it possible to work with multiple individuals in a reduced space and controlled conditions, as well as the possibility of adding selection agents to the culture medium. Thus, it is also possible to obtain plants resistant to drought, herbicides, and diseases. Other important biotechnologies are medium and long-term conservation by cryopreservation (storage of plant tissues such as seeds, apices, buds, and cells under ultra-low temperature conditions, using liquid nitrogen [-196 °C]) (Chaves, 2021).
Micropropagation or in vitro propagation is one of the applications of plant tissue culture that contributes to accelerating the selection processes within genetic improvement programs. It has been applied for the genetic improvement of various crops. For example, sugarcane has been decisive in developing varieties resistant to biotic and abiotic factors and in increasing biomass and sucrose yields. In the in vitro regeneration of sugarcane plants, the cultivation of protoplasts, cells, callus, tissues, and various organs has been used (Castañeda-Castro et al., 2014).
Soil salinity affects food production on a global scale by causing physiological and biochemical changes in plant metabolism that determine their survival and productivity under these conditions, for which plants have developed tolerance mechanisms. Genetic improvement offers an increase in the recovery of underutilized areas and in yields in areas where salinity is a limiting factor. The selection of salinity tolerant plants is a long process, so efficient indicators are sought for the early selection of genotypes with better agronomic performance (Lamz and Gonzalez, 2013).
Modern genetic improvement has two stages: the first is to obtain genetic variability and the second consists of selecting the genotypes thus obtained to develop varieties adapted to specific conditions. Crossing between different genotypes of the same species is the conventional tool for generating variability through classical breeding.
Biotechnology makes it possible to make use of all existing genetic variability, circumvent the sexual barrier to incorporate genes from other species, integrate artificial genes, or edit the genome (making it possible to generate genetic variability). Another advantage is the possibility of introducing a single new gene in the genetically improved plant, preserving the rest of the genes of the original plant in its offspring. This technology has been used in crops to increase insect resistance and herbicide tolerance; organisms to which genetic material has been introduced in this way are called “genetically modified organisms” (GMOs).
In this sense, Bacillus thuringiensis (Bt) is a Gram-positive bacterium in Bacillus form, which produces Cry proteins with biological activity against insect pests. Due to these proteins, B. thuringiensis presents toxicity against insect-pest larvae of the orders Lepidoptera, Coleoptera, and Diptera, among others. It is common in the development of commercial products and transgenic plants based on Cry toxins in the agricultural sector, such as cotton and corn. The transformation of plants, through the incorporation of Cry protein-coding genes, confers protection to internal tissues, maintains a constant production of protein, and does not generate a harmful environmental impact (De Maagd, Bravo, and Crickmore, 2001). Transgenic foods are those generated from genetic engineering and genome editing techniques. Golden Rice is a variety with nutritional improvement due to the increase in vitamin intake in which two genes are inserted through genetic engineering techniques, resulting in an increase in the amount of β-carotene in the grains. It is contemplated that this variety will serve to attenuate blindness in juvenile populations while it will reduce child malnutrition and malnutrition.
Prospects for achieving sustainable agriculture
Bioprospecting is the research conducted to identify species, varieties, genes, and valuable natural components of organisms in order to develop products for commercial purposes with current or potential use for their application in agriculture, industry, and the environment. Considering the diversity of microscopic organisms that inhabit the planet, as well as the multiple bioactive substances present in higher organisms; it is important to give continuity to bioprospecting, as this will allow the development of bioproducts, culture media or identify suitable substrates for the growth of beneficial micro and macro organisms for agriculture, such as Trichoderma (Páramo-Aguilera, Fonseca, Delgado, Cabistán, and Ríos, 2018), which is important for its ability to preserve and restore soil microbiota, for its potential for the synthesis and release of enzymes such as cellulases, xylanase, and chitinases, for the production of auxins and crop growth regulators, as well as a biological agent that can significantly reduce the incidence of diseases in crops under greenhouse conditions (Andrade-Hoyos et al., 2023).
In this sense, modern agriculture is facing new challenges, integrating ecological and molecular approaches to achieve higher crop yields and minimize the impact on the environment. In order to generate higher yields, chemical pesticides are used, and the dose of synthetic fertilizers per unit area has increased significantly, inputs that can cause pollution, damage to health, and loss of soil fertility, becoming one of the most important concerns in agricultural production. To improve production without the use of fertilizers of synthetic origin and pesticides, research has been oriented towards the development of new biotechnologies: causing there to be a growing interest in beneficial soil microorganisms as these can promote plant growth and, in some cases, prevent infections of plant tissue by pathogens.
These challenges are determined by the demographic increase and the demand for raw materials for human and animal food, by preferences in consumption, by environmental and legal aspects, and by the globalization of the economy (Melgarejo, Romagosa, and Durán, 2014; Moreno, Carda, Reyes, Vásquez, and Cano, 2018).
Endophytic fungi are microorganisms that inhabit plant tissue without causing apparent symptoms of damage and are capable of producing bioactive metabolites that confer benefits to the host. With the purpose of obtaining secondary metabolites, it has been possible to isolate endophytic fungi from plants and soils, which have shown antimicrobial activities, as well as stimulating plant development. However, further research is still needed to provide information on the toxicity and safety of secondary metabolites so that they can be used as agrochemical agents. In addition, it is essential to conduct studies on the role that these metabolites may have in nature and the protection they provide to their host against pathogens and herbivores (Sánchez-Fernández et al., 2013).
The use of basidiomycetes fungi in agriculture has aroused interest due to their ability to efficiently degrade aromatic and heterogeneous compounds because they present a nonspecific enzyme complex with oxidative activity against a wide variety of toxic and recalcitrant substances, and some species have shown antifungal, phytotoxic and nematicidal activity. In addition, basidiomycetes have the ability to efficiently degrade lignin (a process that involves the synergistic action of extracellular enzymes). They can also transform simpler substances into environmental pollutants. For these reasons, they are promising species as biological controllers with an interest in agriculture; they can influence the control of plant diseases and thus become a potential biotechnological tool. The bioactivity of basidiomycetes has not been sufficiently investigated and constitutes a source for bioassays, suggesting new lines of development of different metabolites with a wide range of applications (Rojas, 2013).
On the other hand, macroalgae are a source of macro and microelements, bioactive compounds, growth regulators, and organic matter; they have nutritional value, stimulate plant defense against pathogens, improve soil characteristics, and increase agricultural production in a sustainable manner. Seaweed hulls are a resource with numerous possibilities for their use as raw material to produce flour, extracts, compost, and biotechnological products, with the aim of reducing the use of chemicals in soils. Therefore, it is transcendental to continue trials with algae species that have not been studied in order to know the compounds they contain and the mechanism of action in plants, in soils, and the activity on pathogens of agronomic importance, both in field and greenhouse conditions (Durán-Hernández, Uribe, Mateo, and González, 2022).
The production of organic fertilizers and biostimulants for the development of commercial formulas must first be developed at the laboratory scale, optimizing the working parameters to be scaled up, in addition to demonstrating the absence of phytotoxic substances and pathogenic microorganisms. In this sense, it is important to continue promoting sustainable strategies for more efficient and cost-effective waste disposal for the benefit of organic agriculture, aquaculture, and the environment, in addition to framing it in a circular economy model (Florez-Jalixto et al., 2021).
On the other hand, research for breeding transgenic plants will continue to grow, and the long-term outlook is that transgenics will continue to be introduced into world agriculture, occupying larger areas and partially or totally replacing traditional varieties (Ardisana, Álvarez, Macías, Gaínza, and 2016).
Conclusion
Biotechnology techniques applied to agriculture are oriented towards sustainability since they focus on the development of products or innovations that reduce the pollutants generated by this productive sector. This may be due to the indiscriminate use of chemical fertilizers and insecticides or to the extensive generation of residues and leachates that accumulate on the surface, in the air, and in bodies of water. Thanks to biotechnology and microbiology, it has been possible to develop bio-inputs, the purpose of which is to gradually replace the use of synthetic products to reduce pest populations and control the spread of diseases that affect crops of agricultural interest. In addition to the use of waste from various sectors to incorporate them into what is known as the circular economy. However, it is still necessary to continue with research to incorporate potential species in the formulation of bioproducts, characterize them, and define their interaction with their environment to determine the degree of stimulation of reactions they cause in plants or other microorganisms, whether beneficial or pathogenic. It is also necessary to deepen research that contemplates meta-omics and bioinformatics with impact and improvement of agricultural activities.
Consent for Publication
All authors have read and approved the final manuscript and have given their consent for publication.











 nueva página del texto (beta)
nueva página del texto (beta)